Lesson 5: Forensic Geology
Overview
Lesson 5 will take us one week to complete. This lesson is about mineralogy and forensic geology. First we'll examine a case study of sorts by reading an account about FBI agents and geologists who tracked down the origin of some soil adhering to the body of a murdered DEA agent in Mexico in the 1980s. Then you will come up with a short learning activity of your own in which students would examine local mineralogy.
What will we learn in Lesson 5?
By the end of Lesson 5, you should be able to:
- Describe the unique mineral assemblage present in the samples of soil discussed in "Death of an Agent."
- Differentiate between different types of volcanoes and the igneous rocks they produce.
- Guess which minerals to expect in soil or sand based on local knowledge of bedrock characteristics.
- Construct a short activity in which students investigate different soils or sands.
What is due for Lesson 5?
The chart below provides an overview of the requirements for Lesson 5. For assignment details, refer to the lesson page noted.
Lesson 5 will take us one week to complete. 8 Jul - 14 Jul 2020.
| Requirement | Submitted for Grading? | Due Date |
|---|---|---|
| Reading assignment "Death of an Agent" | No | |
| Activity: Design your own forensic mineralogy activity. | Yes - submit to Canvas assignment called "Forensics Lesson". | 14 Jul 2020 |
Questions?
If you have any questions, please post them to our Questions? Discussion Forum (not e-mail). I will check that discussion forum daily to respond. While you are there, feel free to post your own responses if you, too, are able to help out a classmate.
Reading Assignment
The reading assignment for this lesson is an account of some detective work done by geologists working for the FBI during the 1980s. They used forensic mineralogy to find the burial site of an undercover US DEA agent who had been killed in Mexico. One of the reasons I like this article is that it presents a case study. It's almost a story with a plot line, so it is a nice break from reading scientific articles. Furthermore, it puts into context why studying mineralogy and knowing how to identify certain types of rocks and minerals has important applications. Another reason I like it is that it synthesizes some of the material we've already covered in this course and in other courses in the M.Ed. program, while bringing up some new information, too. For example, we already know how to read geologic maps to get a feel for the plate tectonic structure of a region. After reading this article, we will study the tectonics of western Mexico and then extend our thinking to subduction zones, and volcanoes in particular.
Reading assignment
McPhee, J. (1996). The gravel page. The New Yorker, 71(46), 44, pp. 60-69.
Note that this reading is an excerpt from a larger article. Your assignment begins on page 60 under the heading "Death of an Agent," and ends on page 69 before Annie Leibovitz's article about showgirls in Vegas.
Overview
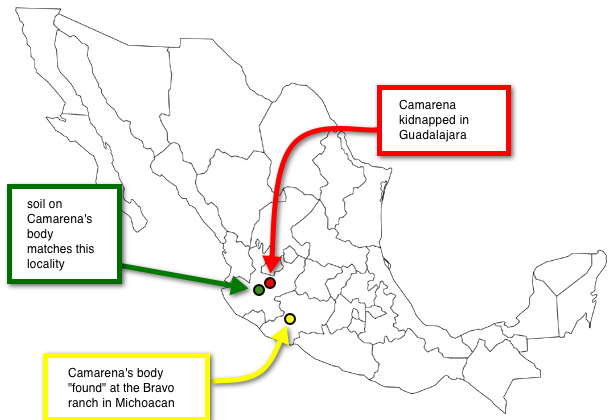
Here is a quick overview of the main points of McPhee's article: Witnesses saw Enrique Camarena kidnapped in broad daylight in Guadalajara (red dot on the map of Mexico, below). His body was later "found" by the MFJP at the Bravo family ranch in Michoacán (yellow dot on the map below). However, the soil from his body didn't match the soil at the Bravo ranch, it matched the soil in Bosques la Primavera state park in Jalisco (green dot on the map below).
The story hinges upon not just being able to differentiate between the two soils, but also upon being able to locate the real source of the soil on Camarena's body. In order to appreciate how the FBI geologists were able to do this, we will step back and discuss some basic points regarding mineralogic classifications and we will think about this in the wider framework of the mineralogic products of volcanoes. Knowing why certain volcanoes produce certain types of mineral assemblages comes from putting together a tectonic history of the volcanoes of interest, so we'll do this, too.
