3.2. Basic Rules of Light Quantification
Reading Assignment
- J.R. Brownson, Solar Energy Conversion Systems (SECS), Chapter 3: Laws of Light.
While reading, pay attention to the basic rules of light measurement. Our historical affinity for burning fuels has been paired with an affinity and strong awareness of temperature, but we lack an awareness for measures of light, found through radiometry. Think of how you “know” what 80 °F versus 65 °F means. Why not measure the irradiance/irradiation along with the temperature? What if we, as a solar culture, were to learn what 1200 W/m2 meant versus 500 W/m2 for an average hourly or minute irradiation upon an exposed surface (vertical wall or sloped rooftop)?
Radiometry
For solar resource assessment, radiometry (the measure of electromagnetic or radiant energy) is more valuable than thermometry (the measure of temperature). Solar energy is comprised of the shortwave band of light found between 250-2500 nanometer wavelengths of light ( , or one billionth of a meter). Within the solar field, we measure the shortwave band in terms of irradiance, as W/m2 (Watts per square meter, a flux of light per receiving area). If we group irradiance over a block of time, say an hour, we call the measure irradiation, in units of Wh/m2 (Watt-hours per area, similar units to electrical energy measure).
Quick Terms and Concepts:
- Radiometry: measure of radiant energy
- Shortwave: solar band of interest in measurement for the applied solar field
- Nanometer: wavelength units for light/photons (sometimes micrometer)
- Electron volt: energy unit for light/photon wavelengths ( )
- Irradiance: metric for flux of light received on a surface (W/m2)
- Irradiation: solar field convention, metric for energy (Wh/m2 Watt-hours per area or Joules per area)
- Photometry: measure of optically perceived light (illuminance, illumination). Photometry is not used for solar resource assessment.
Technical Shortcut:
To convert energy values as electron volts from nanometer wavelengths: divide 1239.8 nm•eV by the wavelengths that you found in nanometers to find their respective energies in electron volts (eV). Because the error is negligible for us in the field, we can even use 1234.5 nm•eV (just count to five and use the right decimal point).
For more information on how we get to this equation, see the Key Equations [1] page in the Resources box.
Traditional silicon photovoltaics collect energy for electricity production from wavelengths <1100 nm. Other systems like solar hot water panels will collect almost all of the spectrum from the Sun. In fact, almost all of the opaque surfaces about our environment (buildings, roads, water) tend to absorb strongly over the entire shortwave range (leading to an increase in temperature!).
And yet, our wonderfully adaptive human eyes only capture the tiny visible band from 380-780 nm. Thanks to the complex feedback systems from our irises, lenses, and eyelids, eyes can adapt well to extreme conditions of bright or dim lighting but cannot be used to quantitatively gauge irradiance conditions that energetically drive our buildings and photovoltaic systems. Our eyes impart important information, but because they are highly sensitive and adaptive systems, our eyes cannot be used to reliably distinguish and evaluate the solar resource (bare skin may actually be a better receptor for the shortwave band on days that are not windy).
Notice that the "visibility" of a photon is really limited only by the type of detector being used. Let's review the distribution of photons across the electromagnetic spectrum, and then we will discuss types of photon detectors beyond the rods and cones in our eyes. As a reminder, 1 micrometer wavelengths (also called 1 micron wavelengths) are equivalent to 1000 nanometer wavelengths (by unit conversion). It is often easier to use units of nanometers (nm) for the shortwave band, while using units of micrometers (um) for the longwave band. Either will work, but it is helpful to be aware of the relationship.
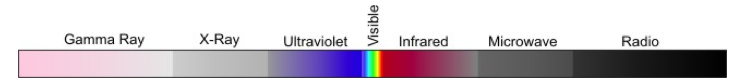
Diagram of the broad possible spectrum of electromagnetic radiation from left to right: Gamma Ray, X-Ray, Ultraviolet, Visible (small section), Infrared, Microwave, Radio
Now, let's zoom in to the shortwave and longwave bands important to SECSs. Wavelengths are expressed either in nanometers (nm) or micrometers ($\mu m$, also called microns). For wavelengths greater than 380 nm, electromagnetic radiation transitions from the ultraviolet (UV) to the violet visible range for the human eye. As we will observe in the Granqvist figure below, the red visible range ends for wavelengths greater than 780 nm (to the human eye). Notice how the "infrared" crosses over both the shortwave and longwave bands of light.
- SECS have a different operational "grouping" of bands than traditional spectroscopy or remote sensing. Get used to shortwave and longwave bands, as we describe below.
Video: Temperature and Wavelengths (0:57)
Let’s explore a simple model of how oscillation frequency is tied to the wavelength of electromagnetic radiation.
The frequency at which electrons oscillate is essentially set by the temperature of the matter in which the electron resides. Lower temperatures yield lower frequencies of oscillation. Here, we’ve set our temperature on the low side, and you can see the molecule oscillating fairly slowly, or in other words, at a low frequency. The wavelength of the emitted radiation is also relatively long.
But, when temperature increases, the oscillations get faster, which makes for a higher oscillation frequency. This high frequency means that the emitted electromagnetic radiation has a relatively short wavelength. For comparison again, we can decrease our temperature to watch the oscillation frequency slow, and the wavelength of the emitted radiation increase.
All objects glow with energy in proportion to temperature. As temperatures increase, the wavelength oscillations become shorter and the density of photons measured is effectively increased. So, the light becomes more intense (more photons), and each photon is packed with more energy.
Most photons are emitted from a thermal surface, where the constituent atoms are vibrating in the material (this means: temperature). Every material has a thermal property, and hence "everything glows." This concept works well for something called "blackbody emission" (zero emittance) or "greybody emission" (non-zero emittance) but not so well to describe your standard laser pointer or microwave oven. So, just to clarify all cases, there are also photons emitted from the Sun and from solid state devices that are "stimulated emission" (lasers), which can allow for photon emission from surfaces that are high in energy, but not high in thermal temperature.
First rule of Solar Energy: Light is Directional.
This is our first principle of seven for the behavior of light. So, let's begin with the simple yet important statement: Light is directional. Light, as a photon, is born (emitted) from a surface in many directions and then impinges upon other surfaces, where it is either absorbed, reflected, or transmitted (and refracted). We use that directionality to describe the resource. In the text chapter, we have defined a visual shorthand for light that is emitted, transmitted, reflected (scattered), and/or absorbed by various surfaces.
- Irradiance (W/m2): light that impinges upon a receiver surface, interpreted as the instantaneous rate of change in energy (called power, or Watts) per unit area. Remember that one Watt is equivalent to one Joule per second (a rate of energy exchange).
- Irradiation ($J/m^2$ or Wh/m2): light that impinges upon a receiver surface over a period of time, interpreted as the energy (in Joules or Watt-hours) per unit area.
- Radiance (W/m2/sr): light emitted from a surface, interpreted as the instantaneous rate of change in energy (call power, or Watts) per unit area per steradian (a unit of solid angle). If we were to generalize radiance over all exiting directions or angles ($W/m^2$), we term the energy emittance as radiant exitance.
- Radiation (J/m2 or Wh/m2): here, using the convention of the field, radiation means light emitted from a surface over a period of time, interpreted as the energy (in Joules or Watt-hours) per unit area. This is a loose operational definition, as "radiation" can mean many different things to different fields of study.
Notice that the "Ir-" instructs us that the light is incident upon a surface. For example, we measure "irradiance" on a pyranometer, because light is absorbed by the device. Also, notice that my choice of "-iance" or "-iation" determines if the light is a measure of rate (as in power: energy per time) or energy (rate integrated over a span of time).
