Lesson 4c: Acid Rain and the Ozone
Introduction to Acid Rain and the Ozone
Acid rain is a serious environmental problem around the world, particularly affecting Asia, Europe, and large parts of the U.S. and Canada. The acidic pollutants such as SO2 and NOx are emitted into the environment by combustion of fossil fuels.
Most of the sulfur in any fuel combines with oxygen and forms SO2 in the combustion chamber. This SO2 when emitted into the atmosphere slowly oxidizes to SO3. SO3 is readily soluble in water in the clouds and forms H2SO4 (sulfuric acid).
Most of the NOx that is emitted is in the form of NO. This NO is oxidized in the atmosphere to NO2. NO2 is soluble in water and forms HNO3 (nitric acid).
Acid Deposition
Sunlight increases the rate of most of the SO2 and NO reactions. The result is a mild solution of sulfuric acid and nitric acid. "Acid rain" is a broad term used to describe several ways that acids fall out of the atmosphere. A more precise term is acid deposition, which has two parts: wet and dry.
- Wet deposition - refers to acidic rain, fog, and snow. As this acidic water flows over and through the ground, it affects a variety of plants and animals. The strength of the effects depend on many factors, including:
- the acidity of the water;
- the chemistry and buffering capacity of the soils involved;
- the types of fish, trees, and other living things that rely on the water.
- Dry deposition - refers to acidic gases and particles. About half of the acidity in the atmosphere falls back to earth through dry deposition.
- Acidic particles and gases are blown by the wind onto buildings, cars, homes, and trees.
- Dry deposited gases and particles can also be washed from trees and other surfaces by rainstorms. When that happens, the runoff water adds those acids to the acid rain, making the combination more acidic than the falling rain alone.
Process of Acid Deposition
Prevailing winds blow the compounds that cause both wet and dry acid deposition across state and national borders, and sometimes over hundreds of miles. Please watch the 1:22 presentation below to learn more about the process of acid deposition.
In this diagram, we are seeing how the acid deposition occurs. When the sources emit pollutants such as SO2, NOx, mercury, and volatile organic compounds, primarily SO2 and NOx, which are acidic gases, are deposited in two ways. One is dry deposition, the other one is wet deposition. The SO2 and NOx when they deposit back either gaseous pollutants or as particulates, it's called dry deposition. When these pollutants dissolve in water, cloud water, and then deposit, it's called wet precipitation. Or that is what we call acid rain. The dry, gaseous pollutants or particulate matter can sometimes get dissolved in water and come down again as wet precipitation. Receptors are the species that receive this acid rain and get affected. These receptors can be materials that we care about, or aquatic life, human beings, or lakes and streams.
pH Scale
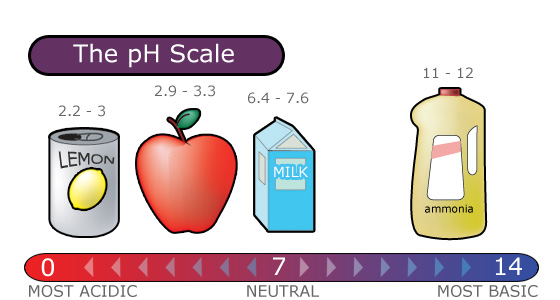
Acid rain is measured using a pH scale.
pH is a measure of hydrogen ion concentration, which is measured as a negative logarithm. In other words, acids produce hydrogen ions and alkalis produce hydroxyl ions, so pH is the power of a solution to yield hydrogen ions [H+].
The pH scale ranges from 0 to 14 and indicates how acidic or basic a substance is.
- A pH of 7 is neutral.
- A pH less than 7 is acidic.
- A pH greater than 7 is basic.
The lower a substance's pH, the more acidic it is. Each whole pH value below 7 (the neutral point) is ten times more acidic than the next higher value.
- For example, a pH of 4 is ten times more acidic than a pH of 5 and 100 times (10 times 10) more acidic than a pH of 6.
