Part 3: Policies for Data Sharing and Access

Kim Addresses Issues of Data Access and Restrictions
Having explored data repositories for their metadata requirements and studied the disciplinary standards offered by organizations such as the Digital Curation Center in the U.K., Kim has begun work on applying metadata standards for documenting Dr. Smart's data. Dr. Smart has also informed Kim that he intends to make the data publicly available. He instructs Kim to make certain there are no issues that would prevent broad sharing. Kim will be seeking guidance on the issues and questions that pertain to sharing and access to ensure that management of this research data does not create security or confidentiality breaches, or impinge on any intellectual property rights.

In this part, you will learn about the importance of providing safeguards for protection of confidential information and requirements for restricting data. You will also consider how and when access to your data will be provided, and to whom. We also touch on the importance of data citation.
3.1 Access and Availability of Data
A major reason why federal funding agencies require DMPs is to encourage researchers to think as early in the project as possible about how and when data will be shared and made available. Funding agencies expect you to be clear in the DMP about your approach or policy for sharing and giving access to your data. Sharing also makes possible reuses and repurposing of data, as explained later in this tutorial.
Ways of sharing data can vary. They depend on the research domain, on the availability of services, on the size of the data set, and other factors. Many of these are covered later in this tutorial in the section which addresses long-term preservation of data. Preserving data ensures long-term access to it. What you want to make sure your DMP states is that you will deposit your data in a repository - whether it's a repository dedicated to data from your research domain or an institutional repository that accepts data sets. Penn State has such a repository - ScholarSphere [2], about which you may learn more here [3], in the tutorial.
You should note in the DMP when data will be made available - i.e., during the project, or afterward? Will there be any embargoes? If so, why and for how long? Will data be shared indefinitely, or will there be temporal constraints?
Will all the data be shared, or only a portion? Have you adequately addressed levels of access, e.g., who is allowed to use the data, etc.? (You may already have addressed this in the section of the plan related to data access.)

Click to expand to provide more information
3. Policies for Access and Sharing
After publication of manuscripts based on the data we collect, we will share our data and metadata with the NutNet community via data updates sent annually as .csv files from the existing central relational database. Other NutNet users will need to contact Lind for access to the data.
We will also submit both of our datasets (abundance and stoichiometry) to the U of M Digital Conservancy, an archive for digital preservaton. Borer has access to this resource as a faculty member. This will occur within a year of publication. The data will be publicly available via the Digital Conservancy, which provides a permanent URL for digital documents.
In DMPs, researchers often state that their findings will be published as articles and other genres relevant to their particular disciplines. Increasingly, publications are linking to data sets that have been deposited into repositories; this is another reason why depositing your data into repositories is important.
The NSF and other funding agencies that require DMPs now frown on the previously common practice of sharing data sets upon request, such as via email. Researchers' email addresses are not permanent, for one thing, and on-demand access unnecessarily burdens the researcher with the redundant act of locating and attaching, or pointing to, the requested data set. By submitting the data set to a repository, data become publicly discoverable, findable, and accessible.
While typically not mentioned in the DMP, the citation of data marks another method of sharing data and attributing the researchers who created them. The organization DataCite [5] has examples of citing data [6]. The most crucial component of such a citation is a Digital Object Identifier or DOI [7]. DOIs are assigned to ensure persistent access. Even though the digital object may undergo changes over time, the DOI stays the same.
Another benefit to researchers of having your data made available in a repository is that it becomes more widely accessible and citable. Citation of data is increasingly required by editors and having a permanent URL or repository identifier helps others find your data, as Andrew Stephenson describes in the following video:
Making publicly funded data available is a common good, however, there are instances where it is important to restrict data. Thus, in a DMP researchers should note what data will be shared and whether there are any restrictions to sharing.

3.2 Confidentiality and Ethics Issues
Confidential data usually includes personally identifiable information (PII), such as names, addresses, and Social Security numbers - anything that might point to the identity of a person. Health information data, for example, is protected under the Health Information Privacy and Accountability Act (HIPAA), because it is considered private data.