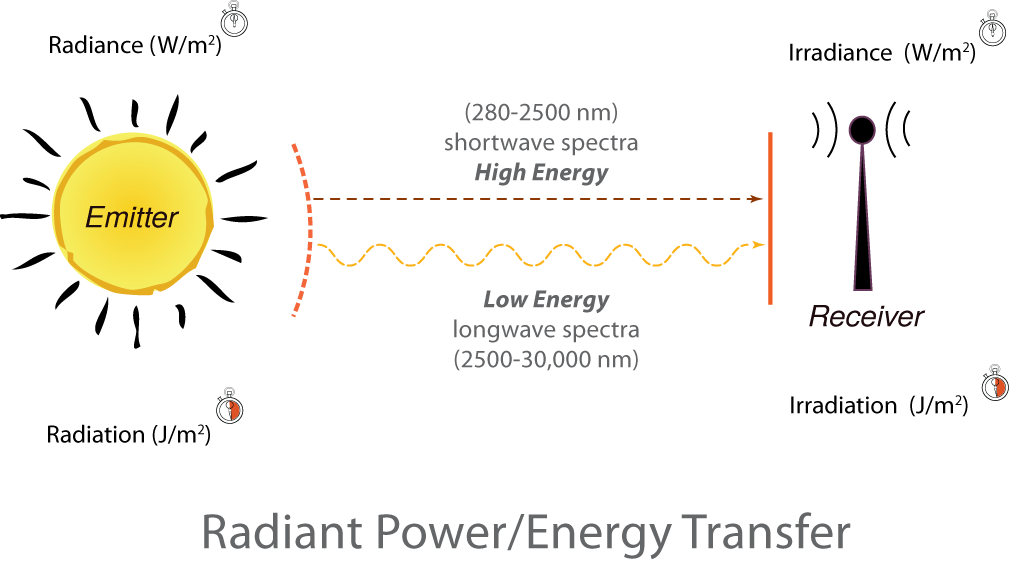
Left to right:
Left: An image of the sun is labeled Emitter. Above the sun is the label: Radiance (W/m2) and the image of a stopwatch. Below the sun is the label: Radiation (J/ m2) and the image of a stopwatch with the right half filled in.
Proceeding to the right: Two lines from the sun travel to a Receiver on the right. The top line is a straight dotted line labeled (280-2500nm) shortwave spectra High Energy. The second line is a wavy dotted line labeled Low Energy longwave spectra (2500-30,000 nm)
Right: Above the Receiver is the label: Irradiance (W/m2) and the image of a stopwatch. Below the receiver is the label: Irradiation (J/ m2) and the image of a stopwatch with the right half filled in.
Second Rule of Solar Energy: Light is Spectral.
All objects glow (even the gases in the sky); it is just the intensity and peak wavelength that shifts with respect to their thermal temperature. This brings us to the second of the seven principles of light behavior. Light (e.g., derived from the sun) has a broad spectrum of energies, which are discussed as wavelengths. We tend to group similar wavelengths in the spectrum according to the properties of their emitting surface or receiving surface.
Short Wave Band (250-2500 or 3000 nm)
This is a "group" of high-energy wavelengths of light that are emitted from the Sun (See Light is Directional), comprising the majority of the total energy collected by the Earth's surface or our designed SECS, given the decrease in the light power density with distance to Earth (93 million miles, 150 million km). This bundle of wavelengths of light emitted from the sun includes the ultraviolet (UV), the visible, and the near infrared (IR) sub-bands.
- Shortwave band: The shortwave band of wavelengths is confined from approximately 250 nm to 2500 nm at Earth's surface, because of the Inverse Square Law [3] for light and the nature of our atmosphere as an absorber/reflector of longwave irradiation. Our measurement devices are also limited to measuring this range by the transparency of low-Fe glass covers.
The Sun is effectively the only surface from our surrounding environment that regularly and naturally emits shortwave radiance. We can deliver shortwave band energy to a new surface using a reflector, though, as is the purpose of light concentration onto a central receiver.
Long Wave Band (2500 or 3000 nm - 50,0000 nm)
Long wave band consists of low energy light. The wavelengths of light that are emitted from our skin, from pavement, the sky, ice cubes, and even hot ovens are coming from surfaces that are quite cool relative to the surface of the Sun, right? We are comparing 250-500K surfaces to a 6000K surface. Planck's Law (discussed in the next page) suggests that the distribution of wavelengths will be very different from cool bodies than from extraterrestrial bodies with internal fusion reactors.
The group of wavelengths from cool bodies on Earth is termed the longwave band. This is >2500nm at Earth's surface, but also can be >3000nm if we were to group the wavelengths from space. The longwave band photons are of lower energy, but there is a very wide band of wavelengths included in the group. The longwave band contributes to the greenhouse effect, and keeps our atmosphere comfortably warm. With the addition of CO2 and water vapor to the atmosphere, we make the sky into a better reflector for keeping longwave energy instead of allowing it to escape into space.