The higher a substance’s pH, the more basic or alkaline it is.
- Each whole pH value above 7 is ten times more alkaline (another way to say basic) than the next lower whole value.
- For example, a pH of 10 is ten times more alkaline than a pH of 9.
Effects of Acid Rain
Overview
Pure water has a pH of 7.0. Normal rain is slightly acidic because carbon dioxide dissolves into it, so it has a pH of about 5.5. As of the year 2000, the most acidic rain falling in the US has a pH of about 4.3.
Below is a video demonstration that replicates the effect of acid rain on plant life. In this video, beans are placed in: a) water, b) slightly acidic water and c) acidic water, and their growth is observed over a period of three days. Please watch the following 5:35 video:
We are learning about the environmental effects of acid rain. Acid rain is basically the compounds that form acid are mixing with the water in the clouds and coming down acidic rain. As it can be created at home using various day-to-day ingredients, one of them is vinegar, or you could use, also, lemon juice, that has a lot of acid. So what we are going to do in this demonstration today is to see the effect of acid rain on spouting off moon beans. These are a special kind of beans. And you can use, actually, most of the types of beans which have hard shell on the outside. So we are using here three different bowls here. Each of these bowls is filled with 20 beans, 20 seeds of these moon beans. And we are going to pour plain water in this one, and slightly acidic water in this bowl, and highly acidic water in this bowl, and see every day-- tomorrow, day after, and a day after that-- how this acid changes the spouting off these beans.
So let's actually prepare this and add plain water to the first one. This is plain water, just water from the faucet. I'm going to add enough water so that these beans are all immersed in the water. Now the slightly acidic water I'm preparing by adding vinegar that I have here. And now this is a bottle of vinegar. And I'm going to add two spoons of vinegar to this. So this water in this cup is slightly acidic. I'm going to mix this and use this water. This is slightly acidic water. And I'm going to pour slightly acidic water in here. There are 20 beans in this cup, in this bowl, also. And I'm going to prepare highly acidic water here with the same vinegar. I'm going to add five spoons-- one, two, three, four, five. This is a little bit stronger than the other acid that we prepared. Now I'm going to add this highly acidic water to this bowl to the same level. And as the water evaporates, whatever type of water-- as the water evaporates, we need to refill with the same kind of water-- plain water in this bowl, slightly acidic water in this bowl. And highly acidic water in this bowl-- and observe the results.
And what I want you to now hypothesize is which one would have the significant effects. Which one would have the significant effect? Or which one of these bowls will have more sprouting than the other? And when you look at day one, day two, day three, I need you to count the number of seeds that are sprouting. And you can prepare a plot. Let's say on day one, the plain water ones sprouted six out of 20, which means 30% have sprouted. Slightly acidic ones, four out of 20. So that's 25%. Highly acidic one, six out of 20. That's, again, 30%. So you can plot that as a function of time and see at the end of four or five days how many of these seeds sprout. Let's watch for three days. OK. We are on day three today. And you can see on day one itself we had all 20 out of 20 sprouted in this plain water. And slightly acidic water, we didn't have any sprouts. Even today, we don't have any sign of sprouting in highly acidic water. That is the impact that you can see of the acidity on sprouting of seeds.
Now you can imagine what it would be like for the entire planet if it is covered with acidic rain. And what would be the impact on agriculture? What would be the impact on food supply chain to the humanity? So that is what we learned from this exercise.
Negative Consequences
Acid rain results in many negative consequences. Place your mouse over the image below to see the effects of acid deposition.
Effects of Acid Rain on Forests and Aquatic Life
Effects of Acid Rain on Forests
Acid rain does not usually kill trees directly. Instead, it is more likely to weaken trees by:
- damaging their leaves
- limiting the nutrients available to them
- exposing them to toxic substances slowly released from the soil
Quite often, injury or death of trees is a result of these effects of acid rain in combination with one or more additional threats. Click on the hot spots in the image below to see the effects of acid rain on the forest:
Effects of Acid Rain on Aquatic Life
Acid rain causes acidification of lakes and streams and contributes to damage of trees at high elevations (for example, red spruce trees above 2,000 feet) and many sensitive forest soils. Several regions in the U.S. were identified as containing many of the surface waters sensitive to acidification. They include the:
- Adirondacks and Catskill Mountains in New York State;
- Mid-Appalachian Highlands along the east coast;
- Upper Midwest;
- Mountainous areas of the Western United States.
Some types of plants and animals can handle acidic waters. Others, however, are acid-sensitive and will be lost as the pH declines. View the image of the fish, shellfish, and insects below to see what pH levels they can tolerate:
Bass - Acid tolerance of up to pH 5.5
Perch - Acid tolerance of up to pH 4.5
Frogs - Acid tolerance of up to pH 4.0
Salamander - Acid tolerance of up to pH 5.0
Clams - Acid tolerance of up to pH 6.0
Crayfish - Acid tolerance of up to pH 5.5
Snails - Acid tolerance of up to pH 6.0
Mayfly - Acid tolerance of up to pH 5.5
Effects of Acid Rain on Materials, Visibility and Human Health
Effects of Acid Rain on Materials
Acid rain and the dry deposition of acidic particles contribute to the corrosion of metals (such as bronze) and the deterioration of paint and stone (such as marble and limestone). These effects seriously reduce the value to society of buildings, bridges, cultural objects (such as statues, monuments, and tombstones), and cars.

Effects of Acid Rain on Visibility
Sulfates and nitrates that form in the atmosphere from sulfur dioxide (SO2) and nitrogen oxides (NOx) emissions contribute to visibility impairment, meaning we can't see as far or as clearly through the air.
Eastern United States
Sulfate particles account for 50 to 70 percent of the visibility reduction in the eastern part of the United States, affecting our enjoyment of national parks, such as the Shenandoah and the Great Smoky Mountains.
Through the Acid Rain Program, SO2 reductions will be completed to improve visual range at national parks located in the eastern United States. Based on a study of the value national park visitors place on visibility, these reductions are expected to be worth over a billion dollars annually by the year 2010.

Western United States
In the western part of the United States, nitrates and carbon also play roles, but sulfates have been implicated as an important source of visibility impairment in many of the Colorado River Plateau national parks, including the Grand Canyon, Canyonlands, and Bryce Canyon.

Effects of Acid Rain on Human Health
Acid rain looks, feels, and tastes just like clean rain. The harm to people from acid rain is not direct. Walking in acid rain, or even swimming in an acid lake, is no more dangerous than walking or swimming in clean water. However, the pollutants that cause acid rain also damage human health.
- Effects of Sulfur Dioxide (SO2): These gases interact in the atmosphere to form fine sulfate and nitrate particles that can be transported long distances by winds and inhaled deep into people's lungs. Fine particles can also penetrate indoors. Many scientific studies have identified a relationship between elevated levels of fine particles and increased illness and premature death from heart and lung disorders, such as asthma and bronchitis.
- Effects of Nitrogen Oxide (NOx): Decrease in nitrogen oxide emissions are also expected to have a beneficial impact on human health by reducing the nitrogen oxides available to react with volatile organic compounds and form ozone. Ozone impacts on human health include a number of morbidity and mortality risks associated with lung inflammation, including asthma and emphysema.
Protecting the Environment
You can do the following to protect the environment:
- Turn off lights, computers, and other appliances when you're not using them.
- Use energy efficient appliances: lighting, air conditioners, heaters, refrigerators, washing machines, etc.
- Only use electric appliances when you need them.
- Keep your thermostat at 68°F in the winter and 72°F in the summer. You can turn it even lower in the winter and higher in the summer when you are away from home.
- Insulate your home as best you can.
- Carpool, use public transportation, or better yet, walk or bicycle whenever possible.
