Module 5: Global Carbon Cycle
Module 5: Global Carbon Cycle
Video: Earth 103 Carbon Removal (1:20)
Good morning, we're here today at the Susquehanna Shale Hills Critical Zone Observatory. And all around me, processes are removing carbon dioxide from the atmosphere. Above me, in the trees, photosynthesis is occurring, and that process removes CO2 from the atmosphere and converts it to oxygen that we breathe. If it weren't for photosynthesis, the earth would not be habitable. And it's going to be incredibly important moving forward, that we preserve the forests of the world, because of their role in photosynthesis and removal of CO2. There's another process that's going on near me here in Shale Hills, and that process is called weathering. Weathering is a much slower process that includes the breakdown of rocks below the surface by chemicals and bacteria. And the mixture of those breakdown products, with the degradation of organic material that's produced by trees and plants, and the mixture of the degraded organic material and the minerals that are produced by weathering of rocks, produces soil. And you can see a beautiful example of a soil profile right behind me. So, in this module, we're going to learn, in detail, what happens to the CO2 that humans put into the atmosphere and what will happen in the future as we continue to put more CO2 in.
Introduction
In summer 2019, the Brazilian Amazon rainforest was on fire. By far the largest rainforest on Earth, the Amazon is often called the lungs of the planet for its role in replacing carbon dioxide with oxygen. The fires are a double whammy: The fires are emitting large quantities of CO2 warming the planet, at the same time the removal of forest reduces the long term ability of this vital “scrubber” of CO2 which will lead to warming in the future.
Only ten years ago, there was a global effort to preserve the rainforest, home to a tenth of the world's species, and a vital part of the climate system due to its role in sequestering CO2. This changed with the election of President Jair Bolsonaro in 2019, and his government is now turning a blind eye to logging in the forest combined with burning to make new agricultural land, and the clearing has been focused in indigenous territory.
There were 39,000 wildfires in the Amazon from January through August 2019, a 77% increase over the same time in 2018. Smoke filled the air in cities and the is clearly visible on satellite images. The impact won’t be felt for a while, but the scale and significance of the Amazon in the carbon cycle are sure for there to be a serious cost in the future of atmospheric CO2 and warming. Now the policies will change with the election of President Lula da Silva in 2022 who has vowed to protect the Amazon.

Carbon is unquestionably one of the most important elements on Earth. It is the principal building block for the organic compounds that make up life. Carbon's electron structure gives it a plus 4 charge, which means that it can readily form bonds with itself, leading to a great diversity in the chemical compounds that can be formed around carbon; hence the diversity and complexity of life. Carbon occurs in many other forms and places on Earth; it is a major constituent of limestones, occurring as calcium carbonate; it is dissolved in ocean water and fresh water; and it is present in the atmosphere as carbon dioxide, the second most voluminous greenhouse gas and the trigger for the bulk of current global climate change.
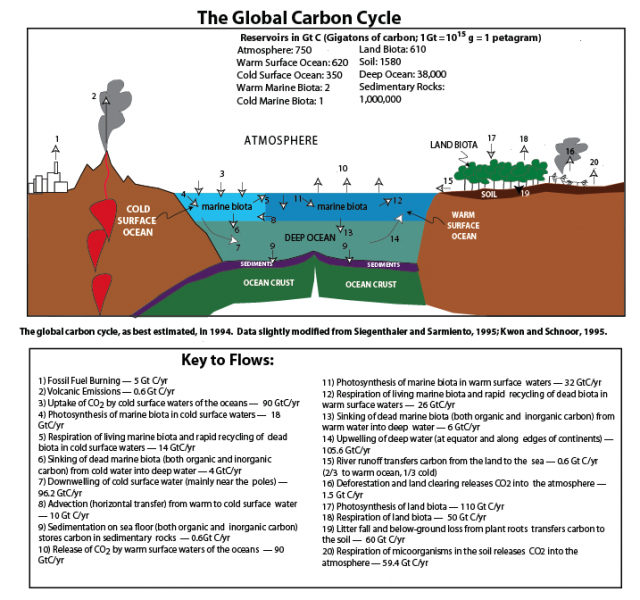
The flow of carbon throughout the biosphere, atmosphere, hydrosphere, and geosphere is one of the most complex, interesting, and important of the global cycles. More than any other global cycle, the carbon cycle challenges us to draw together information from biology, chemistry, oceanography, and geology in order to understand how it works and what causes it to change. The major reservoirs for carbon and the processes that move carbon from reservoir to reservoir are shown in the figure below. You do not need to understand this figure yet, but just appreciate that there are many reservoirs and a lot of exchanges. The carbon cycle is anything but simple! We will discuss these processes in more detail, and then, we will construct and experiment with various renditions of the carbon cycle, but first, we will explore some of the history of carbon cycle studies.
The global carbon cycle is currently the topic of great interest because of its importance in the global climate system, and also because human activities are altering the carbon cycle to a significant degree. The potential effects of human activities on the carbon cycle and the implications for climate change were first noticed and studied by the Nobel Prize-winning Swedish chemist, Svante Arrhenius, in 1896. He realized that CO2 in the atmosphere was an important greenhouse gas and that it was a by-product of burning fossil fuels (coal, gas, oil). He even calculated that a doubling of CO2 in the atmosphere would lead to a temperature rise of 4-5°C -- amazingly close to the current estimates obtained with global, 3-D climate models that run on supercomputers. This early recognition of human perturbations to the carbon cycle and the climatic implications did not raise many eyebrows at the time, but humans' "experiment" inputting massive amounts of CO2 to the atmosphere was just beginning then. We will be referring to this "experiment" throughout the module.
Goals and Learning Outcomes
Goals and Learning Outcomes
Goals
On completing this module, students are expected to be able to:
- explain the different components of the marine and terrestrial carbon cycle;
- recognize that atmospheric CO2 has changed through time and how that variation has impacted climate;
- interpret how the carbon cycle impacts Earth's climate;
- project through modeling how the amount and rate of future carbon emission impact climate and the chemistry of the oceans.
Learning Outcomes
After completing this module, students should be able to answer the following questions:
- Why is the global carbon cycle important to climate change?
- How have humans perturbed the global carbon cycle?
- What are the changes in values of pCO2 since (1) 1900 and (2) the late 1950s?
- What is the current pCO2 value?
- What is the significance of the annual cycle in atmospheric CO2 concentrations?
- What is the correlation of CO2 concentrations with glacial cycles?
- Generally speaking, what are the volumes of the different carbon reservoirs?
- What is the role of photosynthesis and CO2 fertilization?
- What is the role of respiration and permafrost?
- How does the air sea exchange work?
- What is marine carbonate chemistry?
- What are the biologic pump, upwelling, downwelling and sedimentation; explain their role in the marine carbon cycle.
- What are the main types of fossil fuels and their relative contribution to recent CO2 increases?
- What are the big polluters (countries) in total amount and per capita?
- What are the volumes of carbon and their trajectories for the different emissions scenarios?
- What is the role of deforestation and soil erosion in pCO2 changes?
Assignments Roadmap
Below is an overview of your assignments for this module. The list is intended to prepare you for the module and help you to plan your time.
| Action | Assignment |
|---|---|
| To Do |
|
Carbon Dioxide Through Time
Carbon Dioxide Through Time
In the late 1950s, Roger Revelle, an American oceanographer based at the Scripps Institution of Oceanography in La Jolla, California began to ring the alarm bells over the amount of CO2 being emitted into the atmosphere. Revelle was very concerned about the greenhouse effect from this emission and was cautious because the carbon cycle was not then well understood. So, he decided that it would be wise to begin monitoring atmospheric concentrations of CO2. In the late 1950s, Revelle and a colleague, Charles Keeling, began monitoring atmospheric CO2 at an observatory on Mauna Loa, on the big island of Hawaii. Mauna Loa was chosen because its elevation and location away from industrial centers made it as close to a global signal as any other location. The record from Mauna Loa, one of the most classic plots in all of science, shown in the figure below, is a dramatic sign of global change that captured the attention of the whole world because it shows that this "experiment" we are conducting is apparently having a significant effect on the global carbon cycle. The climatological consequences of this change are potentially of great importance to the future of the global population. The CO2 concentration recently crossed the 400 ppm mark for the first time in millions of years! In 2022, the yearly average was 417 ppm (check that number with the curve below!).
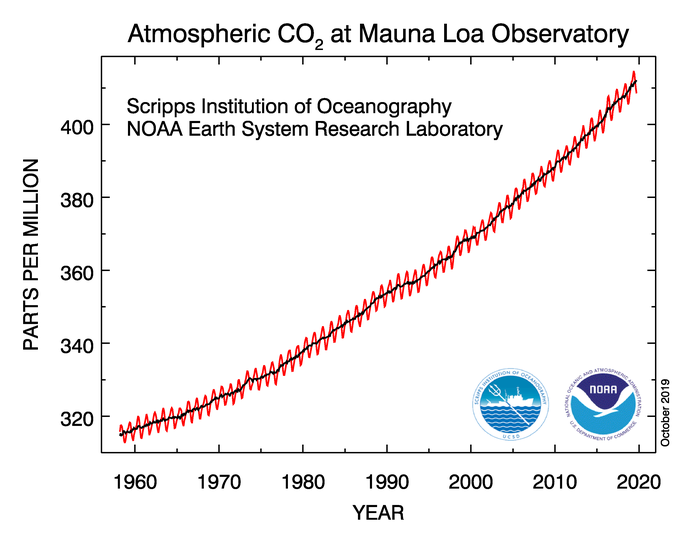
As the Mauna Loa record and others like it from around the world accumulated, a diverse group of scientists began to appreciate Revelle's concern that we really did not know too much about the global carbon cycle that ultimately regulates how much of our CO2 emissions stay in the atmosphere.
The importance of present-day changes in the carbon cycle, and the potential implications for climate change became much more apparent when scientists began to get results from studies of gas bubbles trapped in glacial ice. As we learned in Module 1, the bubbles are effectively samples of ancient atmospheres, and we can measure the concentration of CO2 and other trace gases like methane in these bubbles, and then by counting the annual layers preserved in glacial ice, we can date these atmospheric samples, providing a record of how CO2 changed over time in the past. The figure below shows the results of some of the ice core studies relevant for the recent past -- back to the year 900 A.D.
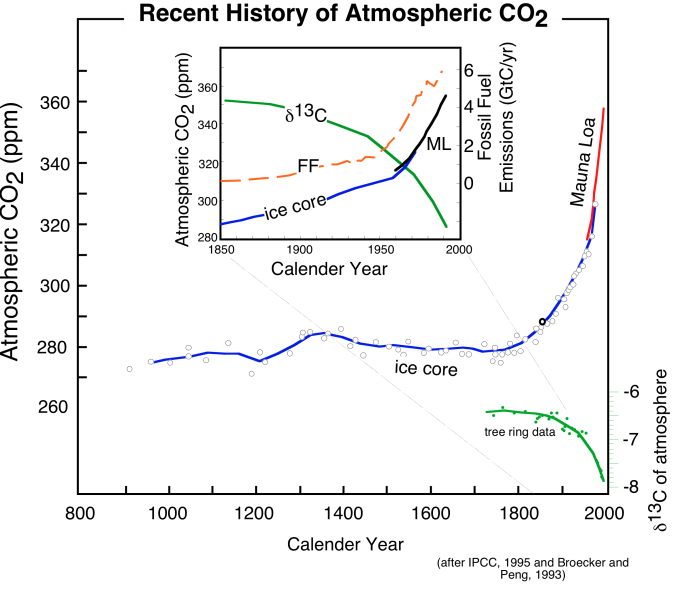
The striking feature of these data is that there is an exponential rise in atmospheric CO2 (and methane, another greenhouse gas) that connects with the more recent Mauna Loa record to produce a rather frightening trend. Also shown in the above figure is the record of fossil fuel emissions from around the world, which show a very similar exponential trend. Notice that these two data sets show an exponential rise that seems to begin at about the same time. What does this mean? Does it mean that there is a cause-and-effect relationship between emissions of CO2 and atmospheric CO2 levels? Although we should remember that science cannot prove things to be true beyond all doubt, it is highly likely that there is a cause-and-effect relationship -- it would be an extremely bizarre coincidence if the observed rise in atmospheric CO2 and the emissions of CO2 were unrelated.
How serious is our modification of the natural carbon cycle? Here, we need a slightly longer perspective from which to view our recent changes, so we return to the records from ice cores and look deeper and further back in time than we did in the figure we have been examining.

In addition to providing a record of the past concentration of CO2 in the atmosphere, as we learned in Module 1, the ice cores also give us a temperature record. By studying the ratios of stable isotopes of oxygen that make up the glacial ice, we can estimate the temperature (in the region of the ice) at the time the snow fell (glacial ice is formed by the compression of snow as it gets buried to greater and greater depths). From these data, shown in the figure below, we can see the natural variations in atmospheric CO2 and temperature that have occurred over the past 160,000 years (160 kyr).
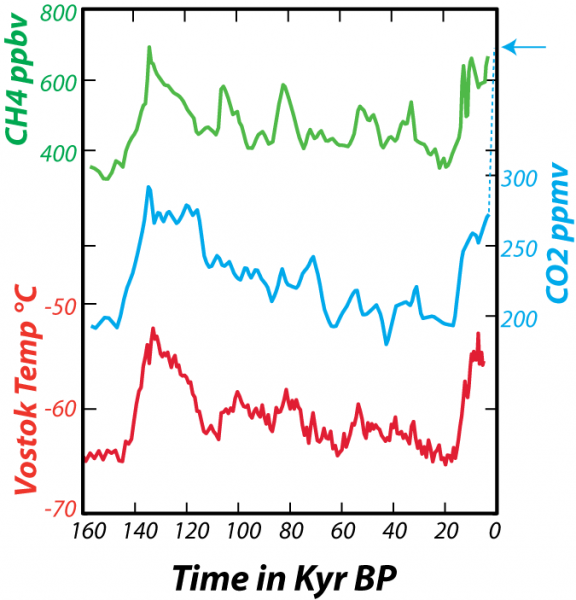
In fact, looking at this much longer span of time enables us to clearly see that the present CO2 concentration of the atmosphere is unprecedented in the last several hundreds of thousands of years. As geoscientists, we are interested in more than just the last few hundred kiloyears, and so we look back into the past using sediment cores retrieved from the deep sea. Geochemists studying these sediments have been able to reconstruct the approximate concentration of CO2 in the atmosphere and the sea surface temperature (SST).
