Lesson 4a: Fossil Fuels and Products of Combustion
Introduction to Fossil Fuels and Products of Combustion
In the first lesson on the world and the U.S. energy supply, we clearly established that the dependence on fossil fuels is still high (still at about 80 percent of the total energy in 2021).
In this section, we are going to look at what the fossil fuels are and the consequences when these fossil fuels are burnt.
As you may recall from an earlier lesson, these fuels, which we primarily depend on, were formed over millions of years by compression of organic material (plant and animal sources) prevented from decay and buried in the ground. They include:
- Coal
- Natural Gas
- Petroleum Oil
Fossil Fuel Elements
Fossil fuels are hydrocarbons comprised primarily of the following elements: carbon and hydrogen and some sulfur, nitrogen, oxygen, and mineral matter. Mineral matter turns into ash when burnt.
The composition and the amounts of these elements change for different fossil fuels (coal, petroleum, and natural gas), but the elements are the same. For example, there is more hydrogen in liquid fuels than in coal per unit mass.
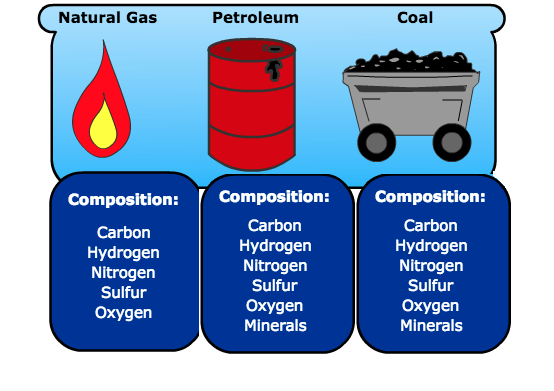
Natural gas is composed of carbon, hydrogen, nitrogen, sulfur, and oxygen.
Petroleum is composed of carbon, hydrogen, nitrogen, sulfur, oxygen, and minerals.
Coal is composed of carbon, hydrogen, nitrogen, sulfur, oxygen, and minerals.
Combustion is rapid oxidation of the fossil fuel’s elements resulting in the generation of heat. When these elements oxidize (or combine with oxygen), products of combustion are formed.
Instructions: Click on the purple hot spot shown above the piece of coal below to determine what products are formed from each during combustion.
Products of Combustion
Some of the fuel (hydrocarbon) may not completely burn during combustion and therefore is released into the atmosphere along with the products. The products that are formed during combustion of fossil fuels are shown in the image below:

Fossil fuels consisting mainly of carbon, hydrogen, nitrogen, sulfur, and oxygen produce the following products during combustion:
The primary pollutants are Carbon Monoxide (CO), Carbon Dioxide (CO2), Sulfur (SO2), Nitrogen Dioxide (NOx), Nitric Oxide (N2O), Volatile organic compounds (VOCs), and Hydrocarbons (HCs).
The particulate matter produced are Course particles less than 10 microns (PM10), Fine particles less than 2 microns (PM2.5), and Ammonia (NH3).
We will now look at six products of combustion:
- Carbon Dioxide
- Carbon Monoxide
- Sulfur Dioxide
- Nitrogen Oxides
- Lead
- Particulate Matter
Carbon Dioxide (CO2)
Carbon dioxide is one of the maor products of combustion with fossil fuels since carbon accounts for 60–90 percent of the mass of fuels that we burn.
China has emerged as the largest single emitter of energy-related CO2 emissions, surpassing the U.S. in carbon dioxide emissions back in 2010. Now, China emits more than 10 million metric tons while the U.S. hovers around 5 million metric tons. The chart below shows the trend in carbon dioxide emissions since 1980. For Asia and Oceania, and particularly for China and India, emissions can be seen to have increased significantly in the past two decades.
Click the Table, Map, and Chart tabs to see how CO2 emissions have changed over time and around the world:

In 2019, 29 % of CO2 emissions were from transportation, 25 % were from electricity production, 23 % were from industry processes the remaining quarter are from commercial, residential and agricultural applications.