Mineralogy Review
What's a Mineral?
A mineral is a homogeneous, naturally occurring, solid inorganic substance with a definable chemical composition and an internal structure characterized by an orderly arrangement of atoms, ions, or molecules in a lattice.
Wow, that's kind of a mouthful, but each part of the definition is easy to explain.
- Homogeneous: The material is the same through and through. Are any of you old enough to remember when milk came in bottles and you had to skim the cream off the top? Now, the all milk available in grocery stores is homogenized, which means it has been processed so that the cream doesn't separate.
- Naturally occurring: True minerals are not formed by the activities of people. For example, manufactured industrial diamonds are “synthetic minerals.”
- Solid: Gases and liquids are not minerals.
- Inorganic substance: Oil, gas, fat, and plastic are organic and therefore not minerals.
- Definable chemical composition: Minerals have a chemical formula you can write down.
- Orderly arrangement of atoms in a lattice: The atom, ions, or molecules occur in a specific repeating pattern called a crystal lattice.
Quiz yourself!
- Is a snowflake a mineral? (check your answer [1])
- Is glass a mineral? (check your answer [2])
Did you know?
It is a common urban myth that glass acts like a liquid over long timescales. This myth, mostly perpetuated by tour guides in cities with old windows, is "proven" by pointing out to onlookers that the window glass in old buildings is thicker at the bottom. This is used as "evidence" that the glass flows downward over time due to gravity and has piled up at the bottom of the pane. Actually, the fact that old windows are thicker at the bottom is an artifact of an old method of glass-blowing, called the "crown glass procedure." In this method, still-molten glass was spun on a disc to flatten it as it cooled. This had the effect of making the disc fatter at the edges. Then the panes of glass were cut with an asymmetrical thickness built in to them. These panes were then usually installed with the thick end down because that is more stable. For a more detailed analysis of this topic, check out Plumb, 1989. "Antique windowpanes and the flow of supercooled liquids." J. Chem. Educ., 66, pp. 994-996.
Isotopes and Polymorphs
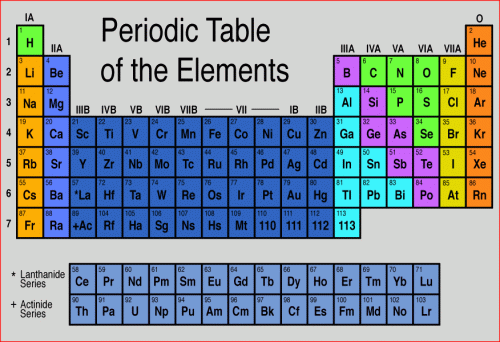
In order to discuss the the classification of various types of minerals, let's take a quick look at the Periodic Table of the Elements and review some simple atomic chemistry. All elements in the periodic table are made of one kind of atom, with a specific number of protons in its nucleus. The number of protons in the nucleus is the atomic number of the element. Below is an image of the periodic table. I assume this is familiar to you from high school and college!
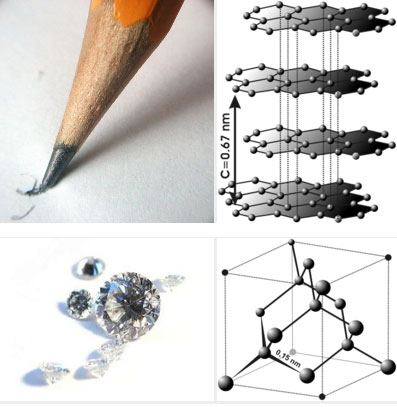
Let's take the element carbon, for example. Its atomic number is 6. That means it has 6 protons in its nucleus and 6 electrons orbiting. It can have different numbers of neutrons in its nucleus (6,7,8 are common). Two atoms with the same number of protons, but different numbers of neutrons are isotopes. Common isotopes of carbon are carbon-12 and carbon-14, denoted like this: C12 and C14. The superscripts in the examples for carbon are the atomic weights of the isotopes. C12 has 6 protons and 6 neutrons in its nucleus, so its atomic weight is 12. C14 has 6 protons and 8 neutrons in its nucleus, so its atomic weight is 14. Two minerals that have the same chemical composition but different crystal lattice structures are called polymorphs. For example, graphite and diamond are both pure carbon, but the way the carbon atoms are arranged is completely different, giving rise to their very different chemical and physical properties (see images below). In graphite, the carbon atoms are arranged in sheets that are weakly bonded to each other. In a diamond, the lattice structure involves a much stronger bond framework.
