Lesson 1: Energy and Sustainability
Overview
This lesson provides an introduction to the course, and covers basic considerations for energy and sustainability. Given that one of the primary goals of this course is for you to be able to critically analyze claims made in contemporary materials, it is important that you have some baseline knowledge about energy and sustainability. Some of this may be review, but even if it is, it is to your benefit to go through all of the material to make sure you understand all of the concepts. If this is new to you and you find anything confusing, please don't hesitate to ask me, preferably by posting to the HAVE A QUESTION Discussion Forum.
Lesson Objectives
By the end of this lesson, you should be able to:
- define energy, energy efficiency, and the First Law of Thermodynamics;
- identify and describe types of energy and energy conversions;
- identify and define fossil fuels, non-renewable energy sources, and renewable energy sources, and their origins and characteristics;
- analyze the energy data provided in charts and graphs;
- identify reliable sources of energy data;
- evaluate the implications of sustainability definitions;
- define the "3 E's" of sustainability and use the 3E framework to evaluate the sustainability of given actions.
What is due for this Lesson?
Please note that the quiz can only be taken once. You have unlimited time to complete it prior to the deadline and can save your progress and pick up where you left off at a later time. See the Assignments and Grading section of the syllabus [1] for tips on how to do this. Once you submit the quiz, you cannot change answers. All saved answers will automatically be submitted at the deadline if you have not submitted them.
| Requirement | Submission Location |
|---|---|
| Lesson 1 quiz | Modules tab > Lesson 1 |
| Start posting to the Yellowdig discussion board | Modules tab > Lesson 1 |
| OPTIONAL Extra Credit: Lesson 1 Extra Credit quiz | Modules tab > Lesson 1 |
Questions?
If you have any general course questions, please post them to our HAVE A QUESTION discussion forum located under the Discussions tab. I will check that discussion forum regularly to respond as appropriate. While you are there, feel free to post your own responses and comments if you are able to help out a classmate. If you have a question but would like to remain anonymous to the other students, email me.
If you have something related to the material that you'd like to share, feel free to post to the Coffee Shop forum, also under the Discussions tab.
Introduction to Yellowdig
As noted in the Orientation, you will be using a discussion board application called Yellowdig. [2]For a review of these activities, see the syllabus [1]. You should start posting to the discussion board this week!
Description of how Yellowdig will be used in EM SC 240N
-
You will be making posts throughout the entire semester. I will periodically post prompts that you must follow, but you can make other comments, as long as they are relevant to the course content. The prompts will focus on the content for that week or lesson. This ongoing assignment (which is worth 25% of your grade!) serves a few purposes: First, thinking and writing about the content helps you internalize it better than reading about it. Second, it helps you learn to communicate these important sustainability issues to others. Third, it gives you an opportunity to have authentic interactions with fellow students.
-
Some of your posts will be graded using a specific rubric, but most of them are auto-graded by Yellowdig.
- Yellowdig is a points-based discussion board app. The app automatically assigns points for length, frequency, commenting on other posts, others commenting on your post, and a few other things. As you accomplish these things, your points go up. The app automatically divides the assignment into "periods," each about a week long. You can always view how many points you have out of your target points that period.
- There is no set amount of posts per se, but more substantive posts, more substantive responses to others' posts, and more frequency of posts all earn you more points. You also get points for others commenting on your post, so make it interesting!
- Please note that Yellowdig keeps a running total of points out of your target points for that period. If you are running low on points, you can bring your score up until the last commenting period is over. The end of the last commenting period is the last day of the semester.
- Each time you make a post you need to add at least one topic. (It forces you to do this, so I won't explain how to add them.) I have pre-loaded topics, all of which match up with the Lesson content.
- At the end of the semester, your score will be your grade. For example, if you get 9,000 out of 12,000 points, that is a 75%. Thus, you would get 75% of the 25% on your Canvas course grade (0.75 x 25% = 18.75%).
I hope you find this engaging, and who knows, maybe even fun! It's a great way to interact with your fellow students.
Please go to the Yellowdig discussion board link in the Lesson 1 Canvas module to read more details about how Yellowdig works.
All interactions on this discussion board and any other personal course interaction must follow the ESP Peer-to-Peer Participation Policy, which can be accessed Course Website [3]. I will be monitoring the board to make sure you adhere to these.