- Buy vehicles with low NOx emissions, and maintain all vehicles well.
Introduction to Ozone
Ozone (O3) is a triatomic oxygen molecule gas that occurs both in the Earth’s upper atmosphere and at ground level. Ozone can be good or bad, depending on where it is found: It is a bluish gas that is harmful to breathe. Therefore, it is bad at the ground level.
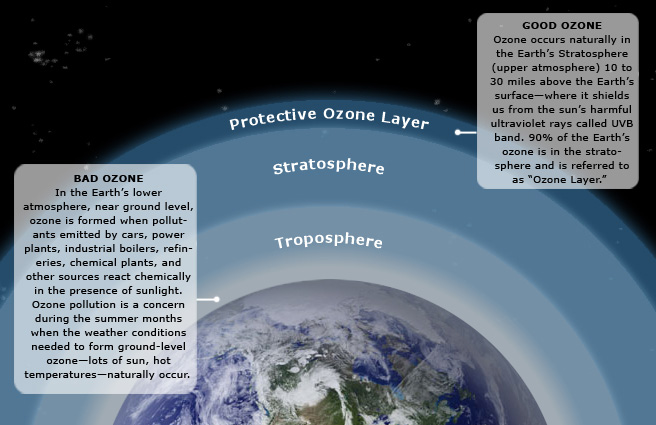
Good ozone occurs naturally in the Earth's Stratosphere (upper atmosphere) 10 to 30 miles above the Earth's surface-where it shields us from the sun's harmful ultraviolet rays called UVB band. 90% of the Earth's ozone is in the stratosphere and is referred to as Ozone Layer.
Bad ozone occurs in the Earth's lower atmosphere, near ground level, when pollutants emitted by cars, power plants, industrial boilers, chemical plants, and other sources react chemically in the presence of sunlight. Ozone pollution is a concern during the summer months, when the weather conditions needed to form ground level ozone (lots of sun and hot temperatures) naturally occur.
The Ozone Cycle
The presentation below shows the process of ozone depletion. Ozone depletion is caused by chlorofluorocarbons (CFCs) and other ozone-depleting substances. Please watch the following 1:16 video.
In this picture we are seeing how the ozone layer is destroyed and the effects of that destruction. Certain compounds such as chlorofluorocarbons, CFC’s, are released into the atmosphere by human activities and these CFC’s particularly, they are very, very unreactive at the atmospherical ground level and they go all the way up to the stratosphere and then the CFC’s dissociate or give up the chlorine. Each chlorine atom is capable of destroying or basically turning ozone into oxygen and which again doesn’t have the same capability as ozone does in shielding us from the UV radiation. And once that ozone layer or the number of ozone molecules in that layer goes down, more and more UV rays can pass through the atmosphere and reach the surface and can cause more skin cancer and cataracts in older people.
Production and Destruction of Ozone
Ozone is constantly produced and destroyed in a natural cycle, as shown in the figure below. However, the overall amount of ozone is essentially stable. This balance can be thought of as a stream's depth at a particular location. Although individual water molecules are moving past the observer, the total depth remains constant. Similarly, while ozone production and destruction are balanced, ozone levels remain stable. This was the situation until the past several decades. Please watch the following 1:32 video about ozone destruction.
Here we are looking at the ozone science. How the ozone is produced and how the ozone is destroyed. In the first step, the oxygen molecules are photolyzed or converted by the UV rays that are coming from the sun into two oxygen atoms; nascent oxygen atoms. Oxygen atoms are very, very reactive, and they react with another oxygen molecule and form ozone, O3. Ozone and oxygen atoms are continually being interconverted as rays break the ozone and turns into nascent oxygen and oxygen molecules. And the oxygen atom again reacts with the oxygen molecules, forms ozone. Our activities, which are producing the CFC’s and liberating into the atmosphere, they are going, and these chlorine atoms are destroying the ozone molecules in addition to the natural process of formation and destruction. That is what is causing the reduction in the concentration of ozone in the stratosphere and when the concentration goes down below certain levels, like 220 Dobson units, we call that ozone hole. Ozone hole does not mean that there is a big hole up there, but what it means is that the concentration is below a certain level.
Large increases in stratospheric chlorine and bromine, however, have upset the balance of the Ozone. In effect, they have added a siphon downstream, removing ozone faster than natural ozone creation reactions can keep up. Therefore, ozone levels fall.
Since ozone filters out harmful UVB radiation, less ozone means higher UVB levels at the surface. The more the ozone is depleted, the larger will be the increase in incoming UVB radiation. UVB has been linked to:
- skin cancer;
- cataracts;
- damage to materials like plastics;
- harm to certain crops and marine organisms.
Although some UVB reaches the surface even without ozone depletion, its harmful effects will increase as a result of this problem.

Ozone-Depleting Substance(s) (ODS) are:
- CFCs;
- HCFCs (used in the energy related to refrigeration and air conditioning in homes, commercial buildings, and cars, and manufacture of foam products);
- Halons (used in fire extinguishers);
- Methyl bromide, carbon tetrachloride;
- Methyl chloroform (used as solvents in chemical industries).
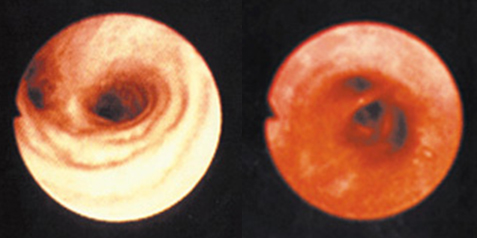
The Ozone Hole
Recent studies by NASA and others have indicated that about 40 percent of the ozone in the Antarctica has been destroyed and that about 7 percent of ozone is destroyed from the Arctic Circle. The destruction of ozone is also called “Ozone Hole."
Ozone hole does not mean that there is no ozone in the region. The ozone hole is defined as the area having less than 220 dobson units (DU) of ozone (concentration) in the overhead column (i.e., between the ground and space).
The image below shows the reduction in ozone concentration over Antarctica. This hole in the Antarctica is unfortunately allowing more Australians to be exposed to UV radiation. However, if this kind of ozone destruction ever takes place in the Arctic zone, more humans (in the Northern Hemisphere) would be exposed to higher levels of UVB radiation.

A Dobson Unit is the measure of the amount or thickness of ozone in the atmosphere. It is based on a measurement taken directly above a specific point on the Earth's surface. One Dobson unit refers to a layer of ozone that would be 0.001 cm thick under conditions of standard temperature (0 degree C) and pressure (the average pressure at the surface of the Earth). The Dobson unit was named after G.M.B. Dobson, who was a researcher at Oxford University in the 1920s. He built the first instrument (now called the Dobson meter) to measure total ozone from the ground.
The size of the Southern Hemisphere ozone hole as a function of the year is shown in the figure below. The graph compares the size of the hole over a twenty-year period, from 1980 to 2010. It can be seen that the size increased each year. Each year, in the spring, the ozone hole is at its largest.
Effects of Ozone Depletion on skin
Effects of ozone depletion can result in 1) increased cases of skin cancer, 2) skin damage, 3) cataracts and other eye damage, and 4) immune suppression.
Skin Cancer
The incidence of skin cancer in the United States has reached epidemic proportions. One in five Americans will develop skin cancer in their lifetime, and one American dies every hour from this devastating disease.
Medical research is helping us understand the causes and effects of skin cancer. Many health and education groups are working to reduce the incidence of this disease, of which 1.3 million cases have been predicted for 2000 alone, according to The American Cancer Society. The figure below shows the sources of ozone depleting substances.
Melanoma
Melanoma, the most serious form of skin cancer, is also one of the fastest growing types of cancer in the United States. Many dermatologists believe there may be a link between childhood sunburns and melanoma later in life. Melanoma cases in this country have more than doubled in the past 2 decades, and the rise is expected to continue.
Nonmelanoma Skin Cancers
Nonmelanoma skin cancers are less deadly than melanomas. Nevertheless, left untreated, they can spread, causing disfigurement and more serious health problems. More than 1.2 million Americans will develop nonmelanoma skin cancer in 2000 while more than 1,900 will die from the disease. There are two primary types of nonmelanoma skin cancers.
- Basal Cell Carcinomas are the most common type of skin cancer tumors. They usually appear as small, fleshy bumps or nodules on the head and neck, but can occur on other skin areas. Basal cell carcinoma grows slowly, and rarely spreads to other parts of the body. It can, however, penetrate to the bone and cause considerable damage.
- Squamous Cell Carcinomas are tumors that may appear as nodules or as red, scaly patches. This cancer can develop into large masses, and unlike basal cell carcinoma, it can spread to other parts of the body.
These two cancers have a cure rate as high as 95 percent if detected and treated early. The key is to watch for signs and seek medical treatment.
Other Skin Damage
Other UV-related skin disorders include actinic keratoses and premature aging of the skin.
- Actinic keratoses are skin growths that occur on body areas exposed to the sun. The face, hands, forearms, and the "V" of the neck are especially susceptible to this type of lesion. Although premalignant, actinic keratoses are a risk factor for squamous cell carcinoma. Look for raised, reddish, rough-textured growths and seek prompt medical attention if you discover them.
- Chronic exposure to the sun also causes premature aging, which over time can make the skin become thick, wrinkled, and leathery. Since it occurs gradually, often manifesting itself many years after the majority of a person's sun exposure, premature aging is often regarded as an unavoidable, normal part of growing older. With proper protection from UV radiation, however, most premature aging of the skin can be avoided.

Protect yourself against sunburn. Minimize sun exposure during midday hours (10 am to 4 pm). Wear sunglasses, a hat with a wide brim, and protective clothing with a tight weave. Use a broad spectrum sunscreen with a sun protection factor (SPF) of at least 15. To be safer, 30 is better.
Effects of Ozone Depletion on eyes and immune system
Cataracts and Other Eye Damage
Cataracts are a form of eye damage in which a loss of transparency in the lens of the eye clouds vision. If left untreated, cataracts can lead to blindness. Research has shown that UV radiation increases the likelihood of certain cataracts. Although curable with modern eye surgery, cataracts diminish the eyesight of millions of Americans and cost billions of dollars in medical care each year.
Instructions: Place your mouse over the image below to see the effect cataracts can have on vision. (Note: This video has no audio.)
Other kinds of eye damage include pterygium (i.e., tissue growth that can block vision), skin cancer around the eyes, and degeneration of the macula (i.e., the part of the retina where visual perception is most acute). All of these problems can be lessened with proper eye protection from UV radiation.
Immune Suppression
Scientists have found that overexposure to UV radiation may suppress proper functioning of the body's immune system and the skin's natural defenses. All people, regardless of skin color, might be vulnerable to effects including impaired response to immunizations, increased sensitivity to sunlight, and reactions to certain medications.
Protecting the Environment- Ozone Depletion
Your “Power” in Protecting the Environment from Ozone Depletion
- Make sure that technicians working on your car air conditioner, home air conditioner, or refrigerator are certified by an EPA-approved program to recover the refrigerant (this is required by law).
- Have your car and home air conditioner units and refrigerator checked for leaks. When possible, repair leaky air conditioning units before refilling them.
- Contact local authorities to properly dispose of refrigeration or air conditioning equipment.
International Action in Protecting the Environment from Ozone Depletion
In 1987, the Montreal Protocol, an international environmental agreement, established requirements that began the worldwide phase out of ozone-depleting CFCs (chlorofluorocarbons). These requirements were later modified, leading to the phase out in 1996 of CFC production in all developed nations.
Ground Level Ozone and Photochemical Smog
Ozone is a secondary pollutant that forms from the primary pollutants such as Volatile Organic Compounds (Hydrocarbons) and nitrogen oxides (NOx) in the presence of sunlight. Its formation is mainly from the automobile emissions.

Below is a demonstration on how ozone forms at the ground level (note ground level ozone is also known as “bad” ozone). Please watch the following 5:29 video:
This demonstration is basically looking at how ozone forms or how photochemical smog forms at the ground level because of the pollutants that we emit. Ozone emission or ozone formation depends on two ingredients, nitrogen oxides that are emitted by pollutants that are going out to the tail pipe of a car and in a power plant with a stack. Nitrogen oxides react with volatile organic compounds. Volatile organic compounds are also emitted by the tailpipe of the cars and some of them are coming from petroleum refineries and petroleum industries. VOCs + NOx = Ozone.
These volatile organic compounds react to form ozone only in the presence of sunlight. That is the critical thing, only in presence of sunlight. So, to see this demonstration, we need sunlight. Obviously, we don’t have the sunlight here, but I can create sunlight or the wave lengths that are important to create the same effects. That is the reason I am using a UV lamp. You see this lamp here? This blue light is producing the UV light that is required to create ozone. And this ozone again reacts with volatile organic compounds to produce photochemical smog.
So first of all I am trying to create here ozone. The way I am creating ozone is not with Nitrogen oxides and volatile organic compounds, but turning the oxygen that is there in the beaker into ozone by using this UV light. So let me close this and in about three or four minutes, the oxygen that is there inside will turn into ozone. So we have one ingredient. The second ingredient that is required is Nitrogen oxides or volatile organic compounds to produce smog. Once ozone is formed, I can introduce one of those ingredients, and we can see the smog. Ok, I guess we have probably enough ozone in there. Now we need to add Nitrogen oxides and volatile organic compounds to form the photochemical smog that we are seeing or that we should be seeing. Ozone + VOCs + NOx = Photochemical Smog.
Since I don’t have my car with me or the tail pipe, I need to somehow produce hydro-carbons in this room. So what I am doing is hydro carbons and Nitrogen oxide, cause even is nitrogen oxides are not there, we can form photochemical smog if we have hydro carbons. The composition will be slightly different. So now I am taking my orange, this is the orange, and you know the beautiful smell that you normally smell with an orange is because of linolenic acid. Linolenic acid is the orange smell that you get. And I’m just taking a small peel of this orange, this is the source of hydrocarbons, and I am trying to put it in here - nothing big deal. And this ozone that is there inside will slowly react with this hydrocarbon that are produced, that are emitted from this orange peel and produce the photochemical smog.
Do you see anything going on here? Are you able to still see through the beaker? Is the atmosphere clear in the beaker, or is it becoming hazy? Let me turn this little bit around, and you may be able to see now, I don’t know. Can you see anything coming out of this? That is the photochemical smog, actually. Let me open this and show you. That is the smog that you see and when the smog or when this smoke kind of smog is in the beaker or in the atmosphere, you can not see through and since this has ozone in it, it is not good for our health. When you live in cities filled with this kind of smog, you generally experience breathing problems and other health problems. Your eyes start to water when you drive home from work late in the afternoon, or your nose starts to run because your body tries to get rid of, get rid of these pollutants. So that is photochemical smog and if we keep this orange peel like this we probably can generate enough smog for about 24 hours to 36 hours like this, with this little orange peel. Now you can see what we are doing to the atmosphere by emitting roughly 10, 12, 13 million tons of nitrogen oxides into the atmosphere and several million tons of volatile organic compounds into the atmosphere.
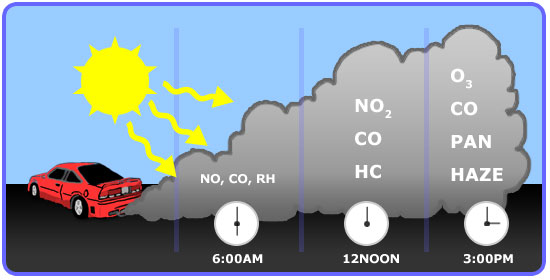
As previously mentioned, the formation of ozone is mainly from automobile emission. A typical profile of pollutants in the air of major cities is well repeatable and is shown in the figure below. Note how the formation changes over the course of a day:
- Early in the morning - During peak traffic hours, NO and Hydrocarbons are emitted along with CO.
- Mid-morning - NO is slowly oxidized to NO2.
- Mid-afternoon - In the presence of sunlight, NOx react with VOCs to form ozone.
Ozone, by itself, is damaging to health and also to the environment. Ozone triggers a variety of health problems even at very low levels and may cause permanent lung damage after long-term exposure. Ozone also leads to the formation of smog or haze, causing additional problems such as a decrease in visibility as well as damage to plants and ecosystems.
Basic Chemistry and Sources
As we have learned, volatile Organic Compounds (Hydrocarbons) combine with nitrogen oxides (NOx) in the presence of sunlight to form ozone.
In turn, sunlight and hot weather cause ground-level ozone to form in harmful concentrations in the air. As a result, it is known as a summertime air pollutant.
Many urban areas tend to have high levels of "bad" ozone, but even rural areas are also subject to increased ozone levels because wind carries ozone and pollutants that form it hundreds of miles away from their original sources.
View the graph below to compare the major sources of NOx and VOC that help to form ozone.
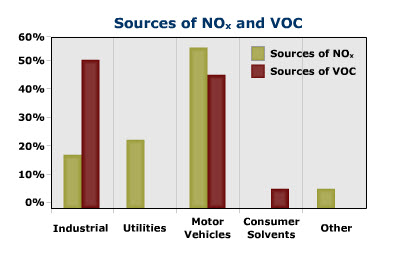
| Source | Amount of NOx | Amount of VOC |
|---|---|---|
| Industrial | 18% | 50% |
| Utilities | 24% | 0% |
| Motor Vehicles | 55% | 45% |
| Consumer Solvents | 0% | 5% |
| Other | 5% | 0% |
Health and Environmental Impact
Several groups of people are particularly sensitive to ozone—especially when they are active outdoors—because physical activity causes people to breathe faster and more deeply. In general, as concentrations of ground-level ozone increase, more and more people experience health effects, the effects become more serious, and more people are admitted to the hospital for respiratory problems. When ozone levels are very high, everyone should be concerned about ozone exposure.
Below is a listing of Health Effects of Ground Level Ozone
- Ozone can irritate your respiratory system, causing you to start coughing, feel an irritation in your throat and/or experience an uncomfortable sensation in your chest.
- Ozone can reduce lung function and make it more difficult for you to breathe as deeply and vigorously as you normally would. When this happens, you may notice that breathing starts to feel uncomfortable. If you are exercising or working outdoors, you may notice that you are taking more rapid and shallow breaths than normal.
- Ozone can aggravate asthma. When ozone levels are high, more people with asthma have attacks that require a doctor's attention or the use of additional medication. One reason this happens is that ozone makes people more sensitive to allergens, which are the most common triggers for asthma attacks. Also, asthmatics are more severely affected by the reduced lung function and irritation that ozone causes in the respiratory system.
- Ozone can inflame and damage cells that line your lungs. Within a few days, the damaged cells are replaced, and the old cells are shed—much in the way your skin peels after a sunburn.
- Ozone may aggravate chronic lung diseases such as emphysema and bronchitis and reduce the immune system's ability to fight off bacterial infections in the respiratory system.
- Ozone may cause permanent lung damage. Repeated short-term ozone damage to children's developing lungs may lead to reduced lung function in adulthood. In adults, ozone exposure may accelerate the natural decline in lung function that occurs as part of the normal aging process.
Protecting the Environment
Click on the hotspots in the image below to find out what you can do to protect the environment.