Unit 3, Lesson 3: Climate (1)
Lesson 3 Introduction

Atmospheric processes and climate (primarily precipitation and temperature) may be the most influential of the factors involved in soil formation because they control weathering over large geographic regions. Bedrock parent material is subjected to physical weathering and disintegration through expansion and contraction associated with wide temperature fluctuations. The presence of water enhances this effect through freezing, thawing, and chemical processes—chemical weathering is accelerated under warm climate settings. In addition, solute and particulate (dust) deposition from the atmosphere plays an important role in Critical Zone and soil biogeochemistry. While continents are more impacted by atmospheric deposition, strong contrasts exist between bedrock and atmospheric sources in ocean island soils.
Climate, as one of the state factors of soil formation, is controlled to a large extent by atmospheric processes dependent on the latitudinal position on Earth. To properly understand the varying effects of climate on the Critical Zone, we must understand Earth's atmosphere and the range of phenomena that occurs within it. The focus of our next three lessons (Lessons 3, 4, and 5) is on understanding the atmosphere and climate systems. In Lesson 3, we will explore the basic structure of the atmosphere, the carbon cycle, basic atmospheric chemistry, atmospheric carbon dioxide and greenhouse gases, radiative forcing, and physical climate processes and feedbacks. In Lesson 4, we will turn our attention toward the evolution of Earth's atmosphere and ocean system through geologic time, specifically focused on our understanding of paleoclimatology (ancient climates). In Lessons 4 and 5, we will consider potential lessons from this knowledge of ancient climate and regional climate issues, in particular how this knowledge can aid our predictions of, and planning for, future climate change. Finally, we will consider how all this knowledge of past, present, and future climate processes is linked to soil formation and the Critical Zone, a topic we will return to in the final lesson (Lesson 12) of this course.
Watch this!
I visited the Adirondacks in upstate New York to examine climate, the hydrologic cycle, and some of the ways in which the forests interact with the Critical Zone during the winter season. Take a look at the video clip below of my visit to the Adirondacks.
Video: A Visit to the Adirondacks with Tim White (3:10)
[MUSIC PLAYING]
TIM WHITE: Hello. I've driven about nine hours north from State College into the northern part of the Adirondack Park in upstate New York. And I'm here to visit my sister, who I've just skied with, out onto Middle Saranac Lake. In the background, you can see of our favorite mountains-- Ampersand Mountain. This is Bodie stumbling around in front of us.
But I thought that while I'm here, I would take advantage of the wintry weather that they're experiencing here that we're not having in central Pennsylvania, to talk to you a little bit about climate and also about the hydrologic cycle and some of the ways in which the forests interact with the critical zone during the winter season.
The difference between weather and climate is an elusive concept for some people, so I'm going to try to describe it to you.
Recall that just moments ago, the sky was clear, and the sun was out to the east. But now, we're in the middle of a nice snowfall. This is an example of the weather. Weather, to me, describes the physical conditions in the atmosphere, and those conditions can change rapidly on time scales of hours to days from region to region and also through time.
Climate, on the other hand, describes prevailing weather trends in a region typically considered over years to decades. So while the weather is different today between the Adirondacks and central Pennsylvania, both regions are characterized as having a humid temperate climate.
One of the things that's unique about the Adirondacks compared to central Pennsylvania is that the mountains, and the topography, and the elevation. So this geographic component is important to understanding climate.
So mountainous regions further from the coastlines typically have colder, longer winters than areas closer to the coast at lower elevations like central Pennsylvania.
In mid- to high-latitude regions that experience a truly cold winter season, critical zone processes slow down and in some cases cease to function. For example, in the frozen Adirondacks, the water that in other seasons would percolate into the subsurface's ground water or flow through the rivers and lakes of the region, is temporarily stored as ice and snow at the surface until temperatures warm, thaw occurs, and critical zone processes begin anew.
