7.5. Thermal - electric power conversion
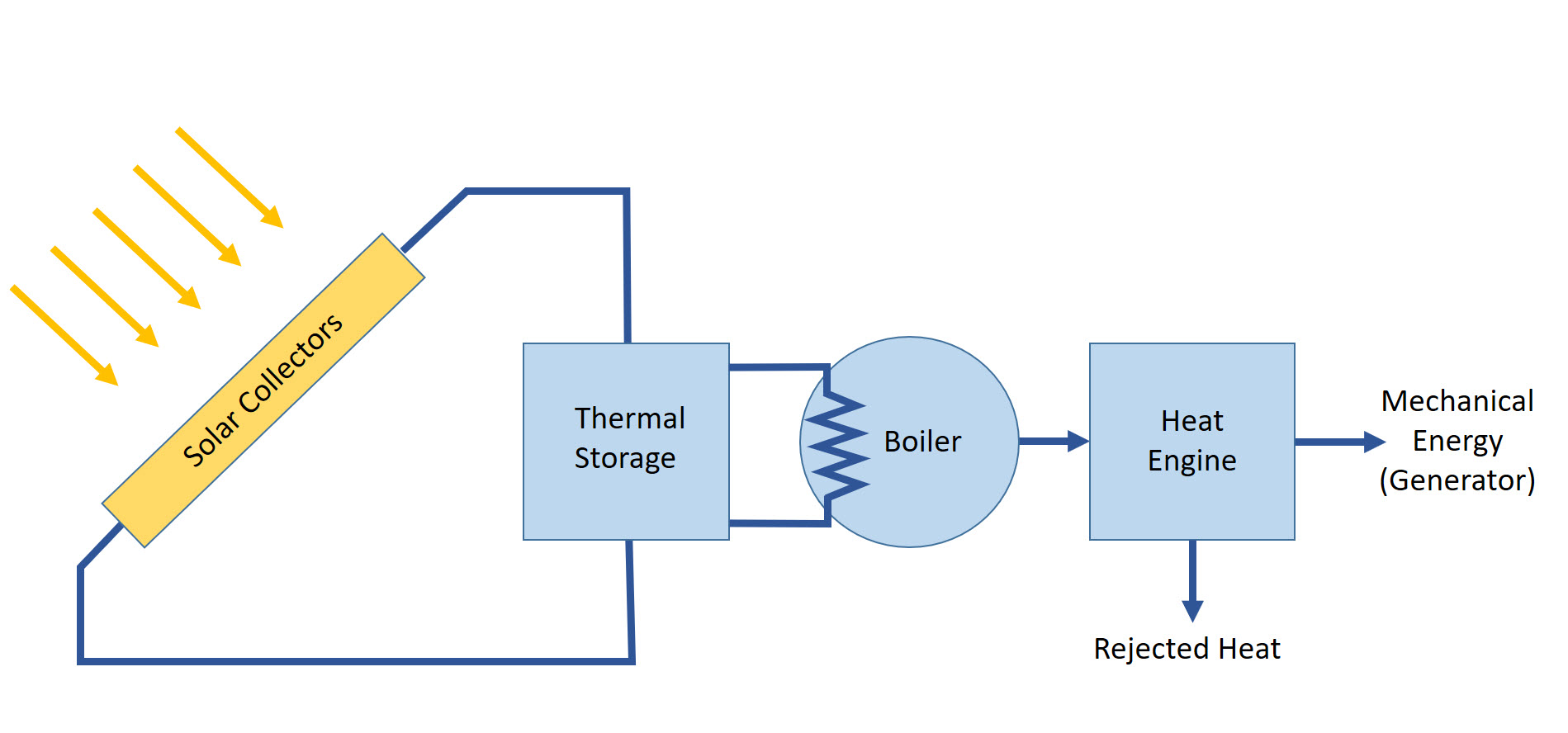
To make usable energy from solar heat collection in CSP plants, thermodynamic power conversion cycles (heat engines) are used. The main idea is quite simple. The heat transfer fluid, which is directly heated in the solar receivers, delivers heat to the boiler, which generates steam. Further steam is used in the heat engine to generate mechanical work to run electric generator. This scheme is very much the same as at conventional fossil fuel power plants, except for the heat being created not through combustion but through concentration of solar radiation.
Note
The physics of thermodynamic cycles is something you might have learned in a typical thermo class (if you have ever taken one). If not, this topic may be somewhat challenging to grasp outside the general thermodynamics context. I should note, however, that the main goal of our activity in this lesson will be to understand the general principle and applicability of different power conversion cycles to solar systems, not learning thermodynamics in detail. So, while going through readings in this lesson, you can try to filter things down to the information that would be useful from the solar project development perspective.
In general, the thermodynamic power cycles can be categorized into gas cycles and vapor cycles. In gas cycles, the working fluid is only present in the gas phase throughout the entire cycle. In vapor cycles, the working fluid can be transformed from the vapor phase to the liquid phase in different parts of the cycle. Rankine cycle, commonly used in stationary steam power plants, is an example of a vapor cycle with water as a working fluid.
In the course of a thermodynamic power cycle, the working fluid goes through a series of temperature and pressure (T,P) parameters. Changes in temperature and pressure result from heating, condensing, pressurizing, and expanding the fluid medium. Expansion creates the physical force to perform mechanical work, which is the main purpose of the system. The efficiency of the cycle is typically higher when the difference between the lowest and highest temperatures is maximized. For example, Carnot efficiency is:
The maximum temperature is set by the heating source - for example, solar concentrator. The minimum temperature is set by the ambient conditions or cooling system - for example, air, river. The Carnot efficiency of 50% is considered good for a real system.
Note
One of the problems of this conversion system is that the efficiency of a solar collector decreases as its operating temperature rises (due to increased heat losses), while it is the opposite for the heat engine part of the system, which benefits from the higher maximum temperatures. So optimization of the system has to be done to find the best operating conditions.
Several thermodynamic cycles that may be considered in connection to solar thermal applications are:
- Rankine Cycle
- Brayton Cycle
- Stirling Cycle
- Kalina Cycle
One of the criteria to combine these cycles with the solar thermal plants is the compatibility of temperature. The concentrating technologies must be efficient enough to generate high temperatures for efficient power conversion in the thermodynamic cycle. So, depending on the technology and type of solar collectors, one or another cycle may be chosen.
Another important criterion to consider is the choice of the working fluid. For higher temperatures (600 oC), steam is the best choice. For lower temperature conversions (100-400 oC), organic fluids are more suitable (Batton, 2000). The desirable properties of the working fluid are:
- low cost
- non-corrosive nature
- thermal stability
- high cycle and turbine efficiency
Possible choices for the working fluids include:
- water
- refrigerants
- organics
- ammonia
- toluene (under development)
- mixtures of above
- fully fluorinated benzene ring fluids
Currently, Rankine cycles are most promising for handling the collector temperatures.