What is Energy?
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
Considering that this course is called “Energy and Sustainability in Contemporary Culture,” let’s start by answering two fundamental questions:
- What is energy?
- What is sustainability?
Energy
Let’s tackle the second question first, since the answer is a little more straightforward, even if it’s not always easy to grasp: What, exactly, is energy?
To Read Now
The National Energy Education Development (NEED) Project [4] is a non-profit organization that provides a lot of useful (and free!) information about energy and energy issues. Please read the first two pages of their Introduction to Energy [5], which provides a good overview of energy. Hopefully, much of this will be a review for you! (Note that we will go over more up-to-date energy use data than the document has - i.e., more recent than 2009 - below.). You are welcome to read the rest, but it is not necessary. (It is helpful, though.)
- Introduction to Energy [5], National Energy Education Development Project (pp. 8 - 9)
Energy is most commonly defined as "the ability to do work." This is a useful technical definition, but from a practical perspective, the NEED Project's indication that energy is also "the ability to produce change" is helpful. A similar way to think of energy is that it "makes things happen." Energy is required to make a TV turn on, a car to move, the sun to generate light and heat, water to vaporize, plants to add biomass, a power plant to generate electricity, and for you to think about this course content as you read it. And even if these things are not actually happening, energy provides the ability to make them happen.
As indicated in the reading, the two categories of energy are potential (stored energy) and kinetic (energy in motion), each of which have several forms. (Note that the categories are listed in parentheses below because they can either be included or not, e.g., chemical energy can be referred to as "chemical" or "chemical potential" energy. Generally, "kinetic" or "potential" is not included.):
- Chemical (potential) energy is stored in the bonds between atoms and molecules. Common examples include the energy stored in food, fossil fuels, and batteries, but anything that is made of more than one atom has chemical energy. Practically speaking, basically everything made of matter has chemical energy.
- Mechanical (potential) energy is "stored in objects by the application of a force." Common examples include a wound spring, a stretched-out rubber band, and compressed air.
- Nuclear (potential) energy is "stored in the nucleus of atoms," and is what holds the nucleus together. Anything made of matter has nuclear energy, but most of the nuclear energy converted by humans comes from the fission (splitting) of uranium atoms and is used to generate electricity. Most of the energy used by humans, however, comes from nuclear fusion (fusing of atoms) in the sun.
- Gravitational (potential) energy is "energy of position or place." Common examples include water (e.g., in a river) at a high(er) elevation, a ball sitting on top of a hill, and you sitting on your chair right now. If you see naturally flowing water, it is moving down hill (tides and waves notwithstanding), so hydroelectric energy (electrical energy generated from flowing water) starts out as gravitational potential energy.
- Electrical (kinetic) energy is "the movement of electrons." The most common example of this is electricity moving through a wire, but discharging static electricity and lightning are also electrical energy.
- Radiant (kinetic) energy is also called "electromagnetic energy." It travels in transverse waves and is produced by anything with a temperature above absolute zero. Common examples include light, sunlight, microwaves, radio waves, and radiant heat emanating in all directions from a fire.
- Thermal (kinetic) energy is the vibration of the molecules of a substance. As an object or substance gets heated up, the molecules vibrate more rapidly, and they slow down as it cools down. Humans cannot see this vibration because it happens at a molecular level, but we can feel it, or at least the results of it. Have you ever accidentally touched a hot stove and gotten burned? That unpleasant sensation is the result of the quickly vibrating molecules of the stove imparting their thermal energy into your skin. Anything above absolute zero has thermal energy, so it is all around us all the time, including everything you see right now.
- Motion (kinetic) energy is the energy in moving objects. Anything with mass that is moving has motion energy. Moving cars, flowing water, a falling object, and even wind (air is made of matter, after all!) are common examples.
- Sound (kinetic) energy moves in waves and is produced by vibrating objects. When you hear something, it is the result of the bones in your ear absorbing and converting these waves into motion energy, which your brain then interprets as sound. Despite what you may have heard, if a tree falls in the woods and there is no one there to hear it, it does generate a sound! Well, it generates sound energy, at least.
Energy efficiency and conservation of energy will be addressed later in this lesson.