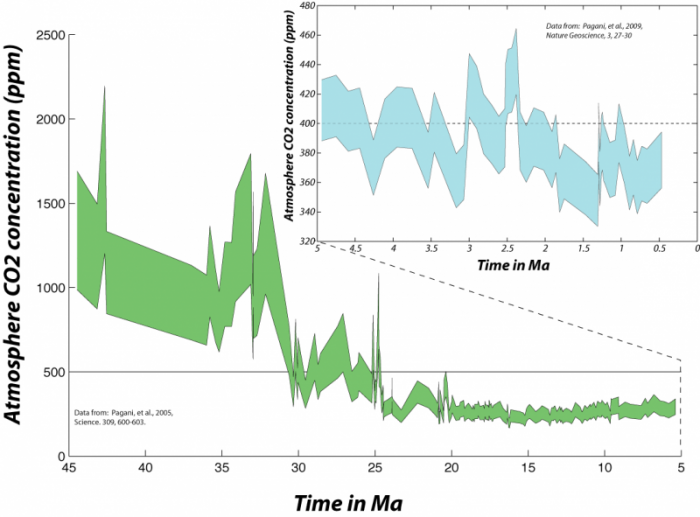
To find atmospheric CO2 levels equivalent to the present, we have to go back 2.5 million years. This means that, to the extent that the state of the carbon cycle is closely linked to the condition of the global climate, we are pushing the system toward a climate that has not occurred any time within the last several million years -- not something to be taken lightly.
The farther back in time we go, the more difficult it is to figure out how CO2 concentrations have changed, but that has not stopped some from attempting:
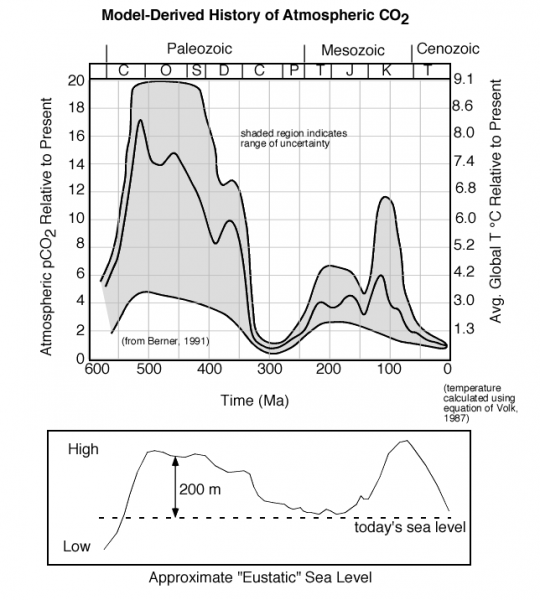
One thing that is clear is that further back in time, CO2 levels have been much, much higher, and the average global temperatures have also been much higher. Why does the CO2 concentration change so much? This is a big question whose answer involves many factors, but consider two that are relevant to what we'll learn about in this module. Photosynthesis only started in the Silurian (S on the timescale in the figure above), and photosynthesis is a major sink or absorber of atmospheric CO2. Sea level was much higher during the two big peaks in CO2 — this leaves less room for photosynthesis and it also decreases the planet's albedo, making it warmer. A warmer ocean cannot absorb atmospheric CO2 and instead, it releases it to the atmosphere.
In conclusion, from this brief look at the record of fossil fuel emissions and atmospheric CO2 concentrations, it is clear that we have cause for concern about the effects of the global CO2 "experiment". Because of this concern, there is a tremendous effort underway to better understand the global carbon cycle. In the remainder of this module, we will explore the global carbon cycle by first examining the components and processes involved and then by constructing and experimenting with a variety of models. The models will be relevant to the dynamics of the carbon cycle over a period of several hundred years -- these will enable us to explore a variety of questions about how the system will behave in our lifetimes and a bit beyond.
Check Your Understanding
Overview of the Carbon Cycle from a Systems Perspective
Overview of the Carbon Cycle from a Systems Perspective
The global carbon cycle is a whole system of processes that transfers carbon in various forms through the Earth’s different parts. The carbon that is in the atmosphere in the form of CO2 and CH4 (methane) doesn’t stay in the atmosphere for long — it moves from there to other places and takes different forms. Plants use the CO2 from the atmosphere in photosynthesis to make carbohydrates and other organic molecules, and from there it may return to the atmosphere as CO2, or it may enter the soil as still different compounds that contain carbon. Some carbon is deposited in sedimentary rocks from the oceans, and much later, this carbon may be released to the atmosphere. So, carbon moves around — it flows — from place to place.
Because CO2 is such an important greenhouse gas, the way the carbon cycle works is central to the operation of the global climate system. Later in this module, we will work with a computer model of the carbon cycle to do experiments that will help us understand how it works, but it will help to begin with an overview of the carbon cycle from the systems perspective.
What is meant by a systems perspective? It just means that we focus on the places where carbon resides (the reservoirs, in systems terminology), how it moves from reservoir to reservoir, how much of it moves from place to place, and what controls those movements. This same perspective is behind the simple climate model we worked on in Module 3.
First, let’s consider the main reservoirs of carbon. These can be seen in the diagram below, where each box represents a different reservoir, and remember that in each of them, the carbon may be in very different forms. Note a GT or gigaton is a billion metric tons or 1015 grams which is a whole lot of carbon!
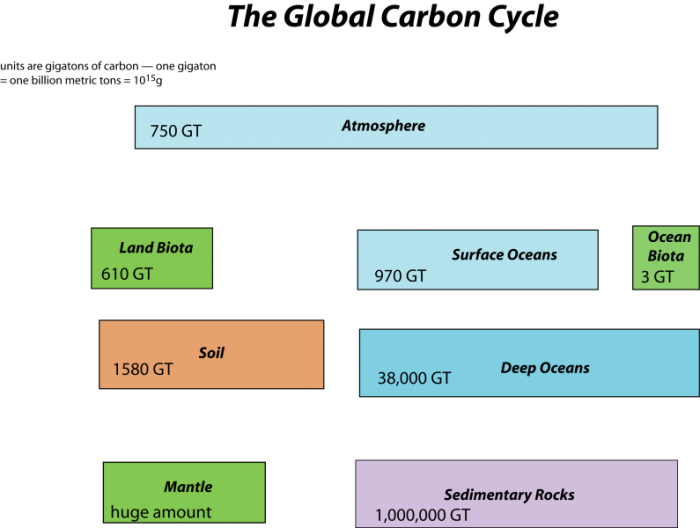
The mantle reservoir is huge and somewhat removed from the other reservoirs, thus we will not really bother with it. Among the other reservoirs, you can see that there is a huge range in the sizes. The ocean biota contain a very small amount of carbon relatively speaking, while sedimentary rocks contain a vast quantity (in the form of calcite — CaCO3 — that forms limestones, and coal, petroleum, etc.).
Now, let’s look at the system with the flows included, as arrows connecting the reservoirs:
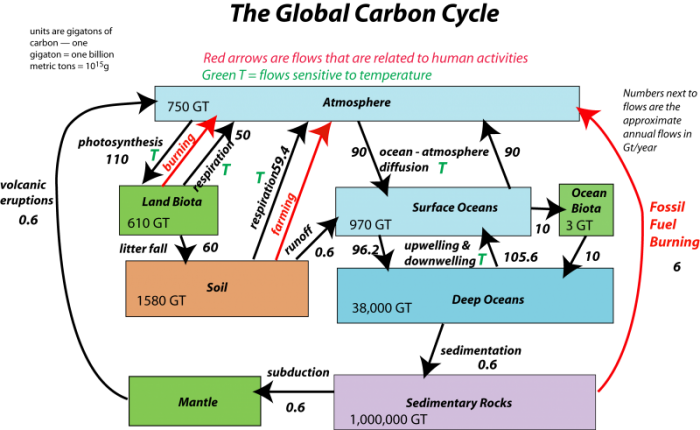
The black arrows represent natural processes of carbon transfer, while the red arrows represent changes humans are responsible for. The magnitudes of the flows are shown in units of gigatons of carbon per year. The diagram as constructed here represents a steady state if we just consider the black arrows; the flows going into each reservoir are equal to the flows going out of the reservoir — in other words, there is a balance. We will step through this system, talking about the processes involved in the flows, but first, let’s try to learn something from the numbers themselves in this diagram.
First, just a bit more on the notion of steady state. The diagram below illustrates some simple systems, one of which is not in a steady state, and the others of which are.
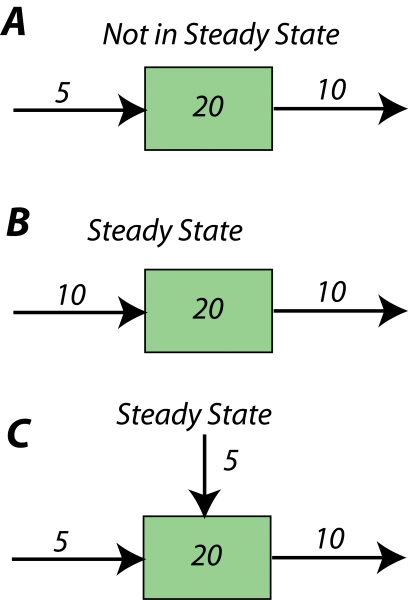
In the first example, A, more (10 units per time) is subtracted from the reservoir than is added (5 units per time), and so over time, the amount in the reservoir will decline — it will not remain constant, or steady. In each time step, it will lose 5 units of whatever the material is. In the other two (B,C), the amount added is the same as the amount subtracted, so these reservoirs will be in a steady state. If you look at example C, you see that the sum of inflows (5+5) is equal to the outflow (10).
When a system is in a steady state, we can say something about the average time something will spend in the reservoir — this is called the residence time for the reservoir. Here is a simple example — if there are 40,000 students at Penn State, and 10,000 students enter each year and 10,000 students graduate each year, then the system is in a steady state. The residence time is the total number of students divided by either the number entering or graduating each year — this give 4 years as the average residence time. Here is a figure to explain the idea further:
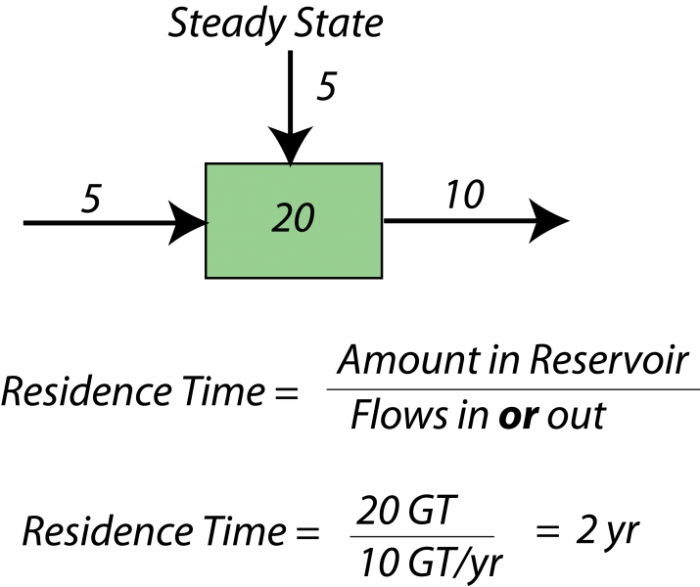
The concept of residence time is a useful one in studying any kind of system because it tells us something about how quickly material is moving through a system, and more important, it tells us how quickly some part of the system, or the system as a whole, can respond to changes. If something has a short residence time, it can respond quickly to changes, whereas if it has a long residence time, it responds very slowly. Mathematically, the residence time is the same as something called the response time which, as the name implies, is a measure of how much time it takes for the system to respond to a change. The word "respond" in this context means "return to a steady state."
Although the residence time and the response time are often the same value, they represent different ideas. As we said earlier, the residence time is a measure of the average length of time something spends in a reservoir — like the average length of time a carbon atom spends in the atmosphere. The response time, instead, is a measure of how quickly something returns to steady state after some disturbance that knocks it out of steady state. So, response time is only meaningful in cases where a system has a tendency to remain in a steady state.
Now, let’s turn to the carbon cycle and consider some of the flows in and out of the reservoirs. What is the residence time for the atmosphere? To get this, we take the amount in the reservoir (750 GT) and divide it by the sum of the inflows or the outflows. Let’s take the outflows: 100 GT C/yr for photosynthesis + 90 GT C/yr going into the oceans. The residence time is thus:
750 GT/190 GTyr-1 = 3.9 years
This is a pretty short residence time. Now, let’s look at the deep ocean (which is the vast majority of the oceans) — its residence time is:
38000 GT/10 GTyr-1 = 3,800 years
We use 10 for the inflow/outflow value because we use the net of the water flux into and out of the deep ocean. The result, 3,800 years, is much longer than the atmosphere, and what this means is that the carbon cycle has some parts that respond quickly, but other parts that respond very slowly, and the very slow parts tend to put a damper on how quickly the other parts can change. In other words, if we suddenly inject carbon dioxide into the atmosphere, you might think that the short residence time of the atmosphere means that the excess CO2 can be removed very quickly, but because these reservoirs are linked together, it turns out that the deep ocean must return to its steady state before the atmosphere can get back to its steady state.
Check Your Understanding
A Simple Analogy
A Simple Analogy
We're going to take a step backwards for a second and think about a much simpler system, but one that has some things in common with the global carbon cycle. First, however, we need to recognize that the natural carbon cycle is something that always varies a bit, but it has some important feedbacks in it that tend to make it stable or steady. And then, along come humans, burning an impressive amount of fossil fuel and creating a new flow in the carbon cycle. To get a sense of how a system with a tendency to remain in a steady state might respond to a new flow, we turn to a simpler model.