We'll reconsider seasonality in later lessons when we discuss the hydrologic cycle and biotic functioning of the critical zone.
[MUSIC PLAYING]
Learning Objectives
By the end of the unit, successful students will be able to:
- Describe the components of the climate system and the basic natural and anthropogenic forces that affect the climate system, including the history of variability and change.
- Respond to questions like: "How much is the world warming?" "Is the recent warming unusual?" and "How rapidly has climate changed in the past?"
- Describe the C cycle and atmospheric chemistry.
- Evaluate C cycle and greenhouse gas models and future projections of atmospheric compositions.
- Determine the relative roles of ozone, aerosols, solar forcing, and land-use change in radiative forcing.
- Further explain the links between the atmosphere, hydrosphere, lithosphere, and biosphere processes and their role in Critical Zone health.
- Compare and contrast various models of future climate change and include region-specific issues.
- Explain how temperature and precipitation patterns are altered by natural events (e.g., El Niño and volcanic eruptions) and by human influences, including deforestation, urbanization, and the production of greenhouse gases (e.g., carbon dioxide and methane).
- Explain how changes in the geometry of Earth's orbital parameters relative to the Sun cause seasonality in climate.
- Describe the different climatic zones and the four major factors affecting climate: latitude, elevation, proximity to bodies of water, and position relative to mountains. Analyze data relating to the concentration of atmospheric CO2 over the past 100 years.
Questions?
If you have any questions, please post them to our Questions? discussion forum (not e-mail), located under the Discussions tab in Canvas. I will check that discussion forum daily to respond. While you are there, feel free to post your own responses if you, too, are able to help out a classmate.
Lesson Overview
To properly understand the varying effects of climate on the Critical Zone, we must understand Earth's atmosphere and the range of phenomena that occurs within it. In this lesson, we will explore the basic structure of the atmosphere, the carbon cycle, basic atmospheric chemistry, atmospheric carbon dioxide, and greenhouse gases, radiative forcing, physical climate processes and feedbacks, and regional climate issues. We will briefly consider the links between the atmosphere and the Critical Zone throughout the lesson.
As you read through the assigned material, you should always make note of and consider the potential link between the subject matter and the Critical Zone. For example, what effect does atmospheric chemistry have on the Critical Zone and how are they linked? We will return to considering these links in more detail later in this lesson and in Lesson 12.
Much of our understanding of the atmosphere and climate system derives from the recent focus on the impact of human society on the greenhouse gas content of the atmosphere and global climate change. The Intergovernmental Panel on Climate Change (IPCC) [2] (go there and read "About IPCC") is a scientific panel established by two organizations of the United Nations in 1988 to evaluate the risk of climate change from human activity. The main activity of the panel is to publish special reports based on assessments of peer-reviewed and published scientific literature—four reports have been published in three volumes each, the most recent in 2007. Much of the background information and reading for Lesson 2 was culled from the 3rd and 4th assessment reports.
Did you know . . . ?
In 2007, the IPCC received a Nobel Peace Prize for its work. Among the 2,000 contributors (including Al Gore), there were five Penn State faculty who shared in the prize: Michael Mann, Richard Alley, Bill Easterling, Klaus Keller, and Anne Thompson were all substantial contributors to the Intergovernmental Panel on Climate Change.
What will we learn about in Lesson 3?
- Fundamentals of the atmosphere
- Overview of the climate system
- C cycle and atmospheric CO2
- Basic atmospheric chemistry
- Greenhouse gases
- Radiative forcing
- Physical climate processes and feedbacks
- Regional climate issues
What is due for Lesson 3?
Lesson 3 will take us one week to complete. As you work your way through these online materials for Lesson 3, you will encounter additional reading assignments and hands-on exercises and activities. The chart below provides an overview of the requirements for Lesson 3. For assignment details, refer to the lesson page noted.