The gentleman in this video (4:35 long) also provides useful information regarding energy and illustrates many of the concepts from the reading above. (In case you are wondering, yes, he is this excited all the time. He also has a number of really good videos regarding many topics. His YouTube channel [6] has over 6,000,000 subscribers, so he must be doing something right!). Please note that you can open this video in YouTube by clicking on the title of the video in the window below.
As you probably know by now, we’ve been working with Google and YouTube to answer ten of the most popular science questions asked on the Internet. And I gotta hand it to you, because there are few questions that are as confounding and complex and fascinating and inspiring, as this one the collective consciousness has spewed forth:
What is energy?
I'm Hank Green, and this is the World’s Most Asked Questions.
[Intro]
Energy is everything. It’s everywhere. It’s one of the true constants of the universe because as long as there’s been a universe, there’s been energy. And while it comes in lots of different forms that can seem different to us, they all amount to the same thing: Energy is the ability to do work. And work is just the act of displacing something by applying force.
So, say you stomp on a stomp rocket. The force of your foot hitting the pedal is turned into the force of air leaving the cannon -- sending your rocket sailing. Or maybe you're enjoying a nice patty melt -- the energy from that food is broken down for all of the quadrillions of cells that you have to do all of the things that they have to do -- make copies of your DNA, assemble and repair proteins, transport materials from one place to another, make muscle cells contract -- you know, all the stuff of being alive. That rocket sailing, your cells toiling away, your phone or computer being on right now to watch me -- that’s all work being done. And the ability to do these things is inherent in everything around you. Even things that look inert, completely lacking in energy. Like this log.
This log, for example, is chock full of chemical energy because it’s made up of combinations of carbon and hydrogen and oxygen formed into lignin, which is the stuff that makes up wood. All of the bonds between all of those atoms, in every molecule of lignin, contain energy. How do I know? Because if I were to apply enough extra energy, like as heat, to break those bonds -- it would release that chemical energy as fire. That chemical energy is also the kind of energy you get from your patty melt -- your body is fueled by the chemical bond energy in sugars, fats, and proteins. But this log also contains nuclear energy! Each atom in this wood has a nucleus, made of protons and neutrons, and the energy that binds them together is one of the most powerful sources of energy in the universe. If you could split one of the atoms of carbon or hydrogen in this log, and rip those protons and neutrons apart, it’d release some of that energy. There’s so much nuclear energy in each atom that, if I could unleash all of it that's in this log? There’d be a giant smoldering crater where I’m standing and everyone in the town of Missoula, Montana, would be dead.
So, everything that’s made of atoms has nuclear energy locked up in it, but also, it turns out, that mass and energy are the same thing! You might have heard of this little equation that a German patent clerk came up with about a hundred years ago: E = mc2. And there are SO MANY OTHER KINDS of energy that I’d love to get into if we had the time ... but even though they may seem different, they can all be used to do work, whether it’s driving a turbine, or moving an engine piston or allowing the screen on your tablet to glow. Or, if it’s that most mysterious of energies, dark energy, causing the universe to expand more than it seems like it should. But here’s the thing to remember.
Once the work is done, the energy isn’t done. Because energy never goes away. It can never be destroyed, and in the same way, it can never be created. It can only be transferred from one source to another -- like, how the energy in the plants and animals that were in the patty melt were transferred into you -- or it can be transferred from one form into another -- like the chemical energy in the wood being transferred to light and heat as fire. You could think of the universe as a constant flow of energy, and we are just little pit stops along the way. Everything your body is doing right now -- whether it’s your lungs absorbing oxygen, your heart pumping blood, your brain cells firing as you watch me and learn things -- all those things are using recycled energy that’s been around since the origin of the universe. And by simply being alive, you’re releasing that energy back into the environment around you, to be used by other things in other ways.
So Internet, to answer your question: Energy is everything. And for those of you who answered our questions on our SciShow survey, where you feel like you get your energy may be keeping you up at night. Survey takers who have a hard time falling asleep nearly every night get their energy from knowledge first, second from purpose. Least likely? From exercise. Of all the fascinating questions in the world, what question do you want to see answered most? Let us know on Facebook or Twitter or down in the comments below and we will aim to answer those questions in a new video at the end of the month.
