2.3 Jet Engines
The first aircraft used engines similar to the Otto four-stroke cycle, reciprocating piston engines. The Wright Flyer was an aircraft with this type of engine. During WWII, powerful 16-cylinder, high-compression ratio reciprocating engines were developed. However, the military was interested in developing engines that would make airplanes go faster, higher, and farther - this was to reduce the length of flights and provide better international communication. In order to achieve high-speed flight, a dilemma ensued: 1) the atmosphere thins at high altitudes, offering less air resistance to a plane which could lead to higher speeds, but 2) in "thinner" air, it is more difficult to get combustion air into the conventional piston engine. The modern jet engine was developed as part of a term paper by Frank Whittle while at the British Royal Air Force College, covering the fundamental principles of jet propulsion aircraft.
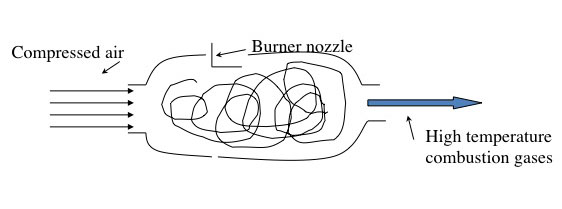
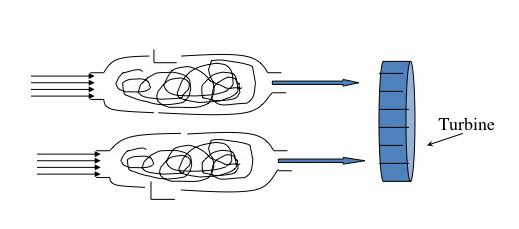
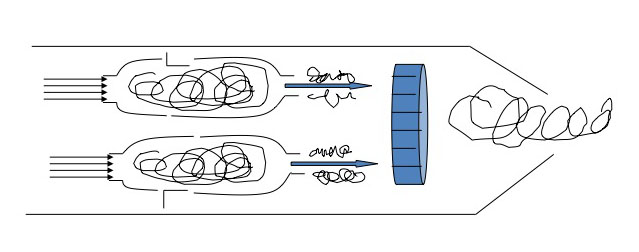
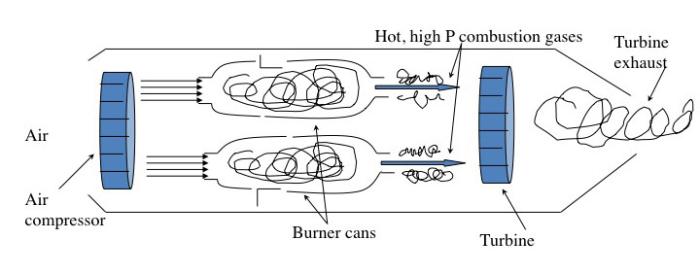
The jet engine begins with a "burner can," where jet fuel is injected and combusted in high-pressure air. The combustion produces a stream of high-temperature, high-pressure gases, as shown in the first figure below. If more power is required, two to four-burner cans can be included, and the high-temperature, high-pressure combustion gases operate a turbine (more about turbines for electricity generation in the lesson on electricity). The second figure depicts these additions. In the third figure, a containment vessel is put around the burner cans; the gases that exit the turbine pass through a nozzle. The gases exiting the nozzle provide thrust for the airplane. The fourth figure shows the completed engine - the high-pressure air comes from the air compressor, which is operated by the turbine.
There are variations on a simplistic jet engine: 1) the fan jet (turbofan), 2) the prop jet (turboprop), and 3) the turboshaft. The fan jet has a large fan in front of the engine to help provide air to the air compressor. It is a little slower than a turbojet but more fuel-efficient. This is the type favored for civilian transport aircraft. The prop jet uses the mechanical work of the turbine to operate a propeller. These types of engines are typically used for commuter aircraft. The turboshaft is a gas turbine engine that uses all of the output of the turbine to turn the blades, without jet exhaust. Helicopters, tanks, and hovercrafts use these types of engines. So, what is the fuel for jets?
Jet Fuel
Conventional jet fuel is composed primarily of straight-run kerosene (straight-chain carbons and accompanying hydrogen, bigger molecules than gasoline). However, there are some purification steps that are needed to ensure that the fuel behaves in jet engines.