Please refer to the Calendar in Canvas for specific time frames and due dates.
| ACTIVITY | LOCATION | SUBMITTED FOR GRADING? |
|---|---|---|
| "Calculating Carbon Footprints" | page 6 | Post to the Lesson 3 - Carbon Footprint discussion forum, then discuss. |
Questions?
If you have any questions, please post them to our Questions? discussion forum (not e-mail), located under the Discussions tab in Canvas. I will check that discussion forum daily to respond. While you are there, feel free to post your own responses if you, too, are able to help out a classmate.
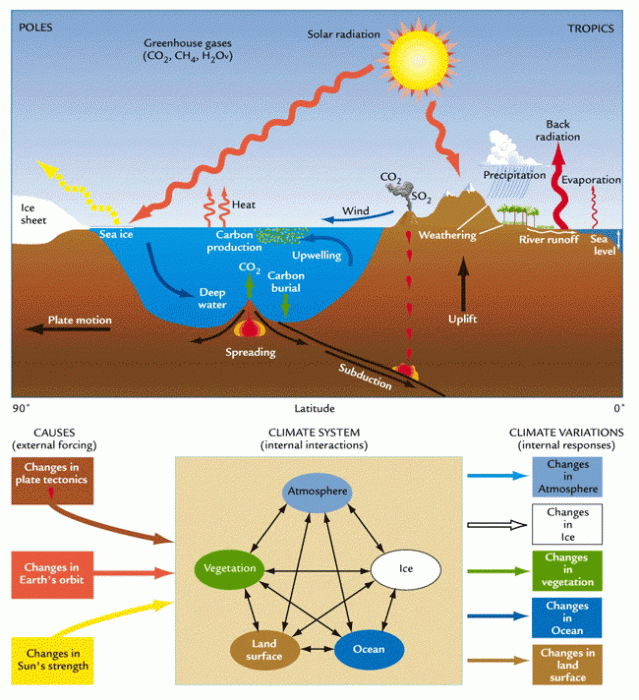
Climate Dynamics
The field of climate dynamics studies the processes that control the climate system and how it evolves. The climate system is controlled by the interaction of the atmosphere, the oceans, [3] the land surfaces, the cryosphere, and the biosphere, averaged over time-scales of weeks to centuries and millennia. Climate can be classified [4] into spatial zones based on, for example, regional similarities in temperature and precipitation—these zones [5] often contain similar biotic elements from region to region.
The primary energy that drives Earth's climate system and sustains life is solar heat [6] and light. The amount of solar energy that reaches Earth’s surface varies because of latitudinal position on the planet, internal solar processes, changes in Earth's orbit, Earth's albedo [7], changes on Earth such as major volcanic eruptions of dust that block the incoming solar energy, or changes in atmospheric composition. Earth's energy balance is complex. About 30% of incoming solar energy is reflected back to space, with 70% being absorbed by the atmosphere and surface of the planet.
To get a better overview of the climate system, there are three reading assignments I would like you to complete before we proceed. Once you have completed these three reading assignments, continue with this online lesson.
Reading Assignment
In succeeding portions of this lesson, you will explore some of the topics described in the chapter in more detail, but it is the generalized overview of the material presented in the chapter that I am most interested in seeing you master. On the first page of the chapter you will find a definition of the climate system as "an interactive system consisting of five major components: the atmosphere, the hydrosphere, the cryosphere, the land surface and the biosphere, forced or influenced by various external forcing mechanisms, the most important of which is the Sun." Confusingly, this sounds a lot like descriptions of the Critical Zone we read in Lesson 1! Remember: While the Critical Zone consists of portions of these "spheres," the central component of the Critical Zone is soil, the consequence of complex processes operating at the interface between the "spheres."