Okay, so if you ask a physicist or energy expert what energy is, she will likely tell you that energy is the ability to do work. This sounds straightforward enough, but you may be thinking, “what is work?” Ask the same (or another) expert, and you will likely hear: “Work is the transfer of energy.” The video below from Kahn Academy (3:16) is optional but does a good job of explaining what this means. If you are still a little confused after watching it, you may want to read through the rest of the energy lesson, then go back to it. The formulas are not important for this course, but the concept of how work is related to energy is important. One thing to note: the narrator uses the term "Joule" a lot in this video. A Joule (J) is the international unit of energy and is simply a way to quantify energy. (More on quantifying energy shortly!)
Optional Viewing
Work as the transfer of energy from the Khan Academy (3:17 minutes)
One way to find the amount of work done is by using the formula Fd cosine theta. But this number for the amount of work done represents the amount of energy transferred to an object. For instance, if you solve for the work done and you get positive 200 joules, it means that the force gave something 200 joules of energy. So, if you have a way of determining the amount of energy that something gains or loses, then you have an alternate way of finding the work done, since the work done on an object is the amount of energy it gains or loses.
For instance, imagine a 50-kilogram skateboarder that starts at rest. If a force starts the skateboarder moving at 10 meters per second, that force did work on the skateboarder since it gave the skateboarder energy. The amount of kinetic energy gained by the skateboarder is 2,500 joules. That means that the work done by the force on the skateboarder was positive 2,500 joules. It's positive because the force on the skateboarder gave the skateboarder 2,500 joules. If a force gives energy to an object, then the force is doing positive work on that object. And if a force takes away energy from an object, the force is doing negative work on that object.
Now imagine that the skateboarder, who's moving with 10 meters per second, gets stopped because he crashes into a stack of bricks. The stack of bricks does negative work on the skateboarder because it takes away energy from the skateboarder. To find the work done by the stack of bricks, we just need to figure out how much energy it took away from the skateboarder. Since the skateboarder started with 2,500 joules of kinetic energy and ends with zero joules of kinetic energy, it means that the work done by the bricks on the skateboarder was negative 2,500 joules. It's negative because the bricks took away energy from the skateboarder.
Let's say we instead lift the bricks, which are 500 kilograms, upwards a distance of four meters. To find the work that we've done on the bricks, we could use Fd cosine theta. But we don't have to. We could just figure out the amount of energy that we've given to the bricks. The bricks gain energy here. And they're gaining gravitational potential energy, which is given by the formula mgh. If we solve, we get that the bricks gained 19,600 joules of gravitational potential energy. That means that the work we did on the bricks was positive 19,600 joules. It's positive because our force gave the bricks energy.
This idea doesn't just work with gravitational potential energy and kinetic energy. It works for every kind of energy. You can always find the work done by a force on an object if you could determine the energy that that force gives or takes away from that object.
[MUSIC PLAYING]
In order for an object to gain or lose energy, work must happen. If you pick up a book from the ground and put it on a table, the book gained gravitational (potential) energy. You performed work on the book, and the amount of work is equal to the amount of potential energy gained. When you pull your car or bike out from a parking spot, the car/bike has motion energy, but when it was parked had none. That energy gain is the result of work done by the car engine (then drivetrain and wheels) or your legs (then pedals, chain, and wheels), and you can figure out the work done by considering the velocity and mass of the moving object. When the vehicle stops, the bike/car performs work on the road and tires, resulting in them heating up.
The sun is constantly generating massive amounts of radiant energy. That energy is provided by hydrogen atoms fusing together into helium and releasing nuclear energy. The amount of radiant energy generated in this process is equal to the amount of work done by the hydrogen atoms on the sun. When this sunlight hits your skin (or any object), it performs work on it, resulting in a gain in thermal energy. This gain in thermal energy is equal to the amount of work done.
I could go on and on, but the key thing to remember is that energy transfer requires work. Any time energy is transferred from place to place or from one form to another, work must be done, and the amount of work is equal to the amount of energy gained or lost.
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Thermodynamics and Energy Transfer
Learning Objectives Self-Check Edit (active tab)
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
Forms of Energy
It can be easy to get bogged down by the formulas used to calculate how much work is done, when, by whom, and to whom. Since this is not a physics class, let’s not dwell on those. As described on the previous page, a somewhat simplified, but very useful way to think of energy is that “energy makes things happen.”
Try This
Take a few minutes to look around you. Based on what you know about energy and what you learned in the reading and videos, what is energy “doing” where you are right now? (Seriously, take a look.)
We could go on and on. But as you probably know, these are all examples of kinetic energy, or “energy of motion.” As stated in the reading and video, there are also a number of types of potential energy. Think of some examples of potential energy around (and in) you right now. You are able to move and think because of chemical (potential) energy inside of your body. In fact, everything around you has chemical potential energy. Any object on the wall, on a table, attached to the ceiling, or just above the ground has gravitational (potential) energy because it is above the ground. There is also nuclear (potential) energy in all matter because all matter has at least one nucleus. Again, we could go on and on, but the point is that everything around you has potential energy - nuclear if nothing else - and thus has the ability to do work, i.e., “to make things happen.”
Conservation of Energy
One of the foundational concepts in the understanding of energy – and something that is very important in the context of this course – is the First Law of Thermodynamics. The simplest way to put the First Law of Thermodynamics is that “energy cannot be created or destroyed – it can only change forms.” This is often referred to as the “Law of Conservation of Energy,” for obvious reasons. Practically speaking, this means that all energy came from somewhere else, and that it does not disappear when it is “used.”
All of the examples of energy that were noted above came from somewhere else. The light coming from a light bulb is converted from electrical energy running through a wire. The heat radiating from non-living things around you was absorbed from another source such as sunlight or the heating system of the building. The motion and electrical energy your body has right now comes from the chemical energy inside of your body. The gravitational energy of things around you came from motion energy required to lift the objects. And so on. And recall that each time energy was transferred, work was done.
Good to Know
NASA defines thermodynamics [11] as "the study of the effects of work, heat, and energy on a system." Thermodynamic principles are relevant to many applications, including things as diverse as nanotechnology, health sciences, refrigeration, climatology, manufacturing, space travel, and much, much more. If you are interested, here are some thermodynamic resources: Kahn [12]Academy [12] and Hyperphysics [13] (from Georgia State University) [13].
Of course, this also means that all of the previous forms of energy also came from somewhere else. Where do you think the electricity used to generate the light coming from the screen came from? It almost certainly came from a power plant somewhere. But where does the power plant get its energy from? If you live in the U.S., chances are it came from either coal, natural gas, or nuclear material (about an 80% chance nationally [14], but it depends on where you live).
Let’s assume the electricity in question is from a natural gas-fired power plant. If so, the electricity used to generate the light on the screen you are looking at right now was originally chemical (potential) energy stored in the molecules of natural gas. Note that before it was converted to electricity, it went through a number of conversions, including being burned (thermal and radiant energy), and spinning a turbine (motion energy). But let’s not stop there. Where did the natural gas get its energy? Before we answer this, please read the short readings below.
To Read Now
Knowing where to get reliable information is an important skill to have. If you want U.S. energy data, there is no better source than the U.S. Energy Information Administration [15] (US EIA, or simply EIA). The US EIA is an outstanding source of information, from specific energy use data to general energy information. For the energy geeks among us <raising hand>, there is so much interesting (and reliable) information that hours can be spent perusing, reading, and watching videos. Please read the following pages before moving on. You are, of course, welcome to explore the rest of the website, but at least read the links below:
- Introduction to natural gas [16] (read through the first 3 subheadings in the "Basics" tab and the "Did you know?" box.)
- Introduction to coal [17] (read through the "Basics" tab)
- Introduction to oil [18] (read through the "Basics" tab and browse the "Data and Statistics" tab). Also browse the imports and exports [19]page.
- Introduction to nuclear [20] (read through the "Basics" tab)
Natural gas is formed from the remains of living organisms over millions of years, as are coal and oil. Most of this is from photosynthetic organisms, such as plants and phytoplankton [21](e.g., diatoms [22]). If so, then the energy came from the sun. If it was an animal that formed the gas, then the energy came from what the animal ate to gain that energy, i.e., a plant or another animal. If it ate a plant, then that energy originally came from the sun, but what if it ate another animal? That animal either got its energy from a plant or another animal.

What this boils down to is that no matter how you slice it, all of the energy in natural gas came from the sun. The implications are kind of mind-boggling (and let’s face it, awesome): The light energy coming from the screen you are looking at right now probably started out as sunlight that hit the earth millions of years ago!
Fossil Fuels
Coal, oil and natural gas are considered fossil fuels because they are derived from remains of ancient organic material. They are also all hydrocarbons (technically, oil and natural gas are made of multiple hydrocarbons), which indicates that they are made primarily of carbon and hydrogen. You will often see coal, oil, and natural gas referred to by either name - fossil fuel or hydrocarbon.

Again, we could go through innumerable examples of energy, and most of them would require tracing multiple steps to find their original source. Almost all sources (aside from some nuclear energy and some geothermal energy) can be traced back to the sun, whether it’s recent or ancient sunlight. But more importantly in the context of this course is that:
- all energy comes from somewhere else, and
- energy is a quantity of “something” that takes many forms and can be converted from one form to another.
As the saying goes, “there ain’t no such thing as a free lunch.” In other words, when we “use” energy, that energy must come from somewhere else, and it does not disappear, it is converted to another form.
Good to Know
Almost all of the energy used on earth came from the sun, but where does the sun get its energy? Sunlight is nuclear energy released when atoms of hydrogen fuse to form helium, in a process called fusion. This reaction releases a HUGE amount of energy - the surface of the sun is nearly 6000 °C (more than 10,000 °F), and the core is more than 20 million degrees C [25] (36,000,000 °F)!
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Quantifying Energy
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
It should be clear by now that energy can take many different forms and is often converted from one form to another. Though different forms of energy cannot always be used the same way (ever tried to watch TV by plugging into a lump of coal?), you can always express the amount of energy present in different forms using the same units by using unit conversions. There are many energy units, but the most common unit you’ll see in the U.S. is the British Thermal Unit or Btu. Joules are considered the international unit of energy (you may see these from time to time in the U.S.), but since we like to make things difficult for scientists in the U.S. by using English units instead of metric, we’ll stick mostly to Btus in this course.
A Btu is defined as the amount of heat required to heat up one pound of pure water one degree Fahrenheit. To give you some perspective, a single match releases about one Btu if it is allowed to burn entirely.
The following are examples of commonly used energy equivalencies, i.e., unit conversions:
- One kilowatt-hour (kWh) of electrical energy is equivalent to exactly 3412 Btus of energy.
- Each time you burn a gallon of gasoline in a car, you convert approximately 120,000 Btus to other forms of energy (mostly as waste heat, it should be noted).
- Every 100 cubic feet (ccf) of natural gas that is burned releases approximately103,700 Btus of energy. (Note that 1 ccf = 100 cubic feet, so there are approximately1,037 Btus in 1 cubic foot, or cf).
- You know the calorie labels on the side of packaged food? Those are actually kilocalories, and each one is equivalent to just under 4 Btus of energy.
- There are about 1,055 Joules (J) in 1 Btu.
These are but a few examples - you can pick any amount and any form of energy, and it can be converted to Btus or any other energy unit. The US EIA has a useful unit converter [27].
This is useful in many ways, one of them being that it is possible to tally up all of the energy “used” by a given person or group of people – including a city, state, country, continent, or even planet – and convert that number to a single quantity to see how much energy is being used. Further, it is often possible to separate total energy use into categories to compare uses. This can provide a nice snapshot of energy use and can tell you a lot about the energy regime in an area, including how much is being wasted.
Visualizing Energy Use
The U.S. Department of Energy (DOE) is part of the Executive Branch of the U.S. government. According to whitehouse.gov [28]: "The mission of the Department of Energy (DOE) is to advance the national, economic, and energy security of the United States." The DOE is another excellent source of information (the US EIA is run by the DOE). In addition to providing information, the DOE funds a lot of research, much of which is performed by people in the national labs. There are 17 national labs [29] in the U.S., each with a different research focus. The national labs host some of the top researchers in the U.S., and because they are funded by taxpayers, all of the non-sensitive information is published for free. These are great sources of reliable and cutting-edge information. (Feel free to browse the national labs [30]' website.)
Apropos to our discussion of energy use, Lawrence Livermore National Lab (LLNL) [31] in California publishes annual energy use data for the U.S. and often for U.S. states. The image below (click on it to see a larger version) shows the most recent estimate of energy use in the U.S., divided by source. IMPORTANT: LLNL uses quads as their fundamental unit. As mentioned in a previous reading, a quad is a quadrillion Btus, which is 1,000,000,000,000,000 BTUs, or 1 x 1015 Btus. (Side note: This is one of my favorite charts! I appreciate the amount of information it provides and the ease with which it can be interpreted. It tells a robust - and important - story about energy use in the U.S. I can't be the only one that has favorite charts, can I? Anyway, moving on...)
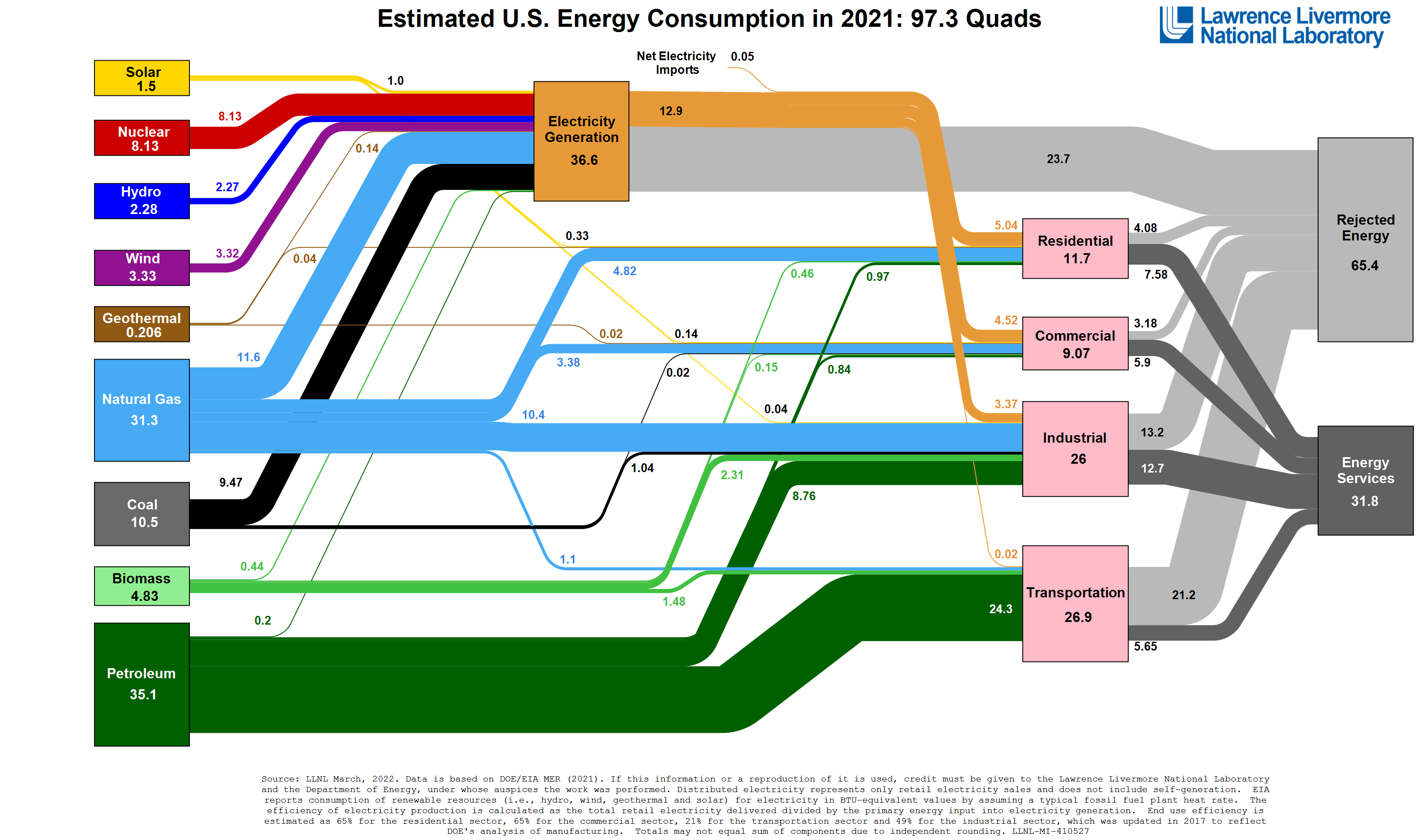 [32]
[32]
You can click on the chart to open a larger version in a new window.
he "blocks" on the left are energy sources (also called primary energy), the pink blocks on the right are end-use sectors (note that electricity is NOT an end-use sector), and the grey blocks to the far right indicate whether or not the energy was successfully used ("Energy Services") or wasted ("Rejected Energy"). All of the numbers in the chart indicate total energy flows or uses. Think of this as a flow chart - follow the lines from left to right to see how energy is used in the U.S.
Let's look at coal as an example. (Find coal on the left side of the chart, then follow the lines coming from coal on the chart and observing the numbers associated with those lines.):
- The U.S. used about 10.5 quads of coal in 2021 (this is up from 9.7 in 2020 and down from 17.9 quads in 2014 [34], by the way).
- Of that, 9.47 quads were burned to generate electricity, and
- 1.04 quads were used in the industrial sector (mostly to create heat for things like making steel), and
- 0.02 quads were used in the commercial sector
You can see where each energy source was "used" by following the chart. Oil is mainly used in the transportation sector but is used in all others as well. Natural gas is used in many sectors too. Nuclear is only used for electricity generation. All of this can be seen by following the energy sources on the left to the end uses on the right of the chart.This type of diagram is called a Sankey diagram and can be used for any number of purposes. Lawrence Livermore creates Sankey diagrams for each state, and many countries have diagrams as well. There are even some used to describe water and carbon flows in the U.S. At any rate, it is a useful tool for analyzing energy and other resource flows.
Check Your Understanding - Chart Analysis
Answer the following 2 questions.
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Energy Efficiency
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
Explain what it means when an appliance is 40% efficient, in terms of input and useful output.
Though many forms of energy can be converted to many others, it is important to consider how efficient the conversion process is. Energy efficiency is the percentage of "useful" energy that is converted from another form.
For example, have you ever thought about what it means to have an "efficient" light bulb, like a light emitting diode (LED)? Think about it - the purpose of using a light bulb is to provide light. Seems obvious enough, but did you know that about 90% of the energy used by an incandescent light bulb is actually converted to heat? Only about 10% is converted to light, which means that incandescents are about 10% efficient. If you are still using these old-style light bulbs, you are wasting about 90% of the money you spend on the electricity used, unless you are purposefully using them to heat your house (this is a very expensive way to heat your house, by the way). This is one reason why CFLs have become so common, and now LEDs ("light emitting diodes") - both of them are around 40% - 45% efficient, which is 4 - 4.5 times as efficient as an incandescent.
Good to Know
Consumers have a wide array of energy efficient lamps available to them. In addition to using electricity more efficiently, CFLs last about 10 times longer than incandescents, and LEDs around 25 times longer.
Efficiency considerations can be made for anything that uses energy. An efficient car is one that gets a lot of miles (useful "output") per gallon (energy input). An efficient home heating system, such as an electric heat pump, releases a lot of heat energy (output) for each kilowatt-hour of electric input. TVs, cell phones, airplanes, refrigerators, you name it - all have a certain efficiency. It can be used in other contexts as well. If you are efficient at work, you get a lot done (output) in a short period of time (input). In an efficient outing by a baseball or softball pitcher, not many pitches (input) were required to retire the batters (getting outs is the useful output).
This leads us to one aspect of the Second Law of Thermodynamics. A full explanation of the 2nd Law goes beyond the scope of this course, but you are welcome to watch the video below (9:29) from the Kahn Academy for a short explanation. One application of this law is that it is impossible to convert energy into a more dense, useful state without adding energy to the system. As Dr. Eric Zencey of the University of Vermont describes it, "the capacity of the energy to useful work is diminished" whenever it is transformed from one form to another (source: Is Sustainability Still Possible? p. 73). In other words, when energy is converted from one form to another, it is impossible to convert all of it. Some is "wasted" in another form, usually heat.
Let's continue with the lighting example to illustrate this. When using a light, electrical energy is converted almost entirely to light and heat (there may be a little sound energy thrown in there, but not much). Electrical energy is relatively dense, useful, and easy to control. You can store electrical energy in a battery. It is relatively easy to transport across distances without losing much. It can be used for many different things. But what about light and heat? Both of them are relatively diffuse and difficult to control. Neither is particularly useful for converting to other forms. It is very difficult to convert heat or light into another form with any kind of efficiency. Sure, you can convert heat back into electricity. In fact, this is exactly what happens in a typical power plant. But this process is very inefficient. Going further back, it is impossible to convert light, