11.1 Anaerobic Digestion
Anaerobic digestion (AD) is a biological process that breaks down organic materials (feedstocks) in the absence of oxygen (anaerobic conditions) into methane (CH4) and carbon dioxide (CO2). It is a process that occurs naturally in bogs, lake sediments, oceans, and digestive tracts. Cows contain one of the most well-known fermentation vats, the rumen, which is part of the stomach (in other animals as well). Fermentation takes place during digestion! The figure below shows a schematic of anaerobic digestion.
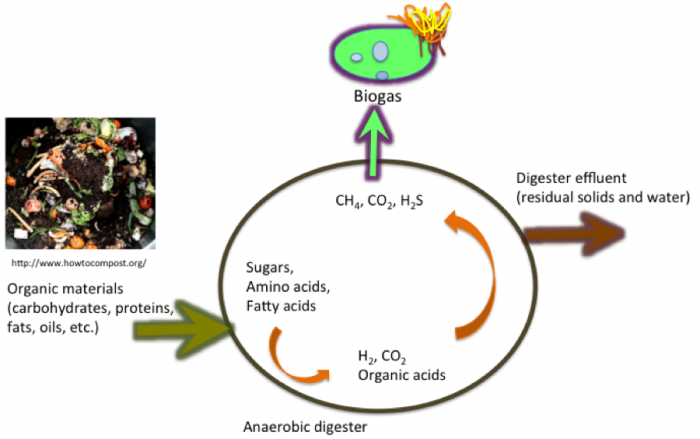
This is the anaerobic digester process. Organic materials (carbohydrates, proteins, fats, oils, etc.) enter the cycle. The sugars, amino acids, and fatty acids become H2, CO2, and organic acids. From there the digester effluents (residual solids and water) are removed. The digestor yields CH4, CO2, and H2S which are removed as biogas.
There are benefits to using an anaerobic digester, particularly when raising livestock. Biogas, which contains methane and hydrogen, will be produced that can be used as a fuel. From a waste treatment point of view, it reduces the volume and mass of the waste, as well as reduces organic content and biodegradability of waste so that the residual matter can be better used as soil amendment and fertilizer. There are also environmental benefits: 1) odors and emissions of greenhouse gases (i.e., methane) and volatile organic compounds are reduced, and 2) the digester will destroy pathogens in the waste.
So, what are the biological processes that occur during AD? The bacteria ferment and convert complex organic materials into acetate and hydrogen. There are four basic phases of anaerobic digestion, which is a synergistic process using anaerobic microorganisms: 1) hydrolysis, 2) acidogenesis, 3) acetogenesis, and 4) methanogenesis. The figure below shows the progression and types of products for each phase.
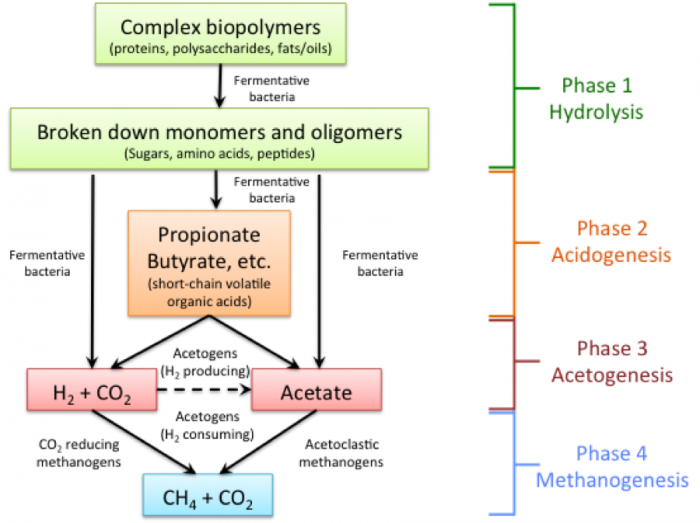
Four Phases of Biogas Production
Phase 1 – Hydrolysis
Complex Biopolymers (proteins, polysaccharides, fats/oils) in the presence of fermentative bacteria become broken-down monomers and oligomers (sugars, amino acids, peptides)
Phase 2 – Acidogenesis
The broken-down monomers and oligomers in the presence of fermentative bacteria become propionate, butyrate, etc (short-chain volatile organic acids)
Phase 3 – Acetogenesis
The broken-down monomers and oligomers from phase 1, in the presence of fermentative bacteria, can also become acetate or H2 + CO2. The propionate and butyrate in the presence of Acetogens (H2 producing or consuming) can also become acetate or H2 + CO2.