Carbon Monoxide (CO)
If a carbon-based fuel and its products are not completely oxidized (i.e. not burned completely), carbon monoxide will be formed. Carbon monoxide, or CO, is a colorless, odorless gas. The figure below shows the contribution of various sources to the emissions of CO:
Carbon Monoxide is a component of motor vehicle exhaust, which contributes about 55 percent of all CO emissions nationwide. Other non-road engines and vehicles (such as construction equipment and boats) contribute about 22 percent of all CO emissions nationwide. Higher levels of CO generally occur in areas with heavy traffic congestion. In cities, 85 to 95 percent of all CO emissions may come from motor vehicle exhaust.
Other sources of CO emissions include industrial processes (such as metals processing and chemical manufacturing), residential wood burning, as well as natural sources such as forest fires. Woodstoves, gas stoves, cigarette smoke, and unvented gas and kerosene space heaters are sources of CO indoors.
The highest levels of CO in the outside air typically occur during the colder months of the year, when inversion conditions are more frequent. An inversion is an atmospheric condition that occurs when the air pollutants are trapped near the ground beneath a layer of warm air.
Sulfur Dioxide (SO2)
Sulfur dioxide, or SO2, belongs to the family of sulfur oxide gases (SOx). These gases dissolve easily in water. Sulfur is prevalent in all raw materials, including crude oil, coal, and ores that contain common metals, such as aluminum, copper, zinc, lead, and iron.
SOx gases are formed when fuel containing sulfur, such as coal and oil, is burned, and when gasoline is extracted from oil, or metals are extracted from ore. SO2 dissolves in water vapor to form acid and interacts with other gases and particles in the air to form sulfates and other products that can be harmful to people and their environment.
Nitrogen Oxides (NOx)
Nitrogen oxides, or NOx, is the generic term for a group of highly reactive gases, all of which contain nitrogen and oxygen in varying amounts. Many of the nitrogen oxides are colorless and odorless.
Nitrogen oxides form when fuel is burned at high temperatures, as in a combustion process. The primary sources of NOx are motor vehicles, electric utilities, and other industrial, commercial, and residential sources that burn fuels as shown in the figure below.
Although many of the nitrogen oxides are colorless and odorless, one common pollutant, nitrogen dioxide (NO2) along with particles in the air can often be seen as a reddish-brown layer over many urban areas.

Lead (Pb)
The major sources of lead emissions have historically been motor vehicles (such as cars and trucks) and industrial sources.
Due to the phase-out of leaded gasoline, metals processing is the major source of lead emissions to the air today. The highest levels of lead in air are generally found near lead smelters (devices that process lead ores). Other stationary sources are waste incinerators, utilities, and lead-acid battery manufacturers.

A man is pictured in a protective suit as he removes lead-based paint from the US Capitol Dome.

Lead is used in the manufacturing of many items, including
glass, rubber, paint, batteries, insecticides, plumbing, and
protective shielding for X-rays.
Particulate Matter (PM)
Particulate matter (PM) is the general term used to describe a mixture of solid particles and liquid droplets found in the air. Some particles are large enough to be seen as dust or dirt. Others are so small they can be detected only with an electron microscope.
Different sizes of Particles include:
- PM 2.5 describes the “fine” particles that are less than or equal to 2.5 µm (micro meter) in diameter.
- “Coarse fraction” particles are greater than 2.5 µm, but less than or equal to 10 µm in diameter.
- PM 10 refers to all particles less than or equal to 10 µm in diameter (about one-seventh the diameter of a human hair). PM can be emitted directly or formed in the atmosphere.
Different Sources of Particles include:
- "Primary" particles are formed from combustion sources and are emitted directly into the atmosphere. Examples of primary particles are dust from roads or black carbon (soot).
- "Secondary" particles are formed in the atmosphere from primary gaseous emissions. Examples of secondary particles are sulfates formed from SO2 emissions from power plants and industrial facilities; nitrates formed from NOx emissions from power plants, automobiles, and other combustion sources; and carbon formed from organic gas emissions from automobiles and industrial facilities.

The chemical composition of PM depends on location, time of year, and weather. Generally, primary particles make up coarse PM and secondary particles make up most of fine PM.
Health and Environmental Effects of Primary Pollutants
The pollutants that are emitted directly from a combustion process – or the products of combustion - are called “primary pollutants.” We just described these products earlier in the lesson, now we will look at their impact on the environment and human health.