Our simple model is a water tub with a drain and two faucets.
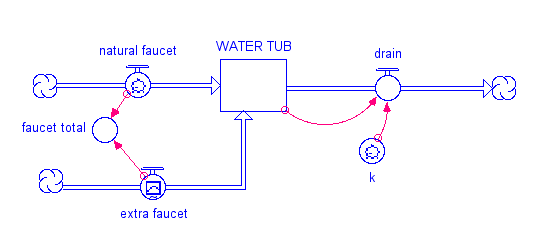
One faucet represents the natural addition of water to the tub, while the other represents a new flow. The tub has a drain in it, and it takes out 10% of the water in a given time interval (the value of k is 0.1 and the drain flow is just the amount in the tub times k). When you first open the model, the extra faucet is set to zero, and if you run the model you will see that it is in a steady state — the faucet and drain are equal, so the amount in the reservoir remains constant. What happens to the system if you increase the faucet flow using the upper knob to the right of the graph? If the faucet flow increases, then at first it will be greater than the drain and the amount in the tub will increase; but as faucet flow increases, so will the drain flow, and it will eventually become equal to the faucet flow and the system will return to steady state, but one with more water in the tub. Give it a try, and see what happens. At steady state, you can write a simple equation that says:
F = k*W
It turns out that the response time for this system is just 1/k. In this example model, k = 0.1 so the response time is 10 time units (the units depend on how the flows are given — liters per second or minute). This response time is not the length of time required for the system to get into a steady state — it is the time required to accomplish 63%, or the change to the new steady state. Why 63%? There is some math behind this choice, but in essence, it is just a convention that helps us avoid the problem of picking the time when steady state is achieved, which is difficult since it does so asymptotically. The figure below shows how this looks. In the graphs below, the blue line for the water tub is hidden beneath the red curve of the drain — these have different scales on the left hand side, but they are the same exact shape, so one plots over the other.
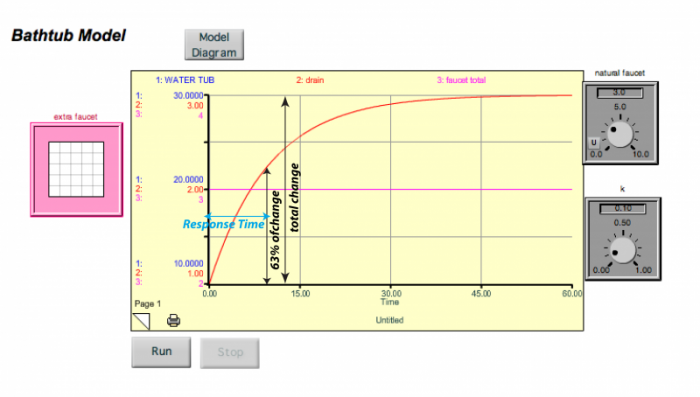
I changed the faucet here to 3, and the water in the tub rose until it reached a value of 30, where the drain value then became equal to the faucet value and the system reached a new steady state. As shown by the blue arrow above, this takes 10 time units to accomplish 63% of the change.
So, this is a system that has negative feedback (the drain) that drives it to a steady state, which means that regardless of the values of the faucet or the drain constant, k, the system will find a steady state.
Now, look at what happens if we turn on a new faucet — add a new flow to a system that is in a steady state. In the figure below, you see what happens if we turn the extra faucet on for a bit and then turn it off.
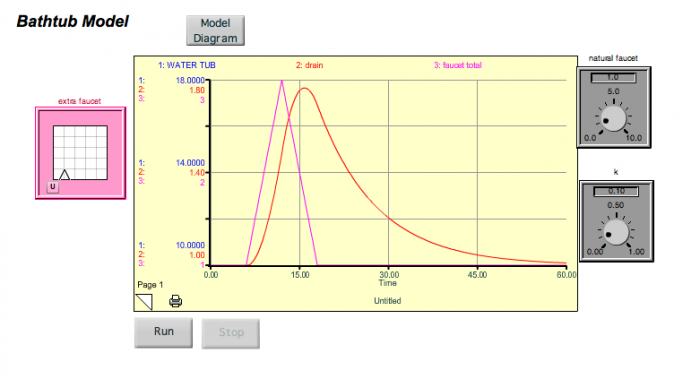
The system gets thrown out of its steady state temporarily, but returns to the original steady state when the extra faucet is turned back off. Note that there is a lag time here — the water tub peaks about 5 time units after the faucet peaks (this is the sum of the two faucets). If we were to decrease k, then the response time would lengthen, and the lag time would also lengthen. Think of this spike in the extra faucet as being equivalent to a short-term addition of CO2 into the atmosphere. But what if we turn the extra faucet on and then leave it on for some time at a steady rate? This might be equivalent to us adding CO2 to the atmosphere and then keeping those emissions constant (sometimes referred to as the "stabilization" of emissions). We can simulate this scenario with our simple model, and the results are shown below.
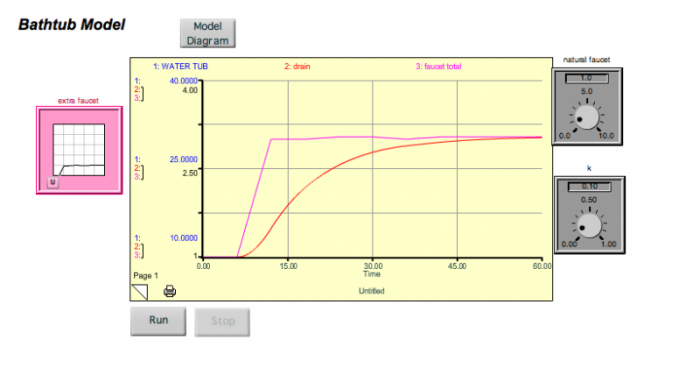
What you see is that the system responds by increasing the water in the tub until a new steady state is reached. The length of time needed to achieve the new steady state is determined by the response time of the system, which again, is governed by the magnitude of the drain constant, k.
In the real carbon cycle, this response time is measured in tens of thousands of years. For example, remember that the residence time of the deep ocean is about 3,800 years. The response time of the whole carbon cycle must be much longer than this because CO2 emissions are cycled through more than just the deep ocean. Unfortunately you can't add up the residence time of the individual reservoirs to get the response time of the whole carbon cycle which is really what we want to know, the system is much more complex than this. The natural carbon cycle will find a new steady state (it will "stabilize") in response to our carbon emissions, but it will take many thousands of years to do so. In the meantime, the system will continue to change as it makes this adjustment.
The Terrestrial Carbon Cycle
The Terrestrial Carbon Cycle
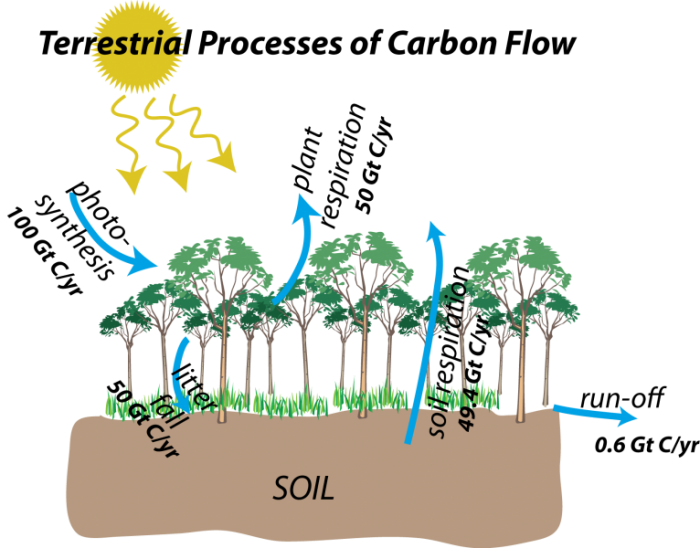
Carbon moves through the terrestrial realm through five main processes, which are represented as blue arrows in the figure below:
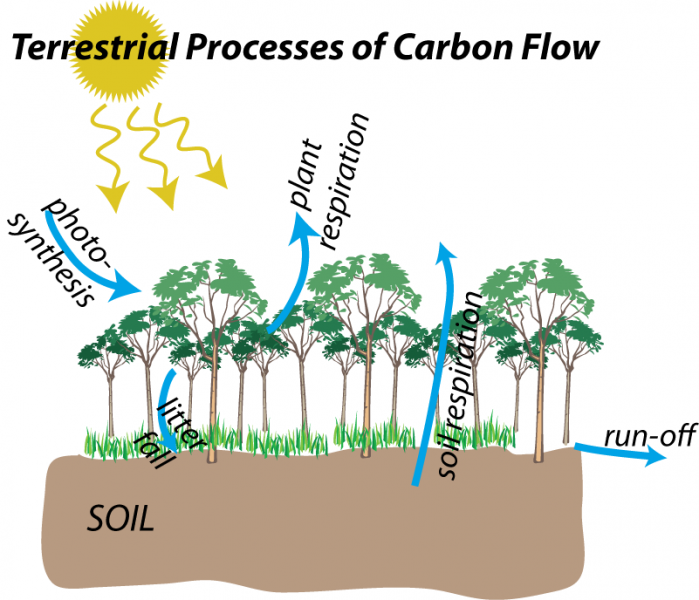
We will briefly explore these processes, beginning with photosynthesis.
Photosynthesis
Photosynthesis
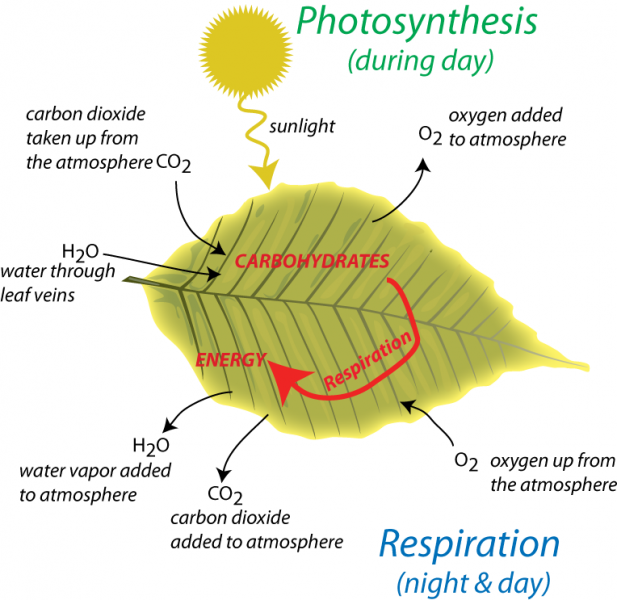
Since its origin over 3 billion years ago, photosynthesis has been one of the most important processes on Earth, helping to make our planet habitable, in stark contrast to the other planets. The basic idea is that plants capture light energy and use it to split water molecules and then combine the products with carbon dioxide to make carbohydrates, which are used for fuel and construction of plants; oxygen which is crucial to making Earth habitable, is a by-product of this reaction, which is summarized as:

This process takes places in the chloroplasts located in the interiors of leaves. Here, chlorophyll absorbs solar energy in the red and blue parts of the spectrum. This energy is then used to split a water molecule into hydrogen and oxygen; in the process, the plants gain chemical energy that is used in a companion process that converts carbon dioxide into carbohydrates represented by C6H12O6 in the above equation.
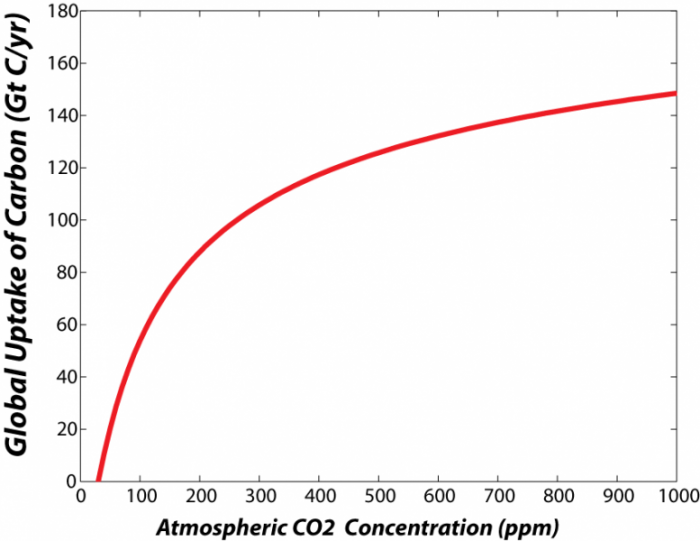
The rate of consumption of CO2 by photosynthesis is mainly a function of water availability, temperature, the concentration of CO2 in the atmosphere, and key nutrients such as nitrogen. The importance of water in plant growth is obvious from looking at the equation above. Temperature is an important factor in many life processes, and photosynthesis is no exception. As a general rule, the rates of most metabolic processes increase with temperature, but there is usually an upper limit where the high temperatures begin to destroy important enzymes, or otherwise inhibit life functions. The fact that photosynthesis depends on the concentration of CO2 is not obvious, but it is very important. Plants take in their CO2 through small openings about 10 microns in diameter called stomata, which the plant can control like valves, opening and closing to adjust the rate of transfer. The more they let in, the faster the rate of photosynthesis and the faster the growth — but if they open their stomata wide to let in a lot of CO2, they can lose a lot of water, which is not so good. However, if there is a greater concentration of CO2 in the atmosphere, then the plants will get a good dose of CO2 by opening their stomata just a little bit, allowing them to conserve water. What this amounts to is increased efficiency of growth at higher levels of CO2. We call this effect CO2 fertilization, and it is an important way in which plants are our friends in helping to minimize the rise of CO2 in the atmosphere. You can see this effect in the graph below, which shows the theoretical relationship between the CO2 concentration in the atmosphere and the uptake of carbon from photosynthesis by land plants, summed up for the whole globe.