Phase 4 – Methanogenesis
H2 + CO2 in the presence of CO2 -reducing methanogens becomes CH4 + CO2. Acetate in the presence of acetoclastic methanogens can also become CH4 + CO2
Hydrolysis Biochemistry
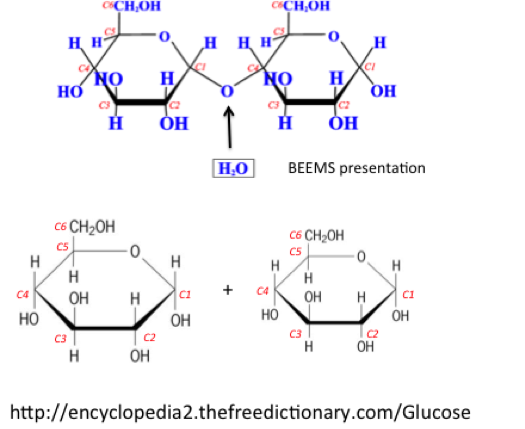
We have talked about hydrolysis in earlier lessons. Hydrolysis is a reaction with water. Acid and base can be used to accelerate the reaction. However, this occurs in enzymes as well. The figure below shows the hydrolysis reaction, and how cellulose, starch, and simple sugars can be broken down by water and enzymes. In anaerobic digestion, the enzymes are exoenzymes (cellulosome, protease, etc.) from a number of bacteria, protozoa, and fungi (see Reaction 1).
(1) biomass + H2O → monomers + H2
(Sources: cellulose, starch, sugars, fats, oils) (Products: mono-sugars [glucose, xylose, etc.], fatty acids)
Acidogenesis Biochemistry
During acidogenesis, soluble monomers are converted into small organic compounds, such as short-chain (volatile) acids (propionic, formic, lactic, butyric, succinic acids – see Reaction 2), ketones (glycerol, acetone), and alcohols (ethanol, methanol – see Reaction 3).
(2) C6H12O6 + 2H2 → 2CH3CH2COOH + 2H2O
(3) C6H12O6 → 2CH3CH2OH + 2CO2
Acetogenesis Biochemistry
The acidogenesis intermediates are attacked by acetogenic bacteria; the products from acetogenesis include acetic acid, CO2, and H2. Reactions 4-7 show the reactions that occur during acetogenesis:
(4) CH3CH2COO- + 3H2O → CH3COO- + H+ + HCO3- + 3H2
(5) C6H12O6 + 2H2O → 2CH3COOH + 2CO2 + 4H2
(6) CH3CH2OH + 2H2O → CH3COO- + 2H2 + H+
(7) 2HCO3- + 4H2 + H+ → CH3COO- + 4H2O
Several bacteria contribute to acetogenesis, including:
Syntrophobacter wolinii, propionate decomposer
Syntrophomonos wolfei, butyrate decomposer
Clostridium spp., peptococcus anaerobes, lactobacillus, and actinomyces are acid formers.
Methanogenesis Biochemistry
The last phase of anaerobic digestion is the methanogenesis phase. Several reactions take place using the intermediate products from the other phases, with the main product being methane. Reactions 8-13 show the common reactions that take place during methanogenesis:
(8) 2CH3CH2OH + CO2 → 2CH3COOH + CH4
(9) CH3COOH → CH4 + CO2
(10) CH3OH → CH4 + H2O
(11) CO2 + 4H2 → CH4 + 2H2O
(12) CH3COO- + SO42- + H+ → 2HCO3 + H2S
(13) CH3COO- + NO- + H2O + H+ → 2HCO3 + NH4+
Several bacterial contribute to methanogenesis, including:
Methanobacterium, methanobacillus, methanococcus, and methanosarcina, etc.
As you can see, the bacteria for anaerobic digestion are different from other enzymes for making biofuels, and could even be in our own stomachs!
Any kind of organic matter can be fed to an anaerobic digester, including manure and litter, food wastes, green wastes, plant biomass, and wastewater sludge. The materials that compose these feedstocks include polysaccharides, proteins, and fats/oils. Some of the organic materials degrade at a slow rate; hydrolysis of cellulose and hemicellulose is rate limiting. There are some organic materials that do not biodegrade: lignin, peptidoglycan, and membrane-associated proteins. The organic residues contain water and biomass composed of volatile solids and fixed solids (minerals or ash after combustion). And it’s the volatile solids (VS) that can be non-biodegradable and biodegradable.