Silicate minerals
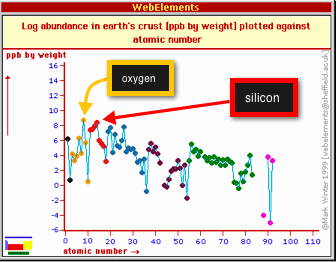
Not all of the elements in the Periodic Table are particularly common in the Earth's crust. Most minerals are formed from different arrangements of just a handful of the most commonly occurring elements. By weight, the two most abundant elements in the crust are oxygen and silicon. The figure below shows the abundance of elements in the crust as parts-per-billion by weight plotted vs. the atomic number of the element. See that oxygen (atomic number = 8, shown by one of the yellow dots) and silicon (atomic number = 14, shown by one of the red dots) are the highest.
Tectonic Setting of La Primavera Caldera
In order to identify the unique suite of minerals found in the soil on Camarena's body, we must first acquire some knowledge about the tectonic history of the area. Then we can narrow our scope to the actual volcano that produced the minerals in question.

Below is a map of the plate boundaries near the western coast of southern Mexico. See if you can apply what you've learned about plate boundaries and map-reading from previous lessons to make a mental picture of the interactions of all the plates and faults depicted on the map, then watch my screencast to hear me explain what I see when I look at this map.
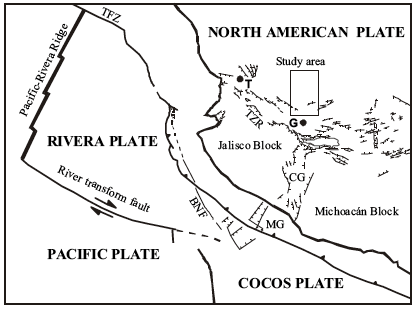
Click here for transcript
What do I see when I look at this figure? There are four plates shown here. The North American plate, the Rivera plate, the Pacific plate and the Cocos plate. Let us make sure we understand how they are all moving relative to each other. Over here there is a ridge between the Pacific plate and the Rivera plate. That is a divergent boundary. So, we can draw some arrows to show which way they are moving. This is a spreading center. These arrows I have drawn luckily jive with the arrows on the transform fault down here between the Rivera plate and the Pacific plate. The Rivera plate is moving from west to east and the Pacific plate is swinging up to the east. This is a kind of complicated area because this is where three plates come together. The authors who drew this figure were not sure exactly how it worked. Since there are some normal faults here my sense is that the Rivera plate and the Cocos plate must have some motion between them that might be pulling apart. Where exactly this boundary is that is between all these plates is kind of complicated. There is still the Cocos plate and the North American plate and there is a great big subduction zone that runs along like this. I will draw the teeth on it a little bigger so you can see them. The teeth are drawn on the overriding plate. That means we have a subduction zone in which the Cocos plate is heading this way and the North American plate is heading that way. The Cocos plate, and the Rivera plate at an oblique angle are diving under the North American plate at this boundary. This boundary here that I am marking in red is the boundary between the ocean and the land. It is the shoreline. The North American plate is everything on land plus this amount of continental shelf before you get to the trench. And then the Cocos plate, the Rivera plate, and the Pacific plate are all out in the ocean. This big subduction zone is probably the most important thing for our discussion. As these plates go down this trench, they are causing volcanism and uplift back here on the continent. That is where the Primavera caldera is located.