The following video explains photosynthesis in great detail:
Video: Photosynthesis (12:26)
Hi, it's Mr. Andersen, and in this podcast, I'm going to talk about photosynthesis. I love photosynthesis because it gives me two things that I need. I need to breathe, so it gives me oxygen. And I need to eat, and so, it's going to give me food. And so, I love photosynthesis. You might think it's only found in these things, plants. But it's also found in bacteria. It's found in algae, and so, it's found in protists, it is found everywhere. And so, photosynthesis has been around a long time, and it’s super important that you understand how it works. So, let's start with the site in eukaryotic cells of photosynthesis, and that's the chloroplast. So, this is a number of cells, and you can see how many chloroplasts we could have in a typical cell, so there's a whole bunch of them. There are a few terms you should be familiar with and where they are. First one is a thylakoid membrane. Thylakoid membrane is going to be organized like this, and basically, that's where the light reaction is going to take place. If you got a stack of thylakoids, like this together, we call that a granum. The other big thing to understand photosynthesis is that this is filled with a liquid, and that liquid is called the stroma. That's going to be the site of the Calvin cycle. If we were to grind up a leaf, what we would find is that there's not only one pigment, chlorophyll A, that does photosynthesis, but there's a number of them that are working together. And so, if you grind up a leaf into some chromatography paper, and then you put it in a solvent, what you'll get is chromatography. It's going to separate into all its different parts. And so, this right here would be chlorophyll A, and chlorophyll B, and this would be like carotene and xanthophylls, and they're all working together. You'll see these other pigments in the fall when the chlorophyll moves back into the leaf and is reabsorbed, but if we look at what light they absorb, here's chlorophyll A and here's B, this is what's called their absorption spectrum, what color of light they're able to absorb. And you can see that they absorb a lot of the blue, a lot of the red, but they don't absorb a lot of this in the middle, this green. And so, a question could be, what is their least favorite color, plants - and the right answer would be green because they reflect that green light. Now, this has actually puzzled scientists for a long time, and we really don't have a definitive answer as to why plants are green. Know this, that if they were black, they probably would get a little bit too hot, they would absorb too much light. And so, let's start with an equation, because this is simply a chemical reaction. It's a chemical reaction with a number of steps. But what are the reactants? Water and carbon dioxide. And so, how does a plant grow? It's basically taking water in from its roots and it's taking carbon dioxide in through its leaves, through its stomata. The other thing it needs is light. And so, it's just taking these simple ingredients, and then it's weaving those together into glucose, this monster molecule here, and then oxygen. And so, this is the food that I get and this is the oxygen that I breathe. Now, are plants just nice? No. They're making this sugar for themselves so they can break it down using cellular respiration. And in fact, if I put this arrow in the other direction, that becomes cellular respiration. So, they're making food for themselves, and they're also going to make some of the structures. So, like the cellulose in the cell walls of a plant is made from that, as well. Okay, so, whenever I try to think what are the different steps in photosynthesis, I always imagine this picture right here. There's photo and synthesis in the word. Photo means “light” and synthesis means “to make”. And so, there are two steps in photosynthesis. The light reaction and those are going to take place in the thylakoid membrane, and then the Calvin cycle. We used to call this the dark reactions, which is a silly term, doesn't happen during the dark, it happens during the light. And so, basically, the person who worked this all out is Melvin Calvin, and so, we named it after him. Where does this take place? You guessed it, it takes place in the stroma or this liquid portion. And so, let's kind of do a cartoon version of photosynthesis. What are the reactants again? Water, light, and carbon dioxide. What are going to be the products that come out of this? It's going to be oxygen and glucose. So, let's watch what happens. In the light-dependent reaction, water and light go into the thylakoid membrane, and they produce two things. They produce oxygen; oxygen is simply waste product. And then, they're going to produce these chemicals, NADPH and ATP, so they have energy now. Let's watch what happens to them. Well, the energy is going to transfer to the Calvin cycle, where carbon dioxide comes in, and then glucose goes out. And so, this is the big picture of photosynthesis. But now, let's kind of dig in a little bit deeper and talk about the light reaction. Okay, so, where are we? We’re in the thylakoid membrane, so we're in this membrane right here. So, if we were to zoom into that membrane right here, that's what this diagram is. Okay, so, what are the two things coming in? Well, the first one is going to be light. So, light's coming in here, light's coming in here. What's the next thing that we're going to have coming in? And that's going to be water. Okay, so, let's look at some of the other big features in this thylakoid membrane. So, this is the outside of the stroma, and this is going to be the lumen or the inside. And so, there's a couple of big things right here. What's in here? Well, these are basically going to be proteins with chlorophyll on the inside of it, and so, we call that whole thing together a photosystem. So, this first one is actually called photosystem 2 and then, we go to photosystem 1. And the reason we go backward is that photosystem 1 was discovered first. So, basically, what comes in? Light. What's that light used to do? Well, that light is used to power the movement of an electron through an electron transport chain. So, that electron is going through proteins, carrier proteins, and eventually that electron is going to go to here. It's going to go to NADPH because remember, that's one of the products of the light-dependent reaction. Okay, what happens to the water then? So, the water is going to be split right away. If you split water, what do you get? Well, you get oxygen, so, that's the O2 that’s going to diffuse out of a cell and that's the oxygen that you're actually breathing right now. And then, we're going to have these protons, which are simply hydrogen ions, so, they're hydrogen atoms that have lost their electron. Okay, so, this is getting kind of messy, so, let's look what happens next. As that electron moves through the electron transport chain, and again, it's powered by the introduction of light here and light here, that electron is going to be moving all the way down here. And every time it goes through one of these proteins, it's pumping protons to the inside, so it's pumping protons to the inside. Now, protons have a positive charge, so, basically, what's happening is that you're building up a positive charge on the inside, so, there's a positive charge in here. If you know how cellular respiration works, you'll realize that this is the opposite of that. So, now, we have all these positive charges on the inside. Where do they go? Well, there's only one hole that they can go through, and that is to go through this protein here. As those protons move out, they're moving through a protein called ATP synthase, and it works almost like a little rotor, and every time a proton goes through, we make another ATP. So, what have we made in the light-dependent reaction? We've made NADPH and we've made ATP. And what's nice about that is, they're now just sitting right here in the stroma and so, they're able to go on to the Calvin cycle, which is going to be the next step in this process. And so, who's providing the energy? Light. Who's providing the electrons? Water. And then, a base or a waste product to that is simply going to be oxygen. Okay, let's go to the Calvin cycle, then. So, what's happening in the Calvin cycle? You can see here's those reactants. So, we've got our ATP here, ATP here, and NADPH. What are they providing? Simply, energy. We also have this molecule here. It's called RUBP. Basically, it's a five carbon molecule. And then, we have carbon dioxide coming in. So, it moves through the stomata of the leaf and it's going to diffuse its way in. Carbon dioxide is a one carbon molecule, so, basically, there's an enzyme here called Rubisco and it's going to attach this one carbon molecule to a five carbon molecule. It immediately breaks into two, three carbon molecules and then it gets energy from ATP and NADPH. And when we're done, it's creating this chemical down here called G3P.What does G3P become? Well, it can be assembled quickly into glucose or sucrose or maltose or whatever they need to do, that's going to be produced right in here by the G3P. So, that's where we're synthesizing. In other words, we're taking carbon and we're fixing it. We're making it usable. Now, some of that G3P is released, but a lot of it is recycled again to make more of this RUBP, and so, that's why it's a cycle over and over again. What's the big picture? If we don't have ATP, if we don't have NADPH, then this process is going to shut down. What's the other thing that could shut it down? If we don't have carbon dioxide. Okay, so, that's basically photosynthesis, and again, it's been working for billions of years. But there's a slight problem, and that problem is called photorespiration. What is photorespiration? Well, photorespiration occurs only when we don't have enough carbon dioxide. So, if we don't have enough carbon dioxide (let me cross that out), well we certainly can't make our G3P, but something worse happens. Oxygen can actually jump into the Calvin cycle and using Rubisco, can form another chemical. Now, that chemical doesn't do anything. In other words, it has no purpose, and the cell actually has to break it down. And so, as a result of that, plants, and we call almost all plants C3 plants, and the reason we call them C3 plants is this G3P is going to be a three-carbon molecule. So, these C3 plants, photorespiration is bad. In other words, they don't get anything out of it. And so, they're going to lose based on that oxygen kind of jumping into the Calvin cycle. And so, you might think, evolutionarily why would this have even evolved? Well, remember photosynthesis shows up first and then oxygen in the atmosphere shows up much later, and so, it wasn't a problem initially, but it became a problem. Another question you might think is, well, when are we not going to have enough carbon dioxide? When wouldn't we have carbon dioxide? Well, how do they get carbon dioxide? A plant is going to have a stomata, and it's surrounded by guard cells. So, basically, when a plant opens up its stomata, carbon dioxide can diffuse in. And so, the only time the plant wouldn't have carbon dioxide, because we have tons of carbon dioxide in the atmosphere, is when it's actually closed. And when would it be closed in a plant? The only time it's closed is when it's really, really hot, and a plant doesn't want to lose water, because through transpiration you're constantly losing water. And so, if you're a plant, if it's a hot day, you have this really tough choice. If you open up your stomata, you're going to lose water; you could shrivel up. If you close it, you can't get carbon dioxide in, and then you're going to start doing photorespiration. And so, of course, nature has come up with solutions to this over time. And it's only going to be found in plants that live in a really hot environment. So, here's the first solution, and this totally makes sense. So, this is in CAM plants. CAM plants, an example would be a jade plant or like a pineapple. Basically, what they do is they only open their stomata at night. And so, at night, they open up their stomata, and then the carbon dioxide will come in, and they'll create malic acid out of it. So, they're going to store it in vacuoles inside the cell. Okay, so now, when it's daytime, what they can do is they can close the stomata because they don't want to lose water, and now, they can actually take that carbon dioxide out of the malic acid and they can use it in the Calvin cycle to make sugars. So, the great thing about a CAM plant is, again, they're only taking in carbon dioxide at night when it's cool. And then during the day, they can close their stomata and they don't lose water. Another example of this would be in C4 plants. What they do is, instead of doing it day and night, what they'll do is they'll take that carbon dioxide in and they'll actually use enzymes to make a four-carbon molecule out of it. That four-carbon molecule will move to some cells on the inside of the leaf, called the bundle sheath cells, and then they can simply introduce carbon dioxide into the Calvin cycle, here. And so, again, both of these solutions are basically taking in carbon dioxide when you can get it, creating a chemical out of it, and then they can introduce that chemical into the Calvin cycle, and they don't have to wait for carbon dioxide to diffuse in. Now, of course, there's going to be extra steps in here. So, it's going to require more energy and so, we only see this in areas where it's really, really, warm. But an example of a C4 plant that we all eat and use a lot of, in fact most of us are just made out of this stuff, is corn. And so, that's photosynthesis. A simple problem is photorespiration, but I hope that's helpful.
If the video does not play above, click here to be directed to the Photosynthesis video on YouTube [8].
Plant Respiration and Litter Fall
Plant Respiration and Litter Fall
If we think of photosynthesis as the process of making fuel (carbohydrates), then respiration can be thought of as the process of burning that fuel — using it for maintenance and growth. This process can be described in the form of a reaction, just like photosynthesis. The chemical reaction is just the reverse of photosynthesis.
Through respiration, plants (and animals) release water, carbon dioxide, and they use up oxygen. Do the carbon flows involved in respiration and photosynthesis balance each other, as the equations seem to imply? The answer is no — otherwise, how could organisms grow?
Experiments on a variety of plants indicate that the ratio of photosynthesis to respiration is generally about 2 to 1. When plants are young, and growing rapidly, but with not much biomass to maintain, this ratio is even higher; in older, larger plants, this ratio is lower since more carbon needs to go towards maintenance.
Litter Fall
Dead plant material enters the soil in two ways -- it falls on the surface as litter, and it is contributed below the surface from roots. The relative importance of these two pathways into the soil varies according to the plants in an ecosystem, but it appears that the two are commonly about equal, which may seem a bit surprising since loss of organic carbon from root systems is a process that we generally don't see. The flow of carbon associated with litter fall is roughly the difference between the photosynthetic uptake of carbon and the return of carbon through plant respiration. If this were not the case, then the size of the global land biota reservoir would be growing or declining, and although some regions are growing, others are shrinking, and they nearly balance out.
Soil Respiration, Permafrost and Runoff
Soil Respiration, Permafrost and Runoff
Respiration (sometimes called decay) also occurs within the soil, as microorganisms consume the dead plant material. In terms of a chemical formula, this process is the same as described above for plant respiration (the reverse of photosynthesis).
There is an unseen but fascinating universe of microbes living within the soil, and they are the key means by which nutrients such as carbon and nitrogen are cycled through the soil system. A great diversity of microorganisms live in the soil, and they are capable of consuming tremendous quantities of organic material. Much of the organic material added to the litter (the accumulated material at the surface of the soil) or within the root zone each year is almost completely consumed by microbes; thus there is a reservoir of carbon with a very fast turnover time — on the order of 1 to 3 years in many cases. The by-products of this microbial consumption are CO2, H2O, and a variety of other compounds, and are collectively known as humus (not the same as Hummus, the Mediterranean chickpea purée!). Humus is a much less palatable compound, as far as microbes are concerned, and is not decomposed very quickly. After it is produced at shallow levels within the soil, it generally moves downward and accumulates in regions of the soil with high clay content. Part of the reason it accumulates in the lower parts of the soil is that there tends to be less oxygen in that environment, and the lack of oxygen makes it even more difficult for microbes to work on this humus and decompose it further. But eventually, due to various processes (animals burrowing, people plowing, etc.) that stir the soil, this humus moves back up to where there is more oxygen, and then the microbes will eventually destroy the humus and release some more CO2. This humus then constitutes another, longer-lived reservoir of carbon in the soil. Carbon 14 (14C) dates on some of this soil humus give ages of several hundred to a thousand years old. Taken together, the fast and slow decomposition processes, both driven by microbes, lead to an average carbon residence time of around 20 to 30 years for most soils. The data used in our global carbon cycle model lead to a residence time of about 26 years for the global soil carbon reservoir.