- Longwave band: The longwave wavelengths are found in the range of 2500 nm to wavelengths greater than 50,000 nm, and again our measurement devices are limited to the range of about 30 or 50,000 nm. This second "group" of wavelengths of light is emitted by surfaces significantly cooler than the Sun! This includes the surface of our human bodies, the surface of Earth, and the effective surfaces of Earth's atmosphere (if we were to simplify the atmosphere to two surfaces in a slice of gas and particles).
Now, we're going to take a moment to explore the following multiple figure graphic from Smith and Granqvist. There is a lot of information in here, so take your time and revisit it as the class progresses.
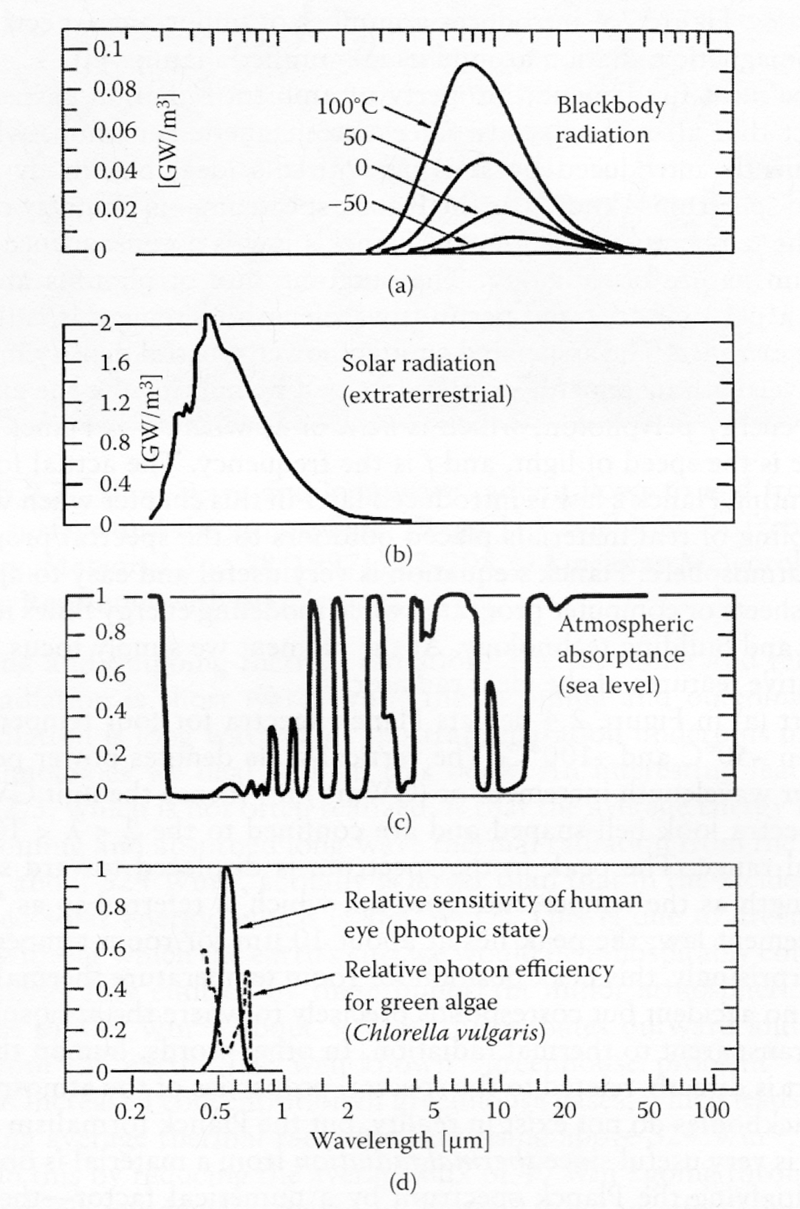
- The top graphic, Figure (a), shows the longwave spectrum of radiance (remember, that means "emitted power") from any surface at "ambient" Earth temperatures (-50C to +150C). You see a series of four humped curves that get more intense (bigger), while the peak shifts toward the left (higher energy) as hypothetical surface temperatures increase from -50C to +100C. These temperatures include the Earth's atmosphere, your body, the interior walls of your homes, even a glacier. Remember, all surfaces "glow." This spectral region is the Near-, Mid-, and Far-Infrared, and you may have seen false color images of these emitted surfaces from IR cameras before. The irradiance scales are from 0 to 0.1 W/m2/nm.