Volcanic history: basalt and rhyolite
The map below zooms in on a section of the first map (note that the rectangular dotted line corresponds to the rectangular box on the first map) and gives details about the volcanic history of this region. We can see from this map that this has been quite an active region volcanically. There are two distinct episodes of volcanism mapped. The shaded areas show basalt flows that have been dated between 9 to 11 million years old. On top of that is a younger (less than 7 million years old) deposit of rhyolitic lava. Each little dot is a separate volcanic vent.
Vents and collapsed calderas
The difference between a vent and a volcano is that a vent emits volcanic products (gas, lava, ash, etc.) but is not necessarily a mountain. Vents can be minor and often occur on the flanks of active volcanoes where lava and gas have found a different weak spot to escape from other than the main crater. Another feature on the map below is the number of collapsed calderas. These are shown as ring-shaped normal faults with the dip lines pointing inwards to show a circular depression. Collapsed calderas are formed when a large explosive volcanic eruption has blown away the magma chamber as well as the entire volcano, leaving behind a giant crater-shaped depression. Many of these calderas will still have active vents or resurgent volcanoes at their centers. Follow along with my screencast to hear me describe the details of the map.
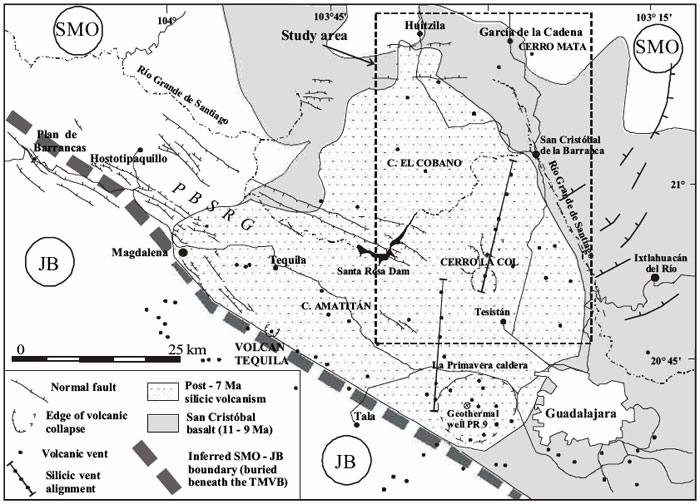
Click here for transcript
Here is a more detailed map of subduction zone volcanism on the west coast of Mexico in the Guadalajara region. Let us orient ourselves. Here is the metropolis of Guadalajara. Here is the town of Tequila over here. True fact for the day. The Jose Cuervo distillery is actually in this town. To get a volcanic history of this region let us go back about eleven million years. That is when the San Cristobal basalt was erupting. That eruption spread out over everything within this boundary here given by the darker shading in this figures. It goes off the map to the southeast and all the way back around so it covers everything within there. Basaltic eruptions are usually high temperature and low viscosity which means they flow pretty readily. Think of Hawaii. And they cover everything. All this area was covered by basalt. Then about seven million years ago the character of the lavas coming out of these volcanoes changed. They became more silicic. An example of a silicic lava is a rhyolite. The area covered by those lavas is shown by the stippled region and I am outlining it, too. Rhyolites are much more viscous. They melt at a lower temperature, and so they are kind of like the toothpaste of lavas. They are so viscous and they often have a lot of gas trapped in them that they lead to very explosive eruptions. Sometimes they are so explosive that they blow away the entire mountain during the eruption. There is evidence that that did happen here. All these circular features denoted like this are collapsed calderas. These happen when you get a giant eruption that blows away the entire mountain and leaves a little depression behind. Volcanism can still happen in there. La Primavera caldera is one of these little calderas and it is right here. It is in a line of other rhyolitic vents. Here is another one. All these other little dots are other rhyolite vents. So this was a very very active region.
Viscosity and Rhyolitic lava
As we were just discussing, collapsed calderas are formed when a large explosive volcanic eruption has blown away the magma chamber as well as the entire volcano, leaving behind a giant crater-shaped depression. Often, calderas are associated with rhyolitic lavas because rhyolite, with its high silica content, is viscous. See the chart below for a comparison of the viscosity of different lavas. Viscosity is a measure of resistance to flow. Lavas with high viscosity tend to form steep-sided volcanoes and often the lava cools right on top of the main vent, essentially plugging it up. Then, pressure builds up in the magma chamber as gases, ash, and lava want to escape but can't. When the pressure finally exceeds the strength of the plug and the eruption happens, it may be so violent that an extremely large volume of ground is blown to smithereens. Historically, large and destructive eruptions, such as Krakatoa and Tambora, happened this way. Note on the map above the sheer size of some of the calderas in the stipple-shaded region that marks the rhyolitic lava flows. La Primavera caldera, at the heart of our story, is shown as one of the many silicic vents in this region.