Carbon Dioxide (CO2)
Carbon dioxide (CO2) is not a pollutant in the sense that would directly harm our health, but it is a proven greenhouse gas. It has an ability to absorb infrared radiation that is escaping from the surface of the earth, causing the atmosphere to warm up. Excessive emission of CO2 along with other greenhouse gases are linked to climate change, which is reaching a critical point.
Carbon Monoxide (CO)
As we learned earlier, Carbon monoxide, or CO, is a colorless, odorless and tasteless gas that is formed when carbon in fuel is not burned completely.
At much higher levels of exposure not commonly found in ambient air, CO can be poisonous, and even healthy individuals can be affected. Exposure to elevated levels of CO may result in:
- visual impairment;
- reduced work capacity;
- reduced manual dexterity;
- poor learning ability;
- difficulty in performing complex tasks.
The health threat from levels of CO sometimes found in the ambient air is most serious for those who suffer from cardiovascular disease such as angina pectoris.
 In the human body, Hemoglobin (an iron compound) in the blood carries the oxygen (O20) from the lungs to various tissues and transports back carbon dioxide (CO2) to the lungs. Hemoglobin has 240 times more affinity toward CO than it does for oxygen. Therefore, when the hemoglobin reacts with CO, it reduces the hemoglobin that is available for the transport of O2. This in turn reduces oxygen supply to the body's organs and tissues.
In the human body, Hemoglobin (an iron compound) in the blood carries the oxygen (O20) from the lungs to various tissues and transports back carbon dioxide (CO2) to the lungs. Hemoglobin has 240 times more affinity toward CO than it does for oxygen. Therefore, when the hemoglobin reacts with CO, it reduces the hemoglobin that is available for the transport of O2. This in turn reduces oxygen supply to the body's organs and tissues.
Sulfur Dioxide (SO2)
High concentrations of SO2 can result in the following health problems:
Short-term exposure
- Adults and children with asthma who are active outdoors will experience temporary breathing impairment.
- Individuals with asthma may experience breathing difficulties with moderate activity and may exhibit symptoms such as wheezing, chest tightness, or shortness of breath.
Long-term exposure (along with high levels of PM)
- Aggravation of existing cardiovascular disease
- Respiratory illness
- Alterations in the lungs’ defenses
The subgroups of the population that may be affected under these conditions include individuals with heart or lung disease, as well as the elderly and children.
The body's reaction to regular and acidic air
Instructions: Click on the types of air and observe what happens for each. (Note: The animation has no audio.)
As a person breathes, regular air enters the lungs through open passageways, filters through the lungs and exits the body through the same open passage as carbon dioxide. However, when the air is acidic, it causes the airways to restrict, and the body must now use twice the energy to evacuate the same amount of carbon dioxide.
Together, SO2 and NOx (discussed on the next page) are the major precursors to acidic deposition (acid rain), which is associated with the acidification of soils, lakes, and streams and accelerated corrosion of buildings and monuments. We will talk more about this in the next section. SO2 also is a major precursor to PM 2.5, which is a significant health concern, and a main contributor to poor visibility.
Health and Environmental Effects of Primary Pollutants, page 2
Nitrogen Oxides (NOx)
Nitric oxide (NO) and nitrogen dioxide (NO2) together are represented by NOx. Most of the emissions from combustion devices (approximately 90%) are in the form of NO.
NOx react in the air to form ground-level ozone and fine particulates, which are associated with adverse health effects.
- Short-term exposures (e.g., less than 3 hours) to low levels of NO2 may lead to changes in airway responsiveness and lung function in individuals with preexisting respiratory illnesses. These exposures may also increase respiratory illnesses in children.
- Long-term exposures to NO2 may lead to increased susceptibility to respiratory infection and may cause irreversible alterations in lung structure.
NOx contributes to a wide range of environmental effects directly and when combined with other precursors in acid rain and ozone.
- Increased nitrogen inputs to terrestrial and wetland systems can lead to changes in plant species composition and diversity.
- Direct nitrogen inputs to aquatic ecosystems such as those found in estuarine and coastal waters (e.g., Chesapeake Bay) can lead to eutrophication (a condition that promotes excessive algae growth, which can lead to a severe depletion of dissolved oxygen and increased levels of toxins harmful to aquatic life).
- Nitrogen, alone or in acid rain, also can acidify soils and surface waters.
Acid rain can ruin a fishing trip!
Acidification of soils causes the loss of essential plant nutrients and increased levels of soluble aluminum that are toxic to plants. Acidification of surface waters creates conditions of low pH and levels of aluminum that are toxic to fish and other aquatic organisms. NOx also contributes to visibility impairment.
Particulate Matter (PM)
Particles smaller than or equal to 10 µm (micro meter or millionth of a meter) in diameter can get into the lungs and can cause numerous health problems. Inhalation of these tiny particles has been linked with illness and death from heart and lung disease. Various health problems have been associated with long-term (e.g., multi-year) exposures to these particles. Shorter-term daily and potentially even shorter term peak (e.g., 1-hour) exposures to these particles can also be associated with health problems.
Particles can aggravate respiratory conditions, such as asthma and bronchitis, and have been associated with cardiac arrhythmias (heartbeat irregularities) and heart attacks. People with heart or lung disease, the elderly, and children are at highest risk from exposure to particles.
Particles of concern can include both fine and coarse-fraction particles, although fine particles have been more clearly linked to the most serious health effects.
- Particles larger than 2 micro meters (µm) do not penetrate beyond the nasal cavity or trachea.
- Particles smaller than 0.1 µm tend to deposit in tracheobronchia tree and are removed when exhaling.
- Particles between 0.1 and 2.0 µm penetrate deep into the lungs and settle in respiratory bronchioles or alveolar sacs
 In addition to health problems, PM is the major cause of reduced visibility in many parts of the United States by scattering and absorbing some of the light emitted or reflected by the body reducing the contrast. Airborne particles can also impact vegetation and ecosystems, and can cause damage to paints and building materials.
In addition to health problems, PM is the major cause of reduced visibility in many parts of the United States by scattering and absorbing some of the light emitted or reflected by the body reducing the contrast. Airborne particles can also impact vegetation and ecosystems, and can cause damage to paints and building materials.
How particulate matter is breathed into the human body
Instructions: See what happens when the name of each size of particulate matter is clicked on. (Note: The animation has no audio.)
How particulate matter is breathed into the human body is dependent on the size of the matter.
Particles smaller than .1 micrometers tend to deposit in the tracheobronchial tree and are removed when exhaling. Particles between .01 and 2 micrometers penetrate deep into the lungs and settle in respiratory bronchioles or alveolar sacs. These particles stay in the lungs. Particles larger than 2 micrometers do not penetrate beyond the nasal cavity or trachea. They exit the body during a cough or sneeze.
Lead
Exposure to lead occurs mainly through inhalation of air and ingestion of lead in food, water, soil, or dust. It accumulates in the blood, bones, and soft tissues and can adversely affect the kidneys, liver, nervous system, and other organs.
- Excessive exposure to lead may cause neurological impairments such as seizures, mental retardation, and behavioral disorders.
- Even at low doses, lead exposure is associated with damage to the nervous systems of fetuses and young children, resulting in learning deficits and lowered IQ.
- Recent studies indicated that lead may be a factor in high blood pressure and subsequent heart disease.
- Lead can also be deposited on the leaves of plants, presenting a hazard to grazing animals and to humans through ingestion.
Instructions: Click the "play" button to see the impact of using unleaded rather than leaded gasoline. (Note: The animation has no audio.)
In the mid-1970s, the use of leaded gasoline caused the amount of lead particles in the air to reach 200 million tons. The use of unleaded gasoline has reduced that amount to 4 million tons of lead particles in the air today.
Secondary Pollutants
The pollutants that are emitted directly from a combustion process are called “primary pollutants.” When emitted into the atmosphere, these primary pollutants combine with other reactants and form “secondary” pollutants.
An example of a secondary pollutant would be ozone. When hydrocarbons are emitted, and they react with NOx in presence of sunlight, they form ozone. Health and environmental effects of secondary pollutants are discussed in the next section: Global and Regional Effects of Pollutants.