As we discussed regarding the pretreatment of biomass for making ethanol, the efficiency of anaerobic digestion improves with pretreatment. Hydrolysis of cellulose and hemicellulose (phase 1 in AD) is improved with pretreatment because it overcomes biomass recalcitrance. As discussed in a previous lesson, pretreatment options include treatments with acids, alkalines, steam explosion, size reduction, etc. Common alkaline agents include: NaOH, Ca(OH)2, and NH3.
Theoretical methane yield (YCH4, m3 STP/kg substrate converted) can be calculated from the elemental composition of a substrate:
CcHhOxNnSs
The table below shows the substrate, a common elemental formula, and the theoretical methane yield for each.
| Substrate | Elemental formula |
Theoretical methane yield (m3 STP/kg) |
|---|---|---|
| Carbohydrates | (CH2O)n | 0.37 |
| Proteins | C106H168O34N28S | 0.51 |
| Fat | C8H15O | 1.0 |
| Plant biomass | C5H9O2.5NS0.025 | 0.48 |
The first figure below shows the biogas yield for several different feedstocks in m3/ton. Be aware that after digestion, there is a biogas yield, and the remainder of the digestion, is known as digestate. The biogas typically contains 50-60% CH4, with the rest primarily composed of CO2 and other trace gases. The digestate contains fiber, nutrients, and water, and these can be used for compost, animal bedding, and composite boards. The second figure below shows a schematic of the components of the digester.
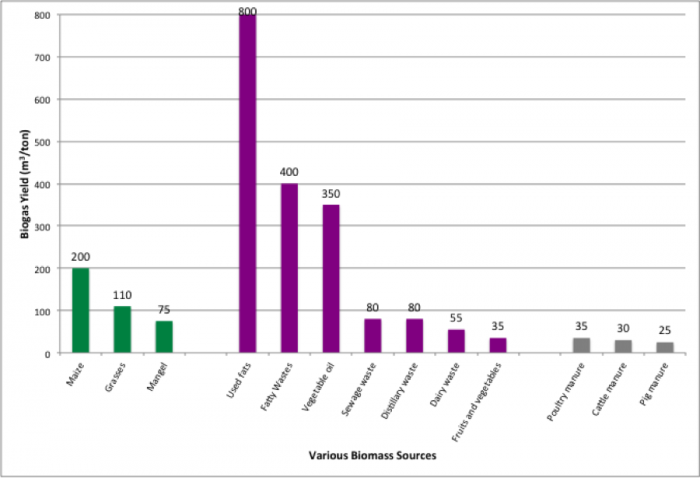
Biogas Yields (m3/ton) of Different Biomass Feedstocks:
- Maize 200
- Grasses 110
- Mangel 75
- Used Fats 800
- Fatty Wastes 400
- Vegetable Oil 350
- Sewage Waste 80
- Distillery Waste 80
- Dairy Waste 55
- Fruits & Vegetables 35
- Poultry Manure 35
- Cattle Manure 30
- Pig Manure 25
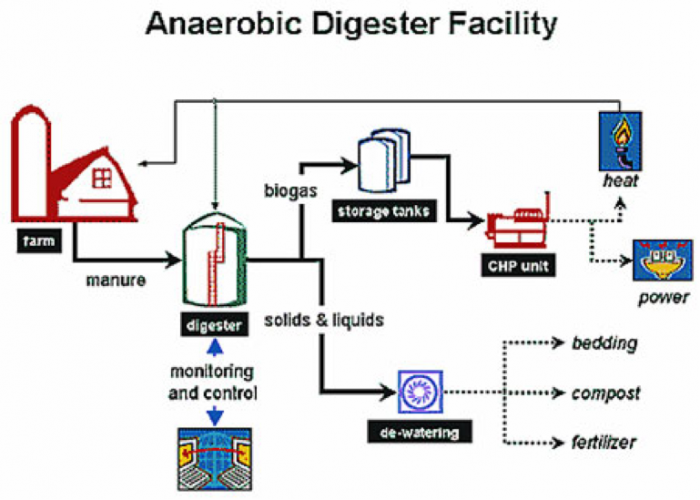
Farm leads to manure which goes to a digester. The products of the digester are separated into solids & liquids, and biogas. The biogas is stored in tanks and then sent to a CHP unit. From there the energy is used to produce power, or heat to warm the farm or the digester. The solids and liquids go through dewatering and are used as bedding, compost, or fertilizer
There are several factors that will affect anaerobic digestion. Different feedstocks will degrade at different rates and produce different amounts of methane (as seen in the Biogas yields graph and table above). That depends on the biological degradability and methane potential, the carbon and nutrients available, and the moisture content of each feed material. As noted in the Biogas yields graph and table above, fats contain the highest volatile solids and can generate the greatest amount of biogas. Solids take a longer time to digest than feedstocks that are soluble. Nutrients are also important. A suitable carbon to nitrogen ratio (C/N) is less than 30, and the carbon to phosphorous ration (C/P) should be less than 50. For example, lignocellulosic biomass has a high C/N ratio, so nitrogen sources must be added. Nutrients also must be free of toxic components. Other factors that can influence digestion are the availability and location of feed materials (transportation costs involved here), logistics of how to get materials to certain sites, and whether size reduction is going to be necessary.