The first step is the removal of sulfur. When sulfur is burned, it forms sulfur oxide compounds, such as sulfur dioxide (SO2) and sulfur trioxide (SO3). Because there are multiple sulfur oxide compounds, they are abbreviated into one chemical formula of SOx. These compounds, when combined with water, form acid rain (more on this in the next lesson on coal for electricity generation). Sulfur compounds are corrosive to fuel systems and have noxious odors. Sulfur is removed by reacting it with hydrogen and a metal catalyst; the processes are known as hydrogen desulfurization processes (HDS) and produce H2S (hydrogen sulfide), which is then reacted to solid sulfur.
Another problem that can occur with jet fuel is if it contains too much aromatic compound content. A small amount is actually necessary to lubricate gaskets and O-rings. However, aromatics are suspected carcinogens, and in combustion, aromatics are precursors to smoke and soot. Too much aromatic content can cause problems such as 1) poor aesthetics, 2) carcinogens, and 3) tracking of military aircraft. The way to remove aromatic compounds is the same as for removing sulfur; the aromatic compound is reacted with hydrogen and a metal catalyst to add hydrogen to the aromatic ring. The resulting compounds are heteroaromatics and cycloalkanes.
Another problem that can occur in the middle distillate fractions can occur if the fuel contains waxes. Waxes are higher molecular weight alkane hydrocarbons that can be dissolved in kerosene. At very cold temperatures at high altitudes, wax can either separate as a solid phase or cause the fuel to freeze and cause plugging in the fuel lines. This can also cause a problem called low-temperature viscosity. Viscosity is a measurement of the flow of a fluid; the thicker the fluid gets (and flow is reduced), the higher the viscosity. While the fuel isn't frozen, it is flowing slower and could cause problems for the engine. Again, the reason for the increase in viscosity is similar to having waxes in the kerosene; high viscosity is caused by bigger molecules within the fuel. The way to improve jet fuel properties is to remove the larger molecules. This is called dewaxing.
The last problem we will discuss has to do with nitrogen. Jet fuels do not typically contain nitrogen, but when combusting fuel using air (which contains primarily nitrogen), nitrogen oxide compounds can form, shown as a formula NOx. Because jet engines burn fuels at high temperatures, thermal NOx is a problem. NOx will contribute to acid rain. If there is any nitrogen in the fuel, it would be removed during the removal of sulfur.
A refinery will make ~10% of its product as jet fuel. The Air Force uses 10% of that fuel, so about 1% of refinery output is for military jet fuel. The figure below shows the additional processes just discussed in our schematic.
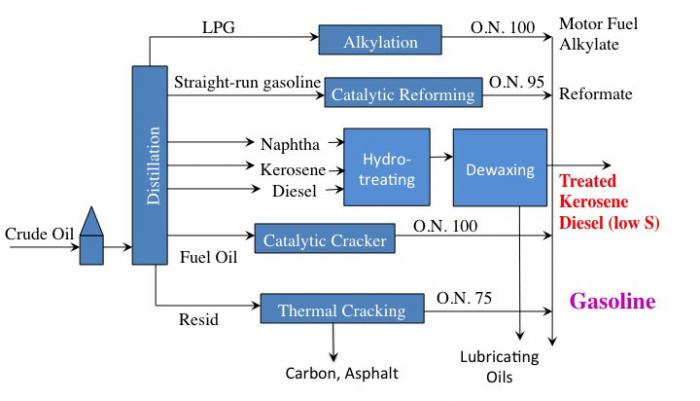
This is a simple flow diagram of a crude oil refinery.
Crude oil enters and goes to distillation. From distillation:
LPG (gases) goes through alkylation to become O.N. 100 Motor Fuel Alkylate which can go on to become gasoline
Straight-run gasoline goes through catalytic reforming to become O.N. 95 Reformate which can go on to become gasoline
Naphtha, Kerosene, and Diesel all go through Hydrotreating and then dewaxing to become either treated Kerosene, Diesel (low sulfur) or lubricating oils.
Fuel Oil goes through a catalytic cracker to become O/N 90-95 Gasoline
Resid goes through Thermal Cracking to become either Carbon, Asphalt, or O.N. 75 Gasoline.