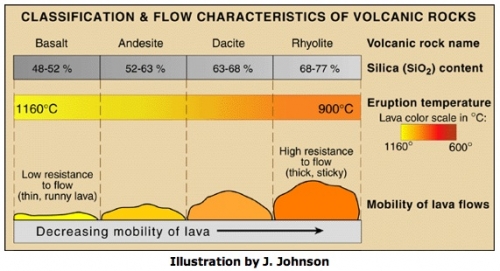
Temperature and composition both affect viscosity. The same substance at higher temperature will usually be less viscous. Think of an ordinary substance like candle wax. When you heat it up it gets more runny, but it is always wax, it has not changed what it is made out of. Two different mineralogies will normally have different viscosities depending on their silica content. The higher the silica content, the higher the viscosity. But note the actual numbers in the classification chart! Geologists talk about basalt as "low silica" but that's only in comparison to other lavas. In fact basalt is about 50% silica!
Bosque la Primavera
Below is a photo of the piney, mountainous state park in Jalisco described in "Death of an Agent."

The photo below shows an outcrop of rhyolite tuff. A tuff is deposited as an air-fall ash layer from an explosive eruption. Up close, you'd see that each grain is full of air bubbles that were trapped in the rock particles when the ash cooled as it flew through the air. The scale of this outcrop gives an indication of the massive eruption that produced this ash layer.

Mineralogy of La Primavera Caldera
The story of two different volcanic soils
The FBI geologists in "Death of Agent" were able to prove that Camarena did not die at the Bravo family ranch because the soil on his body was not at all like the soil at the Bravo ranch. Both soils derived from the weathered products of volcanoes, but the volcanoes couldn't have been the same because the mineralogy was different. The soil from the Bravo ranch was described as containing a lot of obsidian (see image below). Obsidian is a volcanic glass. The obsidian from the Bravo ranch was "globular" meaning that it was probably eroded and rounded by water.
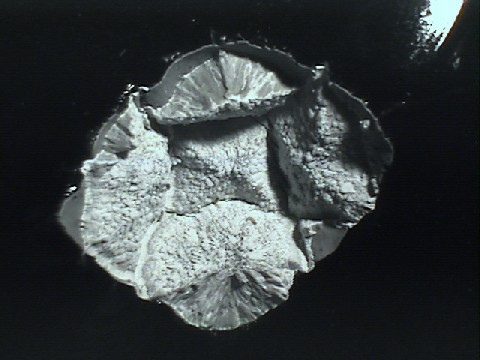
In contrast, the soil on Camarena's body was rhyolite (see image below). It is described as an ash that probably cooled as it flew through the air from an explosive eruption. This process creates small particle sizes and highly vesiculated (full of air bubbles) grains.
What is rhyolite?
What is the mineralogic composition of rhyolite, and specifically this particular rhyolite? Rhyolite is analogous to its more commonly known cousin, granite. The difference is that granite is an intrusive igneous rock that cools underground without erupting, whereas rhyolite is an extrusive igneous rock that forms a lava and cools out of the ground after an eruption. Otherwise, mineralogically speaking, the two rocks have the same composition. Rhyolite has the highest silica (SiO2) content of the extrusive igneous rocks, but it also has lesser components of some other compounds such as aluminum, potassium, sodium, magnesium and iron oxides. See the chart below for the average breakdown of compounds in each of the four most common extrusive igneous rocks.