- Earth's Atmosphere [9]
Before we begin to explore some of the intricacies of Earth's climate, I would like you to review some basic information about the structure and composition of Earth's atmosphere, available on NOAA's Web site. The terminology presented there will provide you with the background to better understand the remainder of the lesson. - Introduction in the IPCC Fifth Assessment Report: "Climate Change 2013 The Physical Science Basis" [10]
This chapter provides an excellent description of climate entitled "Key concepts in Climate Science". Read the chapter, noting that:
- the glossary for the entire report is available at IPCC Glossary [11];
- the other chapters mentioned in the overview are available online but are not required reading for this lesson; and
- the final section of the chapter, entitled "A Road Map to this Report," is also not required reading for this lesson.
- Atmospheric Chemistry [12]
The chemistry of Earth's atmosphere is governed by numerous complex reactions studied by atmospheric scientists. Though the next portion of Lesson 2 is primarily focused on the carbon cycle, I would first like you to explore some of the most basic concepts of atmospheric chemistry presented at Wikipedia.
Want to learn more?
(Optional) Read Chapter 10 (pp. 419–460) in
- Wallace, J. M. (2006). Climate Dynamics. In Atmospheric Science: An Introductory Survey, International geophysics series. (2nd ed.). Amsterdam: Elsevier Academic Press.
This chapter is available through Library Reserves. Do not focus on the details of this chapter and do not worry about references to earlier chapters, figures, etc. Your primary goal should be a very generalized understanding of the climate system, always considering in the back of your mind what implications this information may have to the Critical Zone.
Check out these Climate Education Modules [13] for teachers. The website was created and is hosted by North Carolina State University, and pays particular attention to the southeast of the U.S. However, the website provides links to relevant National Science Education Standards and many pages include classroom activities.
Carbon Cycle and Atmospheric CO2
The Global Carbon Cycle
The biogeochemical cycle in which carbon is exchanged between Earth’s terrestrial biosphere, hydrosphere, geosphere, and atmosphere is called the carbon cycle. The global carbon budget is the balance of the fluxes of carbon between these four reservoirs. The terms source or sink define whether the net carbon flux is out of or into the reservoir, respectively.
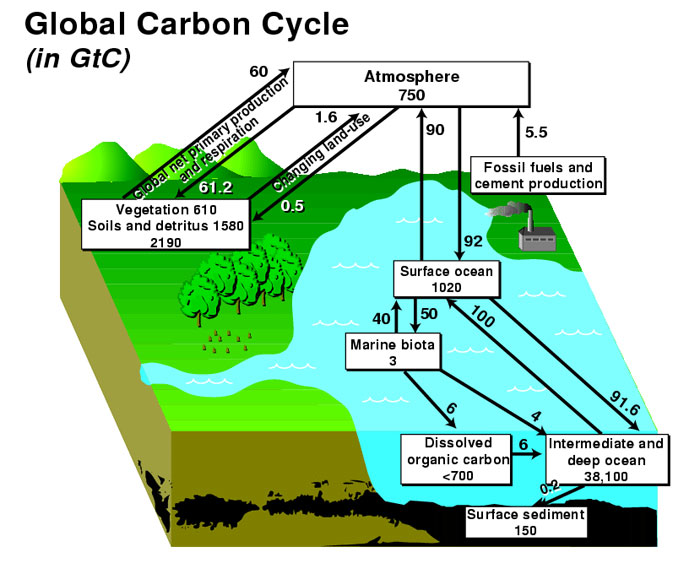
Atmospheric carbon dioxide
The carbon cycle is of interest to understanding climate because it includes two of the most important greenhouse gases: carbon dioxide (CO2) and methane (CH4). Most atmospheric carbon is in the form of CO2, while CH4 is present only in trace concentrations. Because CO2 is chemically inert, it is relatively well mixed within the atmosphere away from forest canopies, whereas CH4 is chemically active and is removed quickly from the atmosphere through oxidation to CO2 and water. The overall atmospheric concentration of these greenhouse gases has increased during the past century and contributed to global warming.