- Next, Figure (b) shows the spectrum of Solar light measured just above the atmosphere. The intensity of solar irradiance (remember, that means "received power") at the atmosphere is reduced in proportion to the inverse square of the distance from Sun to Earth. There is effectively only one temperature for the surface of the Sun, and so only one curve, which is far to the left relative to the curves in Figure (a). Also, notice that the intensity of the curve is significantly greater, with a scale from 0 to 2 W/m2/nm.
- Figure (c) shows spectral regions where shortwave and longwave radiation passes through the atmosphere. This is an absorptance spectrum; so, areas where the curve is near one (e.g., 4-8 micrometers) completely absorb the outgoing longwave from Earth and the surrounding atmosphere. Areas where the curve is near zero (e.g., 8-13 micrometers) will allow longwave light to transmit into outer space. The gap between 8-13 micrometers (8,000-13,000 nm) is called the atmospheric window, or the sky window, where the molecules CO2 and H2O do not absorb or reflect, and through which a portion of the longwave band can escape. Soon, we will design terrestrial emitters to selectively send energy out the window. This is where longwave radiation from the Earth and Atmosphere leaks radiant energy out into space. Compare Figure (c) with Figure (a) and notice how much they overlap each other.
- Finally, Figure (d) shows a solid line with a peak near 550 nm, demonstrating the relatively tiny region of sensitivity of the eye for detecting photons under daylight conditions (called the photopic state). This is a pretty small energy band, and does not include the behavior of the iris, the eyelids, and the direction your eyes are pointing with respect to your eye lenses. The double peaked dashed line (around 420 nm and 700 nm) is the efficiency of light absorption for green algae, again confined to a tiny region of the whole spectrum. Both curves are normalized fractional scales from 0-1.
Third Rule of Solar Energy: Inverse Square Law
Light intensity decreases in proportion to the square of the distance between emitter and receiver. This is called the Inverse Square Law, which applies to solar energy as well as to any other light source. In case of the Sun, the emitted energy flux is 6.33x107 W/m2 at the surface of the Sun drops down to 9126 W/m2 at the surface of Mercury (d=58 million km), and down to 1361 W/m2 at the exterior of Earth's atmosphere (d=150 million km).
As shown in the next figure, the reason for the inverse square law is geometric in nature. As light is emitted from a point, like the Sun at a very large distance, the same quantity of photons is spread out over an increasingly larger area with distance. Area is a spatial term in units of distance squared. This same principle works for ordinary surfaces, as photographers well know.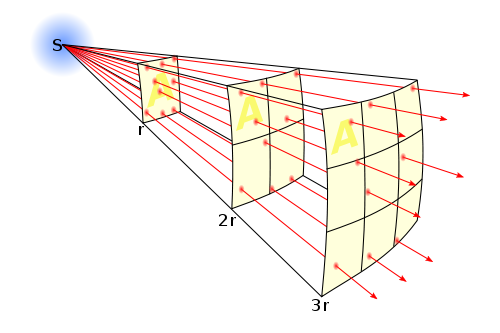
Mathematically, this process of light “dilution” can be represented by the following equation (which is another form of Equation 3.2 in the SECS textbook)
\[G_2^{} = {G_1}\left( {\frac{{d_1^2}}{{d_2^2}}} \right)\]where G1 is the light intensity (or flux) at distance d1, and G2 is the light intensity at distance d2.
If we take G1 as the radiative energy flux at the surface of the Sun, we can then estimate the radiative energy flux at the Earth’s orbit (G2) using this equation simply based on the planetary distances. Try to make this calculation with d1 being the Sun’s radius, and d2 being the distance from the Sun to the Earth and see what you get. You will have a chance to share your result in the Lesson 3 Activity further in this lesson.