Digester performance will also depend on the microbial population in the digester. This means maintaining adequate quantities of fermenting bacteria and methanogens. A recycled stream is used to take a portion of the liquid digestate as inoculum (material used for inoculation of feed materials). Depending on feeds, there may be an acclimation period to reach acceptable conditions.
There are also variations in the operational factors and environmental conditions of the digester. It is important to know the total solids (TS) and volatile solids (VS) in the feeds, the best retention times, and to provide mixing. Operational factors include the amount and strength type of feedstocks added to the digester. The operation also depends on maintaining the microorganism population and organic loading in reactors, whether operating in a batch or continuous reactor. Mixing is also an important factor in any reaction. The goal of mixing is to keep the microorganisms in close interaction with the feed and nutrients. Mixing also prevents the formation of a floating crust layer, which can reduce the amount of biogas percolating out of the slurry. Mixing will benefit the breakdown of volatile solids and increase biogas production, but keep in mind mixing adds energy cost, so this must be balanced. The types of mixing in this system include gas bubbling and/or mechanical mixing.
Environmental conditions include the temperature and pH of the reactor, as well as concentrations of materials, including the volatile fatty acids, ammonia, salt, and cationic ions. Different methanogens react in temperature ranges. The type of methanogens that produce the most biogas are thermophiles, but the digester must be operating between 40-70 °C. Methanogens also prefer neutral pH conditions (6.5-8.2). Accumulation of volatile fatty acids (VFAs) can cause the digestion to stop producing gas – this happens when too much digestible organic material is added, a toxic compound is added, or there is a sudden temperature change. Toxic materials include: 1) oxygen, 2) antibiotics, 3) cleaning chemicals, 4) inorganic acids, 5) alkali and alkaline earth salt toxicity, 6) heavy metals, 7) sulfides, and 8) ammonia. An additional reason for AD process failure has to do with the reaction within being out of balance. In particular, the rate of acid formation and methane production should be equal. This is done by maintaining definite ranges and ratios of the following: solids loading, alkalinity, temperature, pH, mixing, and controlling VFA formation. When the methanogen microorganisms cannot keep up with the fermenting bacteria, the digester becomes acidic – also known as “sour.”
An ambient temperature liquid phase AD reactor is called a covered lagoon. The advantages of the covered lagoon are the low cost, ease of construction, and control of odor control with manure storage. Disadvantages include difficult sludge removal and only seasonal production. However, there are several designs that have controlled temperature, and are typical to different types of reactors: 1) complete mixing, 2) plug flow, 3) sequencing batch, and 4) fixed film. The table below shows a comparison of the variables for each type of anaerobic digester configuration.
| Characteristic | Covered Storage | Plug Flow Digester | Mixed Plug Flow Digester | Complete Mix Digester | Fixed Film Digester | Induced Blanket Digester | Two- Stage Digester |
|---|---|---|---|---|---|---|---|
| Digestion Vessel | Clay or synthetic-lined storage | Rectangle tank in-ground | Rectangle tank in-ground | Round/square in/above ground tank | In/above ground tank | In/above ground tank | In/above ground tank |
| Level of technology | Low | Low | Medium | Medium | Medium | High | High |
| Added heat | No | Yes | Yes | Yes | Optional | Yes | Yes |
| Total solids | 3-6% | 11-13% | 3-13% | 3-10% | 2-4% | <8% | ~5% |
| Solids characteristics | Coarse | Coarse | Medium Coarse | Coarse | Fine | .. | .. |
| Retention time(days) | 60+ | 15+ | 15+ | 15+ | <4 | 3-5 | 10-13 |
| Farm type | Dairy, Swine | Dairy, Swine | Dairy, Swine | Dairy | Dairy, Swine | Dairy, Swine | Dairy, Swine |
| Optimum location | All climates | All climates | All climates | All climates | Temperate/warm | All climates | All climates |