These microbes (considered in terms of their respiratory output) are very sensitive to the organic carbon content of the soil as well as the temperature and water content, respiring faster at higher carbon concentrations, higher temperatures and in moister conditions.
Permafrost - an unknown
In recent years, increasing attention has been directed at permafrost soil carbon, since the polar regions are warming much faster than the rest of the globe. Permafrost is soil that has been frozen for at least two years. As the permafrost melts, carbon that was added to these soils by processes like litter fall will become available for soil microbes to respire and release to the atmosphere. In fact, it is almost surely happening already, but given that much of the permafrost is still frozen, we have probably not seen the real manifestation of this source of carbon. Estimates are variable, but a figure like 1000 to 1500 Gt of carbon reflects the current thinking; this is a huge amount of carbon and has the potential to significantly alter the future of atmospheric CO2 levels. As the permafrost begins to melt, some estimates are that it will contribute something in the range of 2-5 Gt C/yr, which is large compared to the human-related changes. Of course, some of this released carbon will be offset by new carbon sequestered into these formerly frozen soils, but initially, the system will not be in equilibrium and these regions can be expected to be a net source of CO2 to our atmosphere.
Runoff
Although most of the carbon loss from the soil reservoir occurs through respiration, some carbon is transported away by water running off over the soil surface. This runoff is eventually transported to the oceans by rivers. The actual magnitude of this flow is a bit uncertain, although it does appear to be quite small. The most recent estimates place it at 0.6 Gt C/yr.
All Together
All Together
Here are the terrestrial flow processes once again, but this time with the estimated magnitudes of flows included. Most of these are very large flows, and they have a seasonality to them — photosynthesis is obviously big in the growing season, but it is small in the winter. If land masses and land plants were equally divided in the northern and southern hemispheres, this seasonality would cancel out, but in today's world, a large majority of land and plants are in the Northern Hemisphere, so around July, photosynthesis is very strong. In fact, this on and off aspect of photosynthesis is the primary reason for the seasonal changes in atmospheric CO2 concentrations seen in the Mauna Loa record.
Check Your Understanding
The Marine Carbon Cycle
The Marine Carbon Cycle
Processes of Carbon Flow in the Oceanic Realm
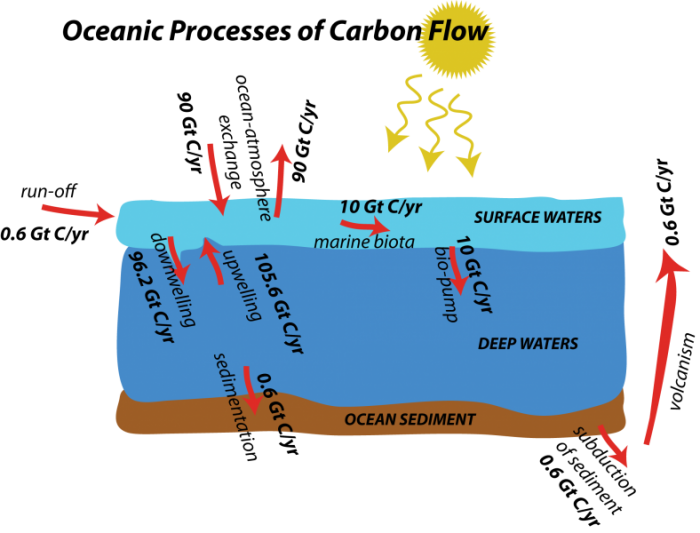
Far less obvious to us than the terrestrial processes we just discussed, the cycling of carbon in the oceans is tremendously important to the global carbon cycle. For example, the oceans absorb a large portion of the CO2 emitted through anthropogenic activities. As with the terrestrial part of the global carbon cycle, we will explore here the various processes involved in transferring carbon in and out of the oceans.
Below, we see a general depiction of the flows involved in the oceanic realm, along with the flow magnitudes.
Ocean - Atmospheric Exchange
Ocean - Atmospheric Exchange
Carbon dioxide can be dissolved in seawater, just as it can be dissolved in a can of soda. It can also be released from seawater, just as the CO2 from soda can also be released. This transfer of gas back and forth between a liquid and the atmosphere is an extremely important process in the global carbon cycle, since the oceans are such an enormous reservoir with the potential to store and release significant quantities of CO2.
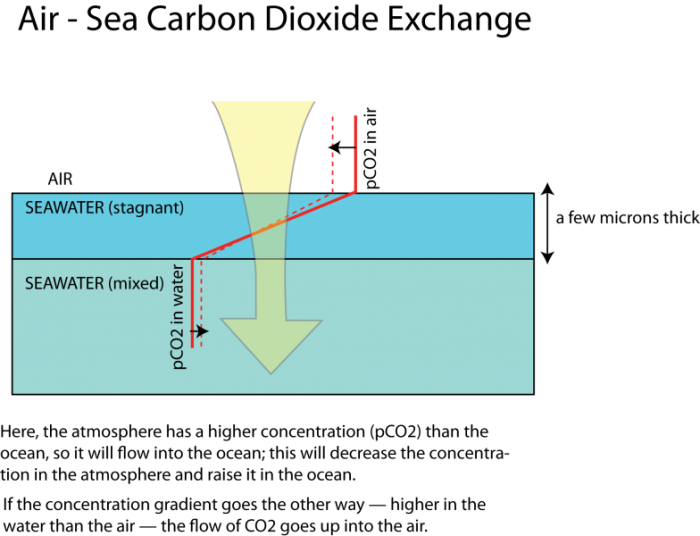
The exchange of a gas like CO2 between the air and seawater is governed by the differences in concentrations, as shown in the figure below, where the solid red line represents the concentration (increasing to the right) in the air and in the ocean. The dashed line indicates what the concentration might look like after some exchange of CO2 occurs — lower concentration in the atmosphere, and higher in the oceans. But note that the change in concentration is less in the ocean than in the atmosphere, which is a result of some chemistry we will explore in just a bit.
Click here to see a text description.
As depicted in the figure above, the concentrations in the air and the sea are relatively constant (spatially, not temporally) since these two media undergo rapid and turbulent mixing that would tend to even out any systematic variations. The exception to this is a thin layer of water, just 20 to 40 microns thick (a micron is one thousandth of a millimeter), which, because of the surface tension of the water, is unable to mix well. This stagnant film is the barrier across which the diffusion has to occur. The rate of gas transfer is a function of the concentration difference and the thickness of the stagnant film of water (which thins when the winds are strong).
In general, this flow depends on the concentration of CO2 in the atmosphere and the oceans, but it gets a little complicated because the concentration of CO2 in seawater depends on a number of other factors. To understand this process, and to see why the atmosphere's concentration changes more than the ocean, we need to have some sense of what happens to CO2 once it gets dissolved in seawater.
Carbonate Chemistry of Seawater
When CO2 from the atmosphere comes into contact with seawater, it can become dissolved into the water where it undergoes chemical reactions to form a series of products, as described in the following:
- CO2 (dissolved gas) + H2O <=> H2CO3 (carbonic acid)
- H2CO3 (carbonic acid) <=> H+ + HCO3- (hydrogen ion + bicarbonate)
- HCO3- <=> H+ + CO3-2 (hydrogen ion + carbonate)
The amount of CO2 as dissolved gas is what controls the concentration of CO2 shown in the figure above. This concentration depends strongly on the temperature — it is low in cold water and it is high in warm water, which means that the colder parts of the ocean absorb CO2 from the atmosphere and the warm parts of the ocean release CO2 into the atmosphere.
All of these reactions mean that in seawater, you can find all of these different forms or species of inorganic carbon co-existing, dissolved in seawater. In reality, though, bicarbonate (HCO3-) is the dominant form of inorganic carbon; carbonate (CO32-) and dissolved CO2 are important, but secondary (see figure below).
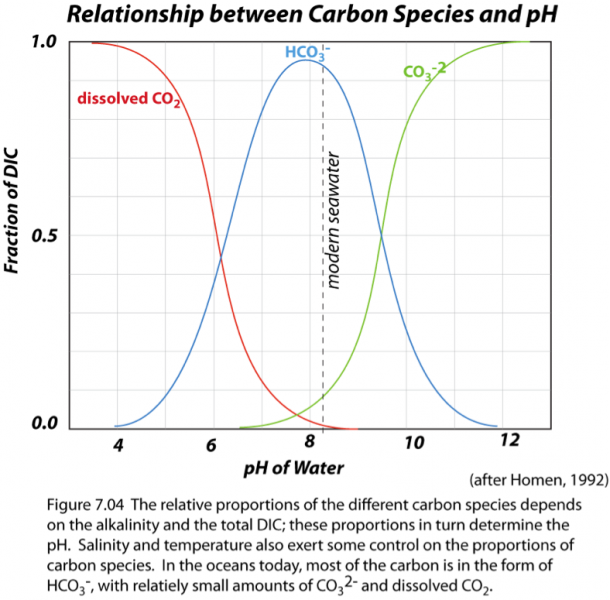
Click here to see a text description.
In the above equations (1-3), the double-headed arrows mean that the reactions can go in both directions, and generally do, until some balance of the different compounds is achieved -- a chemical equilibrium.
Notice that in the equations above, the hydrogen ion appears in a number of places — the concentration of H+ in seawater is what determines the pH of the water, or in other words, its acidity. Remember that low pH means more acidic conditions. It turns out that the ratio of bicarbonate (HCO3) to carbonate (CO3) is proportional to the pH; at lower pH conditions, more of the carbon is in the form of bicarbonate than carbonate, and the result is that the fraction of dissolved CO2 gas also goes up, and this tends to cause CO2 gas to move from the seawater into the atmosphere. As we will see in Module 7, higher acidity (lower pH) also has important implications for organisms that live in the sea.
Obviously, the ratio of these two forms of carbon is important, so we need to ask what controls this. Without getting into the chemistry too much, the ratio depends on two main things, and it depends on these things because they determine the electrical charge balance in the water — the charges from all the positive and negative ions dissolved in the water have to add up to zero, and you can see that if you have more carbonate (minus 2 charge) than bicarbonate (minus 1 charge), you have more total negative charge. So, the 2 important factors here are:
- the total amount of positive charge in the oceans that has to be canceled by the carbonate and bicarbonate — called the alkalinity;
- the total amount of dissolved inorganic carbon in the oceans (DIC for short).
If the alkalinity is high, then more of the DIC has to take the form of carbonate (minus 2 charge), and a result is that the pH is higher, meaning less acidic. If the total DIC is large, then to get the charges to balance, more of the DIC has to be in the form of bicarbonate (minus 1 charge), which leads to lower pH and more acidic conditions.
Let’s return now to the figure that showed the exchange of CO2 between the atmosphere and ocean and the question about why the ocean’s CO2 concentration changed less than the atmosphere. The reason for this is the carbon chemistry reactions that shift some of the CO2 dissolved gas into the forms of bicarbonate and carbonate — this reduces the amount of carbon in the form of dissolved CO2 gas, so the concentration of dissolved CO2 does not increase as much as it would if these reactions did not take place.
As you can see, the chemistry of carbon in seawater is relatively complex, but it turns out to be extremely important in governing the way the global carbon cycle operates and explains why the oceans can swallow up so much atmospheric CO2 without having their own CO2 concentrations rise very much.
Summary of Carbonate Chemistry
Let's see if we can summarize this carbonate chemistry — it is important to have a good grasp of this if we are to understand how the global carbon cycle works.
- Carbon can exist in three main inorganic forms in seawater — CO2, HCO3-, and CO32-, and there is a rapidly-achieved equilibrium between these species.
- The ratio of HCO3- to CO32- along with the water temperature determines the CO2 concentration of seawater and also the pH.
- The temperature of the water also controls how much CO2 occurs in the form of dissolved gas, thus affecting the concentration of CO2 gas in seawater.
- The alkalinity of seawater represents the positively-charged ions that need to be countered by negatively-charged carbonate and bicarbonate ions.
- The concentration of the total dissolved inorganic carbon (DIC), along with the alkalinity, determines the ratio of HCO3- to CO32-, and thus the CO2 concentration of seawater. If we increase DIC without changing the alkalinity, then more carbon must be in the form of HCO3-, which increases both pH and the CO2 concentration of seawater.
- The CO2 concentration of seawater, relative to the atmospheric CO2, determines whether the oceans absorb or release CO2. Currently, the cold parts of the oceans absorb atmospheric CO2 and the warm regions of the oceans add CO2 to the atmosphere.
- The ability of carbon to switch back and forth between these three forms means that only a portion of the CO2 absorbed by the oceans will remain as CO2.
In the real world, there are important variations in the gas transfer between the ocean and atmosphere. This can be seen in the figure below, which represents a kind of snapshot of this transfer across the globe. The units here are grams of C per m2 per year, and each box is about 1e6 m2. The red, orange, and yellow colors represent places where the oceans are giving up CO2 to the atmosphere; the blue and purple areas are places where the oceans are sucking up atmospheric CO2. Summing these up, we find that the oceans are taking up around 92-93 Gt C/yr and they are releasing about 90 Gt C/yr — for a net flow of 2-3 Gt C/yr into the oceans — this represents something like 25-30% of the carbon we are adding to the atmosphere by burning fossil fuels. This exchange is variable in space and time, but a few general features can be pointed out. In general, the colder parts of the oceans absorb CO2 and the warmer parts release CO2 into the atmosphere. This makes sense because CO2 is more soluble in colder water.