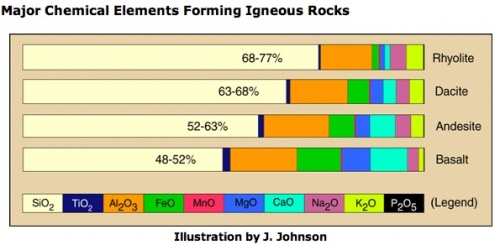
If the soil on Camarena's body had been just a run-of-the-mill rhyolite, his original burial spot would have been impossible to locate. Handily, there were a few diagnostic minerals that were part of the assemblage, and luckily these minerals had been studied and the results published by earlier field geologists. The other minerals found on Camarena’s body were cristobalite (clear and “opalized”), bixbyite, and rose quartz.
Cristobalite is a high-temperature polymorph of quartz often found in volcanic deposits. Remember from earlier in this lesson that polymorphs have the same chemical formula but different lattice structures. Below is a phase diagram of silica showing the different pressure and temperature regimes that produce its different polymorphs.
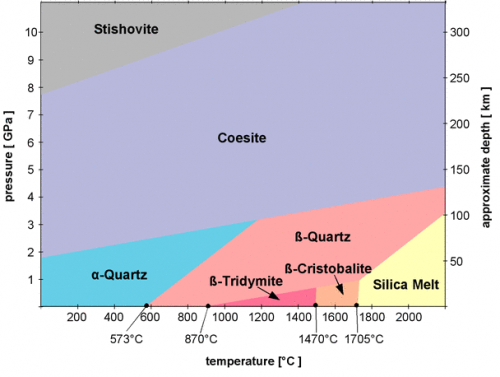
Rose quartz also has the chemical formula SiO2. This is the same formula as regular quartz and cristobalite. So why do they look different? Pure quartz is colorless. However, quartz often contains impurities. In the case of rose quartz, the impurities are titanium and iron. The impurities are at the parts-per-million or parts-per-billion level, so they do not get written in the chemical formula, even though their presence completely changes the look of the mineral!
Bixbyite is manganese iron oxide (Mn,Fe)2O3. It is not such a common mineral, but when it is found, it is usually found in rhyolite deposits.

Recap
We know from this case study where the original burial spot of Enrique Camarena was located because of the unique mineral assemblage of the soil adhering to his body. What do we know about this soil? It was rhyolite. This rhyolite has several characteristics that helped the FBI geologists narrow down its provenance. The crystals were tiny and full of air bubbles, which meant that they probably came from a volcano that had erupted explosively because the small crystals would have cooled as they flew through the air. This knowledge led them to look at the region near the subduction zone west of Guadalajara where all those collapsed calderas indicated past explosive eruptions. They knew they needed to look in a mountainous region because the grains were still sharp and comparatively unweathered. They also knew they needed to find a rhyolite that contained some interesting minor minerals in specific percentages: cristobalite, rose quartz, and bixbyite.
Create Your Own Forensics Lab
Activity
In this activity, I'd like you to create a forensic mineralogy lab or lesson. Make it short and simple (just one or two class periods in length). If you have big ideas for a longer, more involved project, that is fine—why not save that for the course capstone project (Lesson 8) when your assignment is to create a longer lesson?
An example to get you thinking
I made a really simple lab for an undergrad course in which students looked through a low-powered microscope at three samples of sand. One was synthetic sand from a playground sandbox, one was pure quartz with a very narrow grain size ordered from the US Silica Company, and one came from a beach in North Carolina. The students had to figure out which sample was which based on their observations. I had them make some drawings of the grains, and then make educated guesses about the mineralogy with the right reference books at their disposal.
Directions
- Write up your lesson plan. Your lesson plan should include the following:
- A brief overview of what will be taught and why. This overview should include a statement about the background information students will need to know in order to complete your lab successfully.
- A set of learning objectives. (What will your students know or be able to do at the end of your lesson?)
- A description of your plan. (What will the students do?)
- A list of necessary materials. (Feel free to be creative here. This is basically a hypothetical assignment, so if you think you need some equipment or other materials that you don't have immediate access to, don't let that stop you from including it!)
- A list of deliverables. (What will the students turn in? How will you know if they learned what you wanted them to learn?) Note: You need to include a "key" or set of answers to any follow-up questions or other material students will hypothetically turn in. That will help me assess the accuracy of the science you are presenting and it will also give me a clue about the level of thinking you expect from your students.