Carbon is removed from the atmosphere through:
- the photosynthetic conversion of carbon dioxide into carbohydrates by plants, releasing oxygen in the process;
- the formation of carbonic acid as circulating ocean surface waters cool near the poles, thereby absorbing more atmospheric CO2;
- the conversion of reduced carbon to organic tissues or carbonates to hard body parts, such as shells, by marine biota in ocean surface waters; and
- the reaction between carbonic acid and silicate rock that leads to the production of bicarbonate ions, which are carried to the ocean and eventually deposited in marine carbonate sediments.
Carbon can be input to the atmosphere by a variety of processes, including:
- plant and animal respiration, an exothermic reaction that involves the breakdown of organic molecules into CO2 and water;
- decay by fungi and bacteria that break down carbon compounds in dead animals and plants and convert the carbon to CO2 if oxygen is present, or methane if not;
- organic matter combustion (including deforestation and burning fossil fuels), which oxidizes carbon-producing CO2;
- cement production, when limestone (calcium carbonate) is heated to produce lime (calcium oxide), a component of cement, and CO2 is released;
- release of dissolved CO2 back to the atmosphere through warming of surface ocean water; and
- release of water vapor, carbon dioxide, and sulfur dioxide from volcanic eruptions and metamorphism.
Oceanic Carbon
Inorganic carbon is readily exchanged between the atmosphere and ocean, exerting an important control on the pH of ocean water. Carbon is released to the atmosphere at oceanic upwelling sites, whereas regions of downwelling transfer carbon from the atmosphere to the ocean. When carbon (CO2) enters the ocean, carbonic acid is formed by the reaction: CO2 + H2O = H2CO3. Carbonic acid dissociates to form bicarbonate ions (HCO3-), the form in which most of the carbon in the oceans exists; lesser amounts of carbon exist as carbonic acid (H2CO3 or dissolved CO2), and carbonate ions (CO32-) paired with calcium and magnesium and other cations. Marine organisms build their skeletons and shells out of the minerals calcite and aragonite (CaCO3) through the incorporation of bicarbonate ions. These minerals dissolve after the death of the organism, but some of the material settles to the sea floor where it can be buried and stored in the form of limestone.
Carbon in the Biosphere
Carbon is an essential part of life on Earth. It plays an important role in the structure, biochemistry, and nutrition of all living cells. Autotrophs are organisms that produce their own organic compounds using carbon dioxide from the air or water they live in. This lifestyle requires an external source of energy, for example, the absorption of solar radiation in the process called photosynthesis, or the exploitation of chemical energy sources in a process called chemosynthesis. The most important autotrophs for the carbon cycle are trees in forests and phytoplankton in the ocean.
Large quantities of carbon pass between the atmosphere and biosphere on short time-scales: the removal of atmospheric carbon occurs during photosynthesis, following the reaction CO2 + H2O = CH2O + O2, while most carbon leaves the biosphere through respiration, a reversal of the previous reaction in which an amount of energy equivalent to that absorbed during photosynthesis is released as heat. When oxygen is present, aerobic respiration occurs, which releases carbon dioxide into the surrounding air or water. Otherwise, anaerobic respiration occurs and releases methane into the surrounding environment, which eventually makes its way into the atmosphere or hydrosphere.
The biosphere is capable of storing ~10% of atmospheric carbon at any given time. However, carbon storage in the biosphere is influenced by a number of processes on different time-scales: while net primary productivity [15] follows seasonal and annual cycles, carbon can be stored up to several hundreds of years in trees and up to thousands of years in soils. Changes in those long-term carbon pools may thus affect global climate change (view this example of changes to soil) [16].