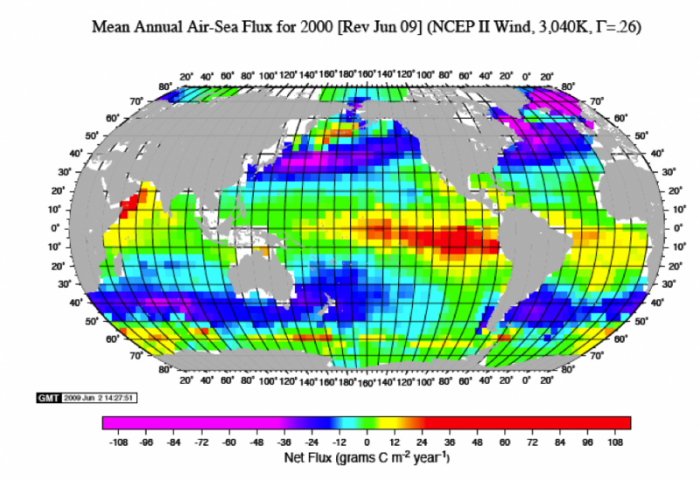
Marine Biota Exchange — The Biologic Pump
Marine Biota Exchange — The Biologic Pump
The surface waters of the world’s oceans are home to a great number of organisms that include photosynthesizing phytoplankton at the base of the food chain. These plants (and cyanobacteria) utilize CO2 gas dissolved in seawater and turn it into organic matter, and just like land plants, phytoplankton also respires, returning CO2 to the surface waters.
At the same time, many planktonic organisms extract dissolved carbonate ions from seawater and turn them into CaCO3 (calcium carbonate) shells. When these planktonic organisms die, their soft parts are mainly consumed and decomposed very quickly, before they can settle out into the deeper waters of the oceans. This decomposition thus returns carbon, in the form of CO2, to seawater.

However, some of the organic remains and the inorganic calcium carbonate shells will sink down into the deep oceans, thus transferring carbon from the shallow surface waters into the huge reservoir of the deep oceans. This transfer is often referred to as the biologic pump, and it causes the concentration of CO2 gas, and also DIC in the surface waters to be less than that of the deeper waters. This can be seen in the figure below, which shows the vertical distribution of DIC (and also alkalinity) in a profile view for some of the major regions of the world's oceans.
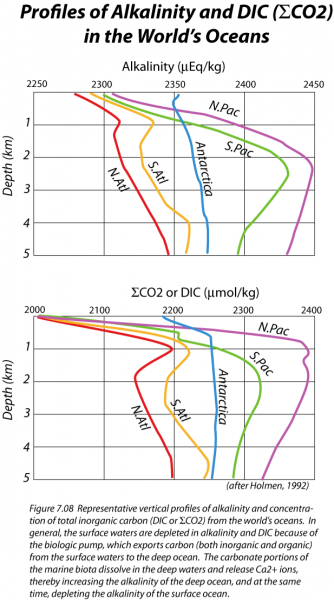
Why is the alkalinity reduced in the surface waters? For the same reason that DIC is depleted. Planktonic organisms make shells of CaCO3, and when these sink to the seafloor, they carry Ca2+ ions with them, thus reducing the alkalinity. Much of this CaCO3 is later dissolved when it reaches deeper parts of the oceans, which explains the higher alkalinity values in the deep waters, as seen in the figure above. By controlling the concentration of CO2 gas dissolved in the surface waters, the planktonic organisms exert a strong influence on the concentration of CO2 in the atmosphere. For instance, if the biologic pump were turned off, atmospheric CO2 would rise to about 500 ppm (relative to the current 408 ppm); if the pump were operating at maximum strength (i.e., complete utilization of nutrients), atmospheric CO2 would drop to a low of 140 ppm. Clearly, this biologic pump is an important process.
Alkalinity is very important, because when it is reduced, species that make shells out of CaCO3 will dissolve. Today, the most sensitive species are those that make shells out of a form of CaCO3 called aragonite which is more susceptible to dissolution than calcite, the other form of CaCO3. Coral is an aragonite group that is very susceptible as we will see in Module 7.
What controls the strength of this biologic pump? The photosynthesizing plankton require nutrients in addition to CO2 in order to thrive; specifically, they require nitrogen and phosphorus. Most of these plants need P, N, and C in a ratio of 1:16:125, and since at present the ratio of P to N in ocean water is about 1:16, both P and N limit the growth of these phytoplankton. Photosynthetic activity of plankton can be mapped out by satellites tuned to record differences in water color due to the presence of chlorophyll. This distribution is shown in the figure below.
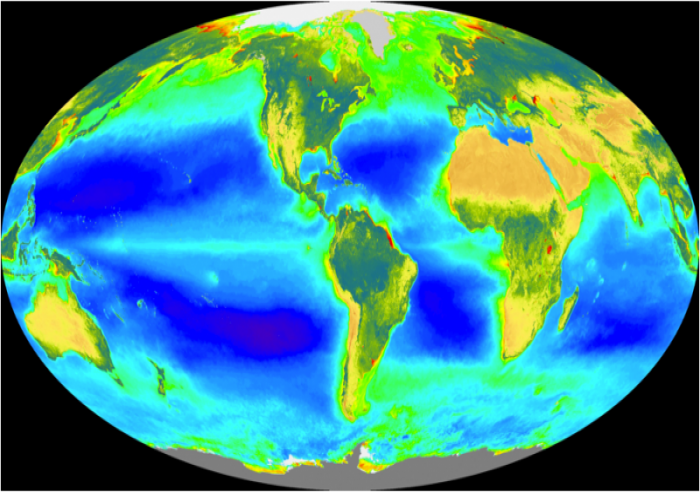
If the nutrients in seawater were being utilized to the maximum extent possible, there would be practically no P and N dissolved in seawater. But in fact, as shown by the figure below, the concentration of P tells us that the biologic pump is not operating at maximum efficiency. In the map below, the purple regions represent regions with no phosphate in the surface water of the oceans, meaning that there is simply a lack of nutrients, or that all the nutrients are utilized. In particular, it is the cold, polar regions that are not utilizing all of the available nutrients. This may be due in part to the temperature, but it may also be related to a paucity of iron, a minor nutrient that is apparently lacking in the colder regions, especially in the Southern Ocean ringing Antarctica.
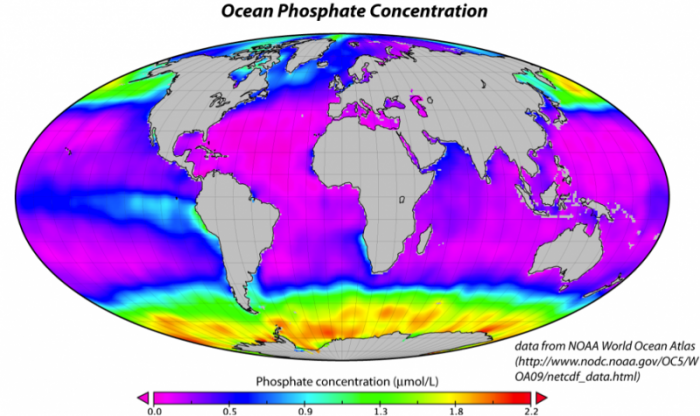
In addition to nutrients, the strength of this biological pump is sensitive to the pH of the ocean water. The organisms in the oceans are adapted to a pH of around 8.3 or 8.4, but as we discussed earlier, if the oceans take up too much CO2 too quickly, the pH will decrease and move it outside the optimum range for the organisms in the oceans. Thus, lower pH levels (i.e., higher acidity) will probably mean a reduction in the strength of the biological pump, which will, in turn, limit the oceans ability to absorb more carbon.
Downwelling
As mentioned above, downwelling (sinking of dense water) transfers cold and/or salty surface waters into the deep interior of the oceans, and as a result, carbon is transferred as well. The magnitude of the flow is thus a function of the volume of water flowing and the average concentration of carbon in the cold surface waters, which is itself a function of the total amount of carbon stored in this reservoir, assuming that the size of the reservoir is not changing appreciably over the few hundred years that this model is intended to be used for.
Downwelling occurs primarily near the poles, where surface waters are strongly cooled by contact with the air. This cooling leads to a density increase. The formation of ice from seawater at the margins of Antarctica increases the salinity of the seawater there, adding to the density of the water. This dense water then sinks and flows through the deep oceans, effectively mixing them on a timescale of about 1000 years or so (the Atlantic Ocean mixes somewhat faster, which helps explain the smaller ΣCO2 and alkalinity gradients seen in the figure above).
Upwelling
Upwelling is just the opposite of downwelling, and as deep waters rise to the surface, they bring with them carbon, nutrients, and alkalinity. The total transfer of carbon is thus a function of the volume of water involved in this flow and the amount of carbon stored in the deep ocean reservoir. Upwelling occurs in areas of the oceans where winds and surface currents diverge, moving the surface waters away from a region; in response, deep waters rise up to fill the "void". Upwelling occurs along the equator, where there is a strong divergence, and also along the margins of some continents, such as the west coast of South America. This upwelling water also brings with it nutrients such as nitrogen and phosphorus, making these waters highly productive.
Note that the amount of carbon transferred by this flow is greater than the downwelling flow. This is not because the volume of flow is different in these two processes, but rather because the concentration of carbon in the deep waters of the ocean is greater than that in the shallow surface waters, due in part to the operation of the biologic pump mentioned above.
Sedimentation
Some of the carbon, both organic and inorganic (i.e., calcium carbonate shells) produced by marine biota and transferred to the deep oceans settles out onto the sea floor and accumulates there, eventually forming sedimentary rocks. The magnitude of this flow is small — about 0.6 Gt C/yr — relative to the total amount transferred by sinking from the surface waters — 10 Gt C/yr. The reason for this difference is primarily because the deep waters of the oceans dissolve calcium carbonate shell materials; below about 4 km, the water is so corrosive that virtually no calcium carbonate material can accumulate on the seafloor. In addition, some of the organic carbon is consumed by organisms living in the deep waters and within the sedimentary material lining the sea floor. This consumption results in the release of CO2 into the bottom waters and thus decreases the amount of carbon that is removed from the ocean through this process. It is worth noting that the process of organic carbon consumption on the seafloor is another microbial process and is very similar to the soil respiration flow described earlier. Since the microbes living on the seafloor require oxygen to efficiently accomplish this task, the supply of oxygen to the seafloor by deep currents is an important part of this process.

Volcanism and Metamorphism
When sedimentary rocks deposited on oceanic crust are subducted [14] at a trench where two tectonic plates of Earth’s surface converge, they may melt or undergo metamorphism; in either case, the carbon stored in calcium carbonate -- limestone -- is liberated in the form of CO2, which ultimately is released at the surface. The CO2 may come out when a volcano erupts, or it may slowly diffuse out from the interior via hot springs, but in both cases, it represents a transfer of carbon from the reservoir of sedimentary rocks to the atmosphere. The magnitude of this flow is quite small and is adjusted here to a value of 0.6 Gt C/yr in order to create a model in steady state. This flow is defined as a constant in the model, although in reality, it will vary according to the timing of large volcanic eruptions. An extremely large volcanic eruption may emit carbon at a rate of around 0.2 Gt C/yr for a year or two, creating a minor fluctuation.
Check Your Understanding
Human Influences on the Global Carbon Cycle
Human Influences on the Global Carbon Cycle
Processes of Carbon Flow in the Human Realm
Humans have exerted an enormous influence on the global carbon cycle, largely through deforestation and fossil fuel burning. In this section, we explore how these processes have led to changes in the dynamics of carbon in the atmosphere.
Fossil Fuel Burning
Another pathway for carbon to move from the sedimentary rock reservoir to the atmosphere is through the burning of fossil fuels by humans. Fossil fuels include petroleum, natural gas, and coal, all of which are produced by slow transformation of organic carbon deposited in sedimentary rocks — essentially the fossilized remains of marine and land plants. In general, this transformation takes many millions of years; most of the oil and gas we now extract from sedimentary rocks is on the order of 70-100 million years old. New fossil fuels take a very long time to form, and we are using them up much, much faster than they are being formed, meaning that if we keep using fossil fuels at the rate we are today, we will run out! This run out date depends on new discoveries and our ability to extract fuels more efficiently by processes like fracking, but we will be close to running out late this century.
These fossil fuels are primarily composed of carbon and hydrogen. For instance, methane, the main component of natural gas, has a chemical formula of CH4; petroleum is a more complex compound, but it, too, involves carbon and hydrogen (along with nitrogen, sulfur, and other impurities). The combustion of fossil fuels involves the use of oxygen and the release of carbon dioxide and water, as represented by the following description of burning natural gas:
CH4 + 2O2 => CO2 + 2H2O
Beginning with the onset of the industrial revolution at the end of the last century, humans have been burning increasing quantities of fossil fuels as our primary energy source.

As a consequence, the amount of CO2 emitted from this burning has undergone an exponential rise that follows the exponential rise in the human population. The magnitude of this flow is currently about 9 Gt C/yr. This number also includes the CO2 generated in the production of cement, where limestone is burned, liberating CO2.
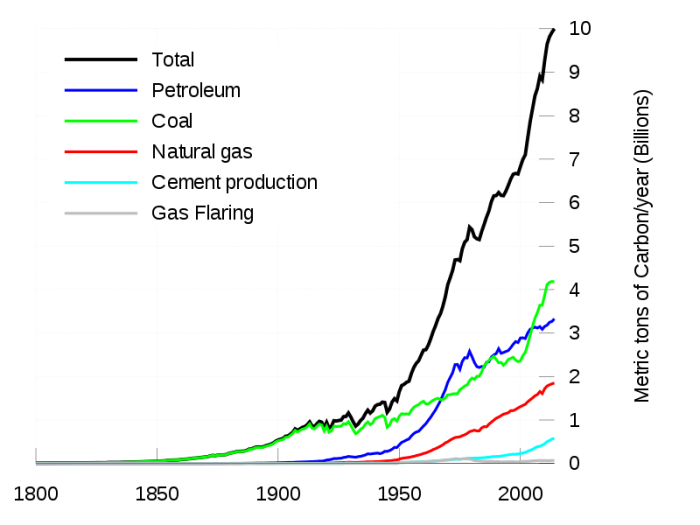
As you can see in the graph above, this flow has changed considerably over time, as human population has increased and as our economies have become more industrialized with a big thirst for the energy provided from the combustion of fossil fuels. The model we will work within the lab activity for this module includes this history, beginning in 1880 and going up to 2010; beyond 2010 is the realm of future projections, which can be altered to explore the consequences of choices we might make or not make in the future. Part of the new energy economy that is key to our future is the use of so-called renewable energy sources, including wind, solar and geothermal energy, that emit little or no carbon. There are some interesting map view representations of this history of fossil fuel carbon emissions in the video below:
Video: Annual Fossil-Fuel CO2 Emissions (2:03) This video is not narrated.