Data collection stemming from human participant research - such as survey data - and thus requiring approval from an Institutional Review Board, or IRB, typically produces confidential data, access to which must be restricted because of PII. Sharing such data would be a violation of both research ethics and human subjects' privacy.
If your research data encompasses any of the above ethics and confidentiality concerns, then you should note these in the DMP. If your project will be generating survey data, then you should also state an intention to comply with Penn State's IRB requirements set by research administration guidelines and policies. Consult Penn State's Human Subjects Research (IRB) site [11]. Review RA 22: HIPAA and Research at Penn State University [12], or, if applicable, RA 23: HIPAA and the Milton S. Hershey Medical Center and Penn State College of Medicine [13].
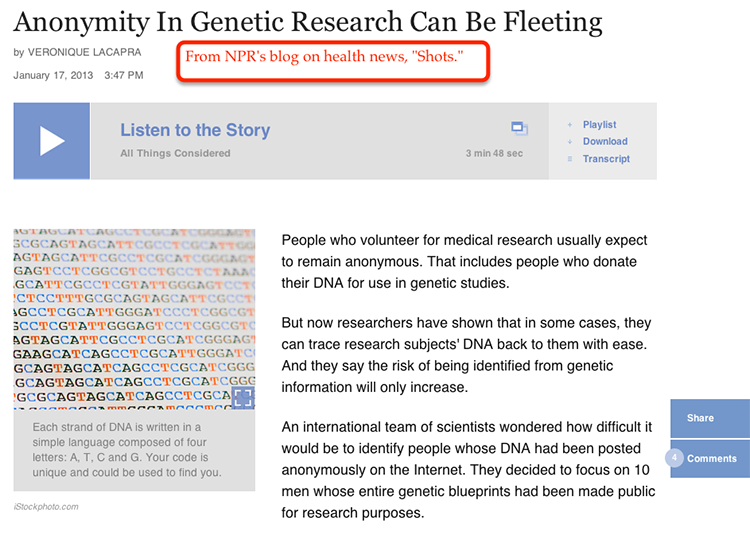
3.3 Intellectual Property, Embargoes, and Protections
One way to help ensure that confidential data, or data related to patented research, for example, is not shared prematurely is to implement an embargo. Embargoes are intended to bar data sharing and access for a limited period of time, thus protecting the data and the intellectual property rights of the researchers. Typically renewable, an embargo allows the researcher to decide when data may be released. The DMP should state whether an embargo will be imposed on the collected data or not.

Most universities provide guidance and policies on intellectual property rights for research. Penn State does at the General University Reference Utility (GURU) site [16]. It advises on ownership and management of intellectual property (Policy IP01 - Ownership and Management of Intellectual Property [17]), as well as guidance on the intellectual property rights of students (Guideline IPG01 - Faculty Guidance on Student Intellectual Property Rights [18]), and (Guideline IPG02 - Special Student Intellectual Property Agreement Forms [19]),While a DMP does not ordinarily contain this depth of information, it is a recommended practice for researchers to review their institutions’ policies and guidelines on intellectual property rights.

In IRB-approved research, an informed consent agreement between the researcher and the study participants is de rigeur. Depending on the nature and sensitivity of the data, however, researchers may wish to consider including an option in the consent form that allows data sharing. Refer to the guidance provided by the UK Data Archive [21] on informed consent forms, as suggested by the MIT Libraries [22].
Finally, in this section of the proposal, where data protections, sharing, and access are discussed, it's important to state outright who on the project will be responsible for monitoring embargoes, consent forms, and non-disclosure/confidentiality agreements. A key part of managing research data is being explicit about who will adopt what role in such management.
Archivists receive formal training related to these issues. Ben Goldman shares some of his expertise regarding security and access to digital data in the video below.
3.4 Summary
While funding agencies created the DMP requirement to encourage sharing of data, there are data produced on funded research projects that are sensitive and need to be restricted for access and use. These kinds of data tend to include personally identifying information (PII), such as might be collected as the result of demographic surveys or those found in health records. Often such data are protected under federal law, such as the Health Information Privacy and Accountability Act (HIPAA) [10].
If the data resulting from your project are likely to have confidentiality or ethics issues (such as human subject research engenders), then these should be noted in your DMP. Be sure to consult Penn State's Human Subject Research (IRB) site, as well as RA22: HIPAA and Research at Penn State University [12]. If applicable, review RA 23: HIPAA and the Milton S. Hershey Medical Center and Penn State College of Medicine [13].
There can be intellectual property rights issues as well, particularly with patent-pending research, that can prevent the sharing of data for a period of time. In such situations, an embargo may need to be implemented for your data, and you should state such in your DMP, specifying when the embargo will be lifted.
A DMP does not need to go into detail regarding confidential data and intellectual property rights, beyond stating what kind of restrictions on access are likely. For guidance during a project, Penn State provides faculty, students, and staff with research guidelines and policies [20] that can be helpful to draw on.
Finally, if possible, the DMP should state whose role it will be to monitor embargoes, consent forms, non-disclosure/confidentiality agreements, and so on.

Check Your Understanding
Which of the following types of data would be considered confidential?
(a) Social Security numbers
(b) Student grades
(c) Personal medical information
(d) All of the above
 Click for answer.
Click for answer.ANSWER: (d) All of the above. Confidential information is often information that might lead to the identification of the human subject or to more information about the human subject who participated in a study. Special measures, such as redaction or anonymization, are typically taken to prevent breaches of confidence.