Carbon in the Geosphere
Organic and inorganic carbon reservoirs in Earth's crust are large with long residence times. Carbon enters the geosphere through the biosphere when dead organic matter (such as peat or marine algae) becomes incorporated into fossil fuels like coal and organic-matter-rich oil and gas source rocks, and when shells of calcium carbonate become limestone through the process of sedimentation briefly described above. These carbon reservoirs can remain intact, that is, the carbon can remain stored within them, for many millions of years. Eventually, most rocks are uplifted and subjected to exposure to the atmosphere where they are weathered and eroded, or they are subducted, metamorphosed, and erupted through volcanoes, returning the stored carbon back into the atmosphere, ocean, and biosphere. Our society's dependence on fossil fuels bypasses this natural process by moving as much carbon from the geosphere to the atmosphere in a single year as what might otherwise require hundreds of thousands or millions of years. In Pennsylvania, when we strip mine and burn coal we are in effect releasing the atmospheric carbon dioxide and stored energy of the sun that has been buried for over 300,000,000 years!
Watch this!
On a trip to Bear Meadows, near the University Park campus of Penn State, I describe atmospheric carbon and organic carbon storage in the Critical Zone.
Video: A Trip to Bear Meadows (5:40)
TIM WHITE: I've traveled to Bear Meadows, a national natural landmark, about eight miles southeast of Penn State's main campus in central Pennsylvania. I've brought you here today to describe the relationship between atmospheric carbon dioxide content and organic carbon storage at the surface of the Earth within the critical zone. Let's go have a look at the bog.
Bear Meadows is a peat bog that began forming nearly 10,000 years ago, shortly after northwestern and northeastern Pennsylvania were covered by vast ice sheets that extended far to the north into the Hudson Bay region and Canada. The closest those ice sheets reached to State College and the Penn State campus here was northeast of Williamsport, nearly 30 miles away. One of the tasks I'd like to accomplish while we're here is to probe the peat in the bog and have a look at what it's made of, and so to do that I'm going to walk along the edge of the bog-- in the background you can see where I'm going to go-- and I'm going to get my soil auger so I have a way to probe and sample the peat.
Recall that plants use water, atmospheric carbon dioxide, and energy from the sun to build their organic framework through photosynthesis. Therefore, plants draw down atmospheric carbon dioxide as part of their life habit. When plants die, the organic matter is oxidized and carbon dioxide is returned to the atmosphere.
This wet, boggy setting is perfect for the preservation of organic matter and organic carbon. If you look beneath the water's surface, you can clearly see various organic constituents related to the plants that live around in the bog. I can see maple leaves and abundant roots associated with these grasses. If plants die and their various organic constituents are preserved, as is the case here in the bog, the original carbon in the atmospheric carbon dioxide that was photosynthesized by the living plant is also stored or preserved, rather than being returned to the atmosphere through decay. We call the site of such processes a carbon sink.
I'm probing the peat with an auger to see how deep I can get to the base of the bog to see if I can get a peat sample from the base of the bog here. You can see that I've penetrated over a meter of the peat and I've obtained a sample from the base of the bog. And you can see that the peat down there is made up of a fine organic muck, along with a number of roots and some grass leaves, similar to what you'd find right here at the surface.
Further out in the bog, there are places where 15 feet of peat have accumulated over the last 8,000 or so years. That means that 8,000 years ago, carbon was removed from the ancient atmosphere, stored here in the peat, and remains today. One last thought before we leave and that relates to the time scale of organic carbon storage in the critical zone. I've told you that organic matter has been accumulating here over the last 8,000 years, but this setting exists within the Appalachian Mountains and the fate of mountains ultimately is to be eroded, which is happening to these mountains today. So while this organic carbon remains stored here now, over long geologic time scales-- hundreds of thousands to millions of years-- this peat will be oxidized and that ancient organic carbon would be returned to our modern atmosphere.
Next, visit a Pennsylvania coal seam and nearby electric generating station to learn about the relationship between Bear Meadows and the burning of coal.
Video: Visit to a Pennsylvania Coal Seam (02:52)
Coal Seams and Electricity Generation near Shawville, PA
[Tim is standing next to a coal seam.]
The geologic, economic, and cultural history of Pennsylvania is steeped in this state's naturally occurring abundance of coal. To give students an opportunity to view coal in its natural setting, I like to come here, Curwensville Dam, about 45 miles northwest of State College.
[Here you can see two coal seams, one directly in front of me and one a little bit further up on the slope. Coal is peat that's been deeply buried in the earth and subjected to high pressures and temperatures. Remember at Bear Meadows, we looked at peat and we were able to see the various plant constituents from which it's made.
We also discussed that those plant constituents at the bottom of the bog were 8,000 years old and contained 8,000-year-old atmospheric carbon dioxide. These coal seams are over 300 million years old. They formed from vast peat swamps that existed along the margins of a large interior seaway that flooded much of North America at the time. So when we dig up and mine these coals, and use them to fire our electric generating stations and the computers you're using today to view this video, we're releasing 300 million-year-old carbon dioxide into our present-day atmosphere.
[looking out a car window at the Shawville Generating Station]
We're approaching the Shawville Generating Station. At this power plant, which is just about 15 miles from the Curwensville coal outcrop we just visited, coal is burned to produce electricity for local and regional consumption.
[Dr. White standing in front of the generating station]
At numerous coal-fired electric generating stations around the state, coal is burned to produce electricity. And it's through this process that Pennsylvania produces 1% of total global human carbon dioxide emissions in any given year.
In subsequent lessons, we will examine and discuss the effects of anthropogenic carbon dioxide emissions and climate change on critical zone processes.
Greenhouse Gases, Aerosols and Radiative Forcing
Earth's surface temperature and climate are controlled by the amount of radiation received from the Sun: when incoming radiation is greater than outgoing radiation the Earth is warmed, and vice versa. Changes in the balance between incoming and outgoing radiation are referred to as radiative forcing. While the intensity or energy output from the Sun varies through time, other Earth-bound processes affect radiative forcing. For example, the gaseous and particulate composition of Earth's atmosphere plays an important role in controlling the energy balance. Changes in the natural landscape and human land-use also influence radiative forcing.
Reading Assignment
- Visit the EPA's Atmospheric Changes [17] site to learn more about the interaction between the greenhouse gas and aerosol composition of Earth's atmosphere and radiative forcing. Note: when you go to this site you'll find that the content is no longer available - I left this link in place so that you can experience the manner in which politics can play a role in science. The same goes on the next page, and assignment, relating to the use of information once provided by the US EPA.
- See https://19january2017snapshot.epa.gov/climate-change-science/causes-climate-change_.html [18] for what used to be the above link.
- Also, visit the UNEP's Radiative Forcing [19] page to better understand the effects of human activity on the composition of the atmosphere and Earth's climate.
Calculating Carbon Footprints
Three processes have contributed to human-induced increases in the carbon dioxide content of Earth’s atmosphere. Two of these processes concern emissions: increasing global economic activity and an increase in the use of fossil fuels to drive the economy. The third process is a suggested decline in the efficiency of CO2 sinks on land and oceans in absorbing human emissions. These changes characterize a carbon cycle that is forcing stronger-than-expected and sooner-than-expected climate change.
The United States ranks first in total and per capita emissions among the top twenty carbon dioxide-emitting nations and is responsible for more than 22% of the total annual carbon dioxide introduced to the atmosphere by human activity. For perspective, the United States population stands at ~4.5% of the total global population—our society’s profligate lifestyle is supported by our consumption of a greater proportion of Earth’s resources than the rest of humanity and therefore translates to a greater share of the waste products of human activities, in this case, human inputs of carbon dioxide to the atmosphere.
Although the United States Federal government has a policy regarding greenhouse gas emissions and climate change (see U.S. Climate Policy and Actions [20]), each citizen, church, school, and business—all of us—can play a role in reducing our nation’s carbon dioxide input to the atmosphere. Reducing the amount of energy we use is one important step in the effort to reduce carbon dioxide emissions. To do so, we must first inventory our carbon dioxide emissions by identifying the energy-intensive activities we partake in throughout our day-to-day lives.
Go here to find information that was once available through the link above: January 19, 2018 Snapshot [21].
You may also be interested in exploring the U.S. Climate Resilience Toolkit [22], designed to help citizens, communities, businesses, and others to manage climate-related risks and to improve resilience to extreme events.
Activity
Directions
For this activity, you are to use three different "calculators" to calculate your own carbon footprint. As you use the three calculators, consider these questions:
- How do the results compare between calculators?
- To what activity(ies) do you attribute a majority of your personal emissions?
- How might you best accomplish a reduction in your carbon dioxide emissions?
- What percentage of total U.S. emissions derive from household emissions versus industry, government, etc.?
- How might widespread reduction (halved) of U.S. household emissions contribute to a reduction in total U.S. emissions?
- In a general sense, how might a business-as-usual approach versus a reductionist approach influence climate change and Critical Zone processes?
- Are there any activities you can engage in that will benefit soil, the central component of the Critical Zone, at the same time as reducing your personal carbon footprint?
I ask you to be honest, with yourselves, with me, and with your classmates. Remember: On average, the U.S. citizenry’s carbon dioxide emissions are grossly out of balance with the rest of Earth’s human inhabitants, so we all play a role in current emission levels. Let’s see what we can learn through a non-judgmental assessment of our community’s lifestyles.
- To begin, use the calculator located at the following Environmental Protection Agency (EPA) Web site:
- Personal Emissions Calculator [23]
- The EPA calculator is no longer easily available, if at all. Please Google Carbon Footprint calculator and choose a calculator to go along with choosing others described in step 2. Provide a sentence in your report that names the calculator and provide the relevant URL.
- Next, use two more calculators, either from the list found on the EPA site or using the one below:
- Once you have accomplished these simple personal calculations, use the same calculators you just used to calculate the carbon footprint for your school or business. Address similar questions as above.
Submitting your work
- Enter the "Lesson 3 - Carbon Footprint" discussion forum in Canvas.
- Post a summary of your responses to the questions above for both your personal carbon footprint and your school/business carbon footprint.
- Read postings by other EARTH 530 students to compare your ideas and experiences.
- Discuss! Respond to at least one other posting by asking for clarification, asking a follow-up question, expanding on what has already been said, etc.
Grading Criteria
You will be graded on the quality of your participation. See the grading rubric [25] for specifics on how this assignment will be graded.
Check this out!
Interested in determining the potential C footprint for a planned building construction or retrofit? See: the Green Footstep website [26] to learn how to plan and design for C neutrality.
Also, visit Global Footprint Network [27] for yet another C footprint calculator as well as an assessment of various country's standing with respect to their C emissions and more.
Summary and Final Tasks
Earth's atmospheric processes and climate may be the most influential factors involved in soil formation in the Critical Zone because processes like precipitation and temperature control weathering over large geographic regions. Once we grasp their importance we begin to properly understand the varying effects of climate on the Critical Zone. In this lesson, you read a lot of material describing these processes, learned about the Intergovernmental Panel on Climate Change (IPCC), performed a carbon footprint calculation, and applied knowledge gained during the calculation to consider Critical Zone-relevant issues of personal and societal behavior.
By now you should feel comfortable describing: components of the climate system and the basic natural and anthropogenic forces that affect the climate system; the carbon cycle and fundamental atmospheric chemistry; and radiative forcing of climate change. In the next lesson you will apply this knowledge to understanding paleoclimates (ancient climates), and from there determine how our knowledge of paleoclimates helps us to understand ongoing and future climate change.
Reminder—Review the Lesson 3 Overview
You have reached the end of Lesson 3! Double-check the list of requirements on the Lesson 3 Overview page to make sure you have completed all of the activities listed there.
Tell us about it!
If you have anything you'd like to comment on or add to, the lesson materials, feel free to share your thoughts with Tim. For example, what did you have the most trouble with in this lesson? Was there anything useful here that you'd like to try in your own classroom?