If the video does not play, you can view it at YouTube: Annual Fossil-Fuel CO2 Emissions [18]
Land-Use Changes - Forest Burning and Soil Disruption
The other form of human alteration of the global carbon cycle is through forest cutting and burning and the disruption of soils associated with agriculture. When deforestation occurs, most of the plant matter is either left to decompose on the ground, or it is burned, the latter being the more common occurrence. This process reduces the size (the mass) of the land biota reservoir, and the burning adds carbon to the atmosphere. Land-use changes other than deforestation can also add carbon to the atmosphere. Agriculture, for instance, involves tilling the soil, which leads to very rapid decomposition and oxidation of soil organic matter. This means that in terms of a system, we are talking about two separate flows here — one draining the land biota reservoir, the other draining the soil reservoir; both flows transfer carbon to the atmosphere. Current estimates place the total addition to the atmosphere from forest burning and soil disruption at around 2-3 Gt C/yr; estimates divide this into 70% to 50% forest burning, with soil disruption making up the remainder. Deforestation is a particular problem in the Amazon, as we will see in Module 9.
Deforestation
The actual history of this alteration to the natural carbon cycle is not well-constrained — not nearly as well known as the fossil fuel burning history — but we include a reasonable history that reflects patterns of land settlement and forest clearing.
Lab 5: Carbon Cycle Modeling (Introduction)
Lab 5: Carbon Cycle Modeling (Introduction)
The goals of this lab are to:
- Simulate the fate of CO2 added to the atmosphere;
- Interpret what happens when we increase the rate of fossil fuel burning;
- Evaluate how the rate of carbon addition affects the maximum temperature and minimum pH attained under fossil fuel burning;
- Simulate the impact of the different emission scenarios on global temperature;
- Evaluate how permafrost melting amplifies warming under the different emission scenarios.
Please make sure that you read the Introduction to the lab. Skipping it will result in a lot of confusion and a lower score on the assignment.
Introduction
In the lab activity for this module, we will be working with a STELLA model of the global carbon cycle that is attached to the climate model we used in Module 3. This model incorporates the processes of carbon transfer in the terrestrial and oceanic realms discussed in the previous sections; it also includes the history (from 1880 to 2010) of human impacts on the carbon cycle in the form of emissions from burning fossil fuels, burning forests, and disrupting the soil. The model is initially set up to represent the carbon cycle in a steady state just before the industrial revolution, at which point human alterations to the carbon cycle began in earnest. We will use this model to explore different carbon emissions scenarios for the future, to see how the climate and carbon cycle might respond.
Here is what the model looks like, in a very simple, stripped-down form:
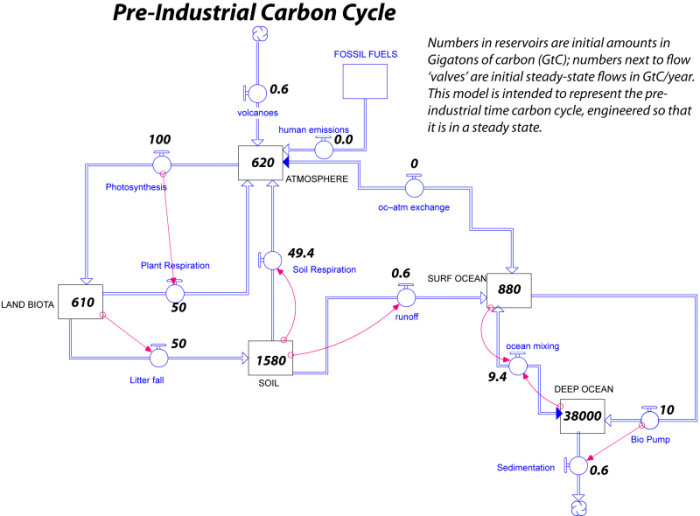
In this figure, you can see the initial amounts of carbon in each reservoir in GT and the annual flows of carbon between reservoirs in GT C/yr. A couple of things should be pointed out about this model. In some cases, flows have been combined into things called bi-flows that have arrows on each end; this means that carbon can flow either way. There is a bi-flow connecting the atmosphere and surface ocean that represents the two-way transfer of ocean-atmosphere exchange. There is another bi-flow connecting the surface and deep ocean that represents upwelling and downwelling combined into one.
The real model is quite a bit more complex-looking, as can be seen below:
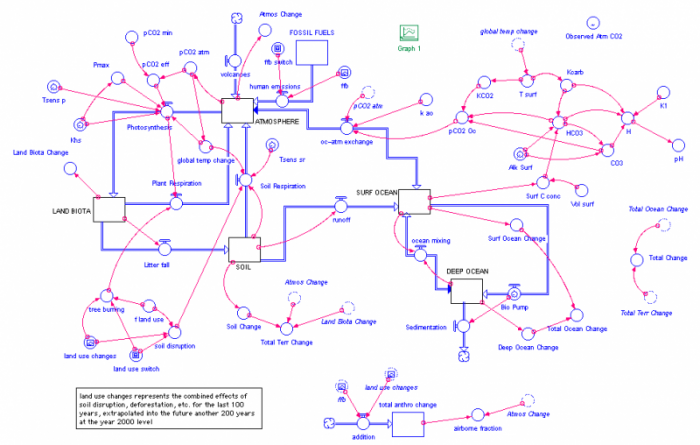
The complications here arise from the fact that most of the flows are expressed by rather complicated equations. Most of the flows have small red arrows attached to them; these show you what things affect the flow rate. Think of the circle in the middle of the flow pipe as being a valve on a water pipe that controls how much water moves through the pipe. In many cases, the red arrows come from the reservoir that is being drained by the flow; this means that the outflow is dependent on how much is in the reservoir — when you have more in a reservoir, the outflow is often greater, and in essence, the outflow is a percentage of how much is in the reservoir.
Note that the atmosphere reservoir is connected to a converter called pCO2 atm — this is the concentration of CO2 in the atmosphere and the units are in parts per million or ppm, the same units that CO2 concentrations are typically given in. In this model, the initial amount of carbon in the atmosphere gives a pCO2 value of 280 ppm (and by now, it is just over 400 ppm). The pCO2 atm converter is in turn connected to the same climate model we used in Module 3, where it determines the strength of the greenhouse effect. The climate model calculates the temperature at each moment in time and then passes that information back to the carbon cycle model in the form of a converter called global temp change, which is the change in global temperature relative to the starting temperature — this is like a temperature anomaly.
The global temp change converter is then attached to a couple of other converters that attach to the photosynthesis flow and the soil respiration flow. Both of these flows are sensitive to temperature and the temperature combines with something called a temperature sensitivity. You can see something above called the Tsens sr — this is the temperature sensitivity for soil respiration. Both photosynthesis and soil respiration are sensitive to the temperature; they increase with temperature. Global temp change is also connected to the surface temperature of the oceans (T surf) via a "ghosted" version of the converter — a dashed line version that helps eliminate so many long connecting arrows running all over the diagram.
The photosynthesis flow has lots of converters associated with it since it is dependent on numerous factors, but the main things are temperature and the atmospheric CO2 concentration.
The model also includes a whole set of connected converters in the upper right that do all of the calculations related to the carbon chemistry in the oceans; this is where the pH or acidity of the surface ocean is calculated.
At the very top right of the model, there is a converter called Observed Atm CO2 that contains the observed history of atmospheric CO2 concentration since 1880. This is in the model so that we can test how good the model is. If our carbon cycle model is good, then it should calculate a pCO2 that closely matches the observed record.
The model includes the history of carbon emissions from burning fossils fuels shown in the previous section; it also includes a history of land use changes that impact the carbon cycle. These land use changes are broken up into tree burning (that accompanies deforestation) and soil disruption (related to farming); they are then additions to the flow of carbon from the land biota and soil back into the atmosphere. These human alterations to the carbon cycle are shown in the model by clicking on the pink graph icons labeled ffb and land use changes on the right side of the model, as shown below.
As before, the model we will work runs on a browser; here is a link to the model [20]. When you follow the link, you should see a screen like this (note the model has changed but the functions are similar):
You can think of this as the control panel for the model, where you can run it, stop it, make changes, and look at the results in the form of different graphs. You can access the different graphs by clicking on the triangular tab at the lower left of the graph window. The controls here consist of a set of switches you can turn on or off — these will determine which of 3 different emissions scenarios are applied to the model. The three buttons on the right-hand side show what the 3 emissions scenarios look like. The video below explains how to work the switches. There is also a switch that can be used to either include or exclude the carbon emissions to the atmosphere from human-related land-use changes such as deforestation and soil disruption, but we will not mess around with this switch — just leave it in the "on" or up position as it is in the diagram above.
This initial model is set up to run from 1880 to 2100. Later, we will work with a model that runs farther into the future.
This is the interface to the carbon cycle model that we will be working with in this module. If we click on this button here, we would see a view of what the carbon cycle looks like that's implemented in this model. It's a complicated thing, but it's a very realistic carbon cycle model that's going to allow us to explore, in a pretty realistic way, what will happen to atmospheric CO2 and to other aspects of the carbon cycle as we change the burning of fossil fuels and the human effect on this particular system. This carbon cycle model here is attached to a little climate model, that you can see the edge of it over here, that's the same one that we worked with in Module 3. And so, the carbon cycle model will control the atmospheric CO2 concentration here, and then that will feed into the climate model here and control the greenhouse effect. And then the temperature, determined by the climate model, will then come back and affect different parts of the carbon cycle model here.
So, let me show you how to work this model. If you run it just the way you first open it, you'll see it calculates the global temperature change over time. So, zero is the temperature at 1880, but this is temperature relative to that time. And then, the temperature rises as we go to the year 2000 and 2100. Now, these switches down in here control different histories or scenarios of fossil fuel burnings. The business-as-usual emission scenario is what we ran first. So, when this switch is on, it uses that business-as-usual scenario, and it looks like this, where the carbon emissions just go up and up and up with no change all the way to 2100.
If we click this off, and leave this switch off, it's going to implement a scenario in which the fossil fuel burning just levels off. So, after the year 2010, it just levels off like this. So, we hold emissions constant, and we can see what happens to the temperature in the rest of the system in that case. If we turn this switch on, like that, it will implement this scenario here. It's a very unrealistic one, where after 2010, we drop quickly down to zero fossil fuel burning for the rest of time. So, this is a really dramatic case, just to see how the system will respond to that change.
Now, you can graph different parts of the system. Here, we can look at the atmospheric CO2 concentration. Here, we can look at the ocean pH. Here, we can look at where the carbon goes, what fraction of it stays in the atmosphere, what fraction goes into the biosphere, and what fraction goes into the oceans. This shows us the details of the fossil fuel burning history that we applied in that case. This plots both the atmospheric CO2 concentration and the fossil fuel burning history together, so you can see how they compare. This shows the values of carbon and all the different reservoirs in the carbon cycle model. This is the cumulative amount of carbon that we've released over time, so it just gets bigger and bigger and bigger. This is not the annual amount, but the cumulative.
Now, this is an interesting one to look at because it goes from 1880 to 2010, so the period of time that we know something about. And it plots in red the actual observed atmospheric CO2 level and in blue the CO2 level that the model calculates. And so, you can see that those two are very close throughout this whole time. And so, that means that our carbon cycle model, coupled with the climate model, is giving us a result that matches a historical record. And so, we say we have a good model, and we can essentially trust what it tells us in terms of projections off into the future.
Lab 5: Carbon Cycle Modeling
Lab 3: Climate Modeling
Download this lab workbook as a Word document: Graded Lab Module 5. [21] (Please download required files below.)
Answer the questions in the workbook. Then, when you are ready, complete the Module 5 Lab 5 Submission in a timed environment. Make sure you have the model ready to run, as we will be asking additional questions as indicated below.
STEP 1. Please Note: The STELLA model in shown in the video below which explains the controls.
Video: Carbon Cycle Intro (4:15)
Hi, everybody, so this is the model that we're going to use in the first part of the lab, and I want you to really familiarize yourself with the controls before you really get started, okay? I think it's really important. And, it can be a lot of fun to experiment with this and really get used to playing with it before you start the lab. And I think if you do that, then the lab will be very straightforward.
So basically, what we have here is a carbon cycle, very complicated. This is the STELLA version of the carbon cycle here, and you can basically see all the knobs and bells and whistles with these STELLA connections. And over on this side is the ocean model that we already used in Module 3 lab. So you really don't need to understand this, of course, but it shows you how complicated it is, and we'll see later on why it's such a good model.
Ok, so the bottom line is we have three different emissions scenarios that are controlled down here by these switches, okay? We have a business as usual (BAU) switch, which is basically the worst case scenario, emission scenario A2. And you can see that is here. It goes all the way up and actually, then it drops off, but still it goes all the way up through 2100. We continue fossil fuel burning through 2100 without stopping. Okay, that's this switch. So that's with this on. And you can read about this down here. So if I turn this switch off and I leave this switch off, and I'll explain this switch in a minute, then what we do is we level off fossil fuel burning around 2010. We just level it off, okay? Which obviously didn't happen because we've gone through 2010. But it just shows you what happens if we slow down fossil fuel burning, don't completely stop it, okay? And then the third switch is what we do is if we all of a sudden drastically stop fossil fuel burning, FFB Halt. And what that shows is that around 2010, we stop. Let's see if I can move this up. But anyway, it's down here. You can see we stop fossil fuel burning at 2010. It goes down to zero. Ok, so that's basically what happens with that third switch.
So the three switches are, the three scenarios are, business as usual (BAU) with this switch on, this switch off. Both switches off, is Leveling Off at 2010. And then with this switch on, we halt fossil fuel burning (FBB) at 2010. Ok, don't worry about the land use switch for this part of the lab.
All right now, the critical thing is we can run, let's run business as usual (BAU). I want to show you one other thing. You run this model here, and let's say you want to then run another model. Let's say we'll turn it off and run the second one. You can see these two curves side by side, so you can see business as usual (BAU), and then you can see the leveling off of fossil fuel burning (FFB). And then if you want to run the third one. Excuse me, sorry about that. We're just going to turn this on. And then you see the third scenario here, 3. So, 1 - business as usual (BAU), 2 - leveling off (Leveling Off), 3 - fossil fuel burning (FFB). But what I wanted to show you also, is that if you want to then start over again and do something different, you restore everything. Okay? In general, it's a good idea between experiments to restore everything, to get everything back to back to usual, back to normal. And then why not?
One other tip that I just thought about is if for some reason the model isn't working for you and you aren't using Google Chrome. Switch to Chrome because it's the most stable. It's the most stable browser for STELLA models.
All right, so let me show you the output that we can get. So let's have business as usual (BAU). This is global temperature change, and as I said, the emissions, the fossil fuel burning stops at 2150, or so. And so you'll see this temperature decrease, which isn't really important for the lab. CO2, you can see it rises steadily here, and then drops here. pH goes down and then rises a little bit, but you can see it's going down. This is ocean pH.
And then one other thing I wanted to show you is make sure you can read the values here. So I'm going to click on the curve and you can see it running along the curve. It shows you the values down on the bottom rung, 8.14987. And then moving down the curve, you can see the values. You can see both the year and the values. So now I'm 2040, and I'm now at 2083, my value is 7.844. And now I'm down at 2040, excuse me, 2153, at the bottom and my value is 7.55. Ok, so make sure that you can run the the cursor along the curve and read the values not only of the year, but the values of pH, temperature, CO2, et cetera. All of these different curves you can do this, which is really, really helpful and very important for the lab. All right? So then we also have how much of the CO2 is absorbed by the air? How much is absorbed by the ocean? How much is absorbed by the biosphere in the purple curve, number 3.
Moving along, page 5 shows you fossil fuel burning, which shuts off as I said earlier, around 2050, or so. This is both CO2 and fossil fuel burning. Fossil fuel burning in red, CO2, atmospheric CO2 total, in blue, comparing those two curves. This here shows you what happens in the biosphere. It's the atmosphere in 1, the land biota in 2. How much CO2 is absorbed by land. How much is absorbed by soil. How much is absorbed by permafrost. And then this here shows how much it's absorbed by surface ocean and deep ocean. This here shows you the gigatons of carbon from fossil fuel burning. And then finally, a comparison of the model and observations.
So this shows you in blue the model run. This is the model run that we have established with this carbon cycle. And in red, we show the observed atmospheric CO2. This is measured in the atmosphere through twenty ten and you can see how close they are together, which shows that this model works really well. All right, so that's a good summary of what this model does. This is for the first part of the lab step one, and I will be producing another video for step two.
To begin with, we will use this model [22]of the carbon cycle (which is coupled to the same climate model we used in Module 3) to learn a few basic things about how the model responds to different scenarios of carbon emissions from fossil fuel burning (FFB). Note, the A2 scenario is also known as “Business-as-Usual” (BAU) in which we make no efforts to limit carbon emissions. Be sure to watch the video that introduces this model and explains how to use the switches to change the FFB scenarios. To get the right answers, it is imperative that you have switches in the correct positions. If in doubt, try reloading the model (reloading the web page) and restoring all devices.
Before running the model, try to guess what will happen to global temperature change under the two reduced emissions scenarios — in one of them (the leveling off scenario), the emissions are held constant after 2010, and in the other (the halt scenario), they drop to zero after 2010.
1. Will the global temperature change level off by 2100 if the emissions level off?
- Yes
- No
2. Will the global temperature change drop to 0 by 2100 if the emissions drop to 0?
- Yes
- No
Now run all three emissions scenarios (keep the land use switch in the on position) in the following sequence: A2 (BAU), then FFB Leveling Off, followed by FFB Halt.
3. Focus on the second scenario — what happens to the global temperature change when the FFB emissions level off (leveling off begins in 2010)?
- Temperature does not level off — it just increases more slowly
- Temperature levels off right away
- Temperature levels off after 20 years
4. Now turn to the FFB Halt scenario — what happens to the global temperature change when the FFB emissions halt (the halt begins in 2010)?
- Temperature drops off right away and goes to 0
- Temperature drops off shortly after 2010 but does not return to 0
- Temperature levels off right away
5. QUESTION IN CANVAS SUBMISSION ONLY.
6. QUESTION IN CANVAS SUBMISSION ONLY.
7. QUESTION IN CANVAS SUBMISSION ONLY.
STEP 2. Please Note: The model in the videos below may look slightly different than the model (linked below) that you will use to complete Step 2. Both models, however, function the same.
Now we turn to a version of the model [23] that has three emissions scenarios from the IPCC.
Video: Carbon Cycle (4:39)
Hi, students, this is the model for step two, the second part of the lab, and I want you to familiarize yourself with this model before you get started. So basically, what we have here are three different switches down the bottom, that I'm going to explain, and we have the scenarios up here on the right side and the carbon cycle model, which is basically the same model as we used in the first part of the lab. So let me explain these switches. So what we have is the business as usual (BAU), which we talked about in the first step, A2, and this is the scenario here, A2 with fossil fuel burning, just keeps going up and up and up and up. And in the second one, we can turn this off. We have B1, which is our best case scenario, and turn that on. And then that is our drastic reduction of fossil fuel burning at around 2010, 2020, somewhere in there. Doesn't really matter when it is, but you can see it comes down significantly.
Ok, so that's the two, A2 with that switch on, B1 with that switch off and this switch on. And the third model, this is what's critical, is with both of these switches off, business as usual and B1. We have the A1B model, the basically likely, the likely scenario where fossil fuel burning starts to decrease in the later part of the century. Ok. It doesn't come down to zero, but it starts to decrease 2030, 2050, somewhere in there.
All right. So basically, those are our three scenarios. A2, B1, and A1B with both of these switches off. Ok. And finally, over here, we have a permafrost burning switch. This is going to this is going to show you what happens when we start burning permafrost and putting all that methane and CO2 into the atmosphere that is with this switch on. Ok, with this switch off, we do not introduce that methane and CO2 into the atmosphere. All right. So let me show you what happens. Basically, it's it's the same controls as we had in the... It's the same graphs we had in the last part of the lab.
All right. So let's run through them again really quickly. Let's do it with the A1B scenario. So we can run the A1B and you'll see temperature in 1, CO2 in 2, and again, if you run your cursor along here, it gives you a value in a year, calendar year, and a value, which you need to be able to do to to do well in this lab. pH comes down, and you can see that doesn't flatten out. And this shows you some of the same things we explained in the last lab. How much of the carbon is absorbed by the air. How much is absorbed by the ocean. How much is absorbed by terrestrial biosphere. This shows you fossil fuel burning in A1B.
This shows you permafrost, which we don't have on, obviously. This shows you how much is absorbed by the atmosphere, how much is absorbed by soil, how much is absorbed by land biota and we don't have permafrost on. This shows you surface in deep ocean. This shows you how much carbon we put in from fossil fuel burning. And this, again, shows you the comparison between the real data and read the observed data and the model that we ran here.
Ok, so let me just show you a couple more things before I set you free to work on this. So what we can do again is we can run different scenarios and compare them side by side so we can do that. This is A1B. Let's turn A2 on. So there's our A2 scenario, business as usual (BAU). Let's turn B1 on. And then that's our third one. That's our business, our B1 scenario, the best case scenario. Ok, and then finally, let me restore everything. Ok, let's run A1B. And now let me show you what happens when I turn permafrost burning on. And you can see a lot more fossil fuels, or a lot more carbon ends up into the atmosphere. And you can compare the curves side by side.
So I think you'll have a lot of fun with this. Make sure you understand these controls before you start. And please let me know if you have any questions.
Again, the A2 scenario is also known as “Business-as-Usual” in which we make no efforts to limit carbon emissions; the A1B scenario is one of modest reductions in emissions; the B1 scenario is one of more drastic reductions. Run all three emission scenarios (A2, A1B, and B1) with the permafrost switch off and the land use switch on, then answer the following questions.
Temperature (Page 1 of the graph pad)
8. Which scenario produces the largest warming in 2100?
9. Which scenario produces the smallest warming in 2100?
10. What is the temperature difference between the highest and lowest emission scenario? (answer to 2.d.p)
11. QUESTION IN CANVAS SUBMISSION ONLY.
12. QUESTION IN CANVAS SUBMISSION ONLY.
Atmospheric CO2 (pCO2 atm on Page 2 of the graph pad)
13. Which emission scenario has the largest impact on drawing down CO2?
14. When does that decrease begin?
15. When does the rate of CO2 increase in A1B start to decrease?
16. QUESTION IN CANVAS SUBMISSION ONLY.
17. QUESTION IN CANVAS SUBMISSION ONLY.
pH (Page 3 of the graph pad)pH is a measure of the acidity of the ocean — it is related to the amount of CO2 dissolved in the oceans. More CO2 in the oceans lowers the pH, which means the water is more acidic (a phenomenon known as Ocean Acidification). We will see in Module 7 that the key variable controlling the precipitation of reefs and other organisms that make shells of CaCO3 is a variable called saturation, which is indirectly related to the pH. Let’s assume for the next four questions that coral framework precipitation in a species of coral declines at a pH of 8.0 and that it can no longer form any below a pH of 7.8 (again this is hypothetical since saturation is key).
18. List the emission scenarios that result in a slow-down in shell precipitation at any time during the model run (i.e., pH drops below 8.0).
19. Which of the scenarios will stop the growth of coral reefs?
20. What year does your answer from the previous question happen?
21. QUESTION IN CANVAS SUBMISSION ONLY.
22. QUESTION IN CANVAS SUBMISSION ONLY.
STEP 3.
Now, we will see what impact permafrost melting might have on the carbon cycle and climate. First, hit the “refresh” button on the browser to return the model to its original state. Run the A2 scenario with the PF switch in the off position, and then run it again with the PF switch turned on.
23. How much additional warming is caused by 2100 in the A2 scenario by the permafrost melting?
24. How much does the pCO2 atm increase by 2100 as a result of the permafrost melting?
Hit the refresh button again, and then run the B1 scenario (the one with the more drastic reductions) with the PF switch off, then run it again with the PF switch on.
25. How much additional warming is caused by 2100 in the B1 scenario by the permafrost melting?
26. How much does the pCO2 atm increase by 2100 as a result of the permafrost melting?
This result might be a surprise to you, but remember how CO2 affects the climate — an increase of something like 100 ppm is much more important at lower concentrations than higher concentrations. So, going from 200 ppm to 300 ppm causes more warming than going from 700 ppm to 800 ppm.
27. QUESTION IN CANVAS SUBMISSION ONLY.
28. QUESTION IN CANVAS SUBMISSION ONLY.
Module Summary and Final Tasks
Module Summary and Final Tasks
End of Module Recap:
In this module, you should have grasped the following concepts:
- Carbon flows through the Earth system by a combination of biological, chemical, and physical mechanisms.
- The global carbon cycle is a critical part of the climate system since it controls the concentration of CO2 in the atmosphere, and CO2 is, because of its long residence time in the atmosphere, the main greenhouse gas "driver" of climate change.
- The carbon cycle is complicated by the fact that it has fast and slow parts to it, with the deep ocean exerting a lot of inertia that limits rapid changes; it also has a variety of positive and negative feedback mechanisms in which the flow rates depend on temperature. Finally, it has a number of important human components to it — burning fossil fuels, deforestation, and soil disruption.
- The global carbon cycle is probably never in a steady-state, but it has negative feedback mechanisms that drive it toward a steady state. The response time of the system is very long, measured in tens of thousands of years due to the vast, sluggish deep ocean part of the system. Some parts of the system can respond (partially) to changes very quickly, as evidenced by the seasonal variations in atmospheric CO2 seen in the Mauna Loa record.
- The human alterations to the carbon cycle are significant — we now add more than 10 times as much carbon to the atmosphere as volcanoes. Roughly half of this fossil fuel carbon has remained in the atmosphere, with the rest being taken up by the oceans and land plants.
- Land plants respond to higher CO2 concentrations in the atmosphere by increasing their efficiency, which enables them to take up increased amounts of CO2 from the atmosphere, but this effect has a saturation point — an atmospheric CO2 level where the uptake of CO2 does not increase any more.
- The oceans are also an important part of the uptake of carbon, aided by chemical reactions that convert CO2 gas into carbonate ions in solutions that are then exported to the deep ocean through the sinking of calcium carbonate shells, a process called the biological pump.
- The chemical reactions associated with the uptake of CO2 by the oceans also influence the acidity of the oceans. If the oceans take up too much atmospheric CO2, the acidity will increase — this acidification of the oceans is made worse if the rate of carbon emissions goes up. If the oceans become too acidic, it may have a detrimental impact on biological productivity in the oceans and thus the biological pump may be weakened, which will limit the amount of carbon the oceans can absorb.
- The various IPCC emissions scenarios all lead to increased temperatures in the next century — even if we reduce the rate of emissions from current levels, the atmospheric CO2 concentration will increase, and thus the temperature will increase.
- There are some important unknowns in the carbon cycle, including how soil respiration will respond to temperature — if it is very sensitive, then at higher temperatures the soil reservoir will become a source of carbon rather than a sink. Furthermore, warming in the high latitudes of the northern hemisphere may lead to important emissions of carbon from permafrost. Permafrost is, in fact, currently melting, and the soil trapped in these frozen soils could increase the global warming of the next century by an additional degree C.
Assignments
You should have read the contents of this module carefully, completed and submitted any labs, the Yellowdig Entry and Reply, and taken the Module Quiz. If you have not done so already, please do so before moving on to the next module. Incomplete assignments will negatively impact your final grade.
Labs
- Lab 5: Carbon Cycle Modeling