- An evaluation rubric. (So that another teacher can assess the students in the manner that you intended.)
- Save an electronic version of your activity and name it like this:
L5_forensicslab_AccessAccountID_LastName.doc (or your file extension).
For example, former Cardinals outfielder and hall of famer Stan "The Man" Musial would name his file "L5_forensicslab_sfm6_musial.doc"
Submitting your work
- Upload your lesson plan file to the Canvas assignment "Forensics Lab" by the due date specified on the first page of this lesson.
Grading rubric
Note on Grading: I am interested in the scientific accuracy of your exercise. I am not going to base my grade on whether you have constructed a lesson plan in some special way (as long as all the components listed above are there). My assumption is that those of you who are teachers already know how to write a lesson plan. For those who are not teachers, I am not going to instruct you on correct lesson-plan making here. However, I am a scientist, so if the facts are not right, or could use clarification, I can assist with that.
- An "A" forensics lesson is complete, clear, and organized. It contains all the components listed above. The science is accurate. I can follow your instructions in the way that you intended without a lot of guesswork (It's fine if part of the lab involves making students guess and hypothesize. I'm talking about whether your directions are clear or not). The lesson is well-designed and would elicit the appropriate amount of thinking and interpretation on the part of the intended audience. Your lesson shows independent thinking.
- A "B" forensics lesson is like that of an "A" project, except that its directions may not be clear enough that I can follow them without having to guess a little bit about your exact intentions. A "B" write-up is complete and contains all the components listed above.
- A "C" forensics lesson may have clarity problems, leading me to have to guess how to follow your instructions. A "C" write-up may also be incomplete with some of the assignment components missing. The science may not be accurate.
- A "D" forensics lesson has such badly written directions that I can't even begin to guess how to follow your instructions. A "D" write-up may be significantly incomplete and it may contain gross factual errors.
Additional Resources and Bibliography
Additional resources
- USGS volcano hazards page [13]
- Science of Sand [14]
Bibliography
Ferrari, L., Pasquaré, G., Venegas-Salgado, S., Romero-Ríos, F. (2000). Geology of the western Mexican Volcanic Belt and adjacent Sierra Madre Occidental and Jalisco block. Geological Society of America, Special Paper, 334, pp. 65-84.
Ferrari, L., Rosas-Elguera, J. (2000). Late Miocene to Quaternary extension at the northern boundary of the Jalisco block, western Mexico: The Tepic-Zacoalco rift revised. Geological Society of America, Special Paper, 334, pp. 41-64.
Murray, Raymond C. (2004). Evidence from the Earth: Forensic geology and Criminal Investigation. Missoula, MT: Mountain Press Publishing Company, p. 226
Rossotti, A., Ferrari, L., López-Martínez, M., & Rosas -Elguera, J. (2002). Geology of the boundary between the Sierra Madre Occidental and the Trans-Mexican Volcanic Belt in the Guadalajara region, western Mexico. Revista Mexicana de Ciencias Geologicas, 19, pp. 1-15.
Do you have another reading or Web site about these topics that you have found useful? Share it in our Teaching/Learning Discussion!
Summary and Final Tasks
I chose a case study to highlight the Neat-o Interdisciplinary Idea for this lesson: Forensics. I like introducing the subject of mineralogy this way because I think it is more exciting to see a real-life example of why you might want to know how to classify rocks and minerals rather than just memorizing a list of classifications, vocabulary, and chemical formulas.
While memorization does have its place and studying science does require learning some new vocabulary, putting these ideas into the context of a true story can make learning seem less burdensome and more like a discovery process--don't you agree?
Reminder - Complete all of the lesson tasks!
You have finished Lesson 5. Double-check the list of requirements on the Lesson 5 Overview page to make sure you have completed all of the activities listed there before beginning the next lesson.
Tell us about it!
If you have anything you'd like to comment on, or add to, the lesson materials, feel free to post your thoughts to our next Teaching/Learning discussion. For example, are you grateful that you were not unlucky enough to stumble onto some traffickers while hiking in Mexico in the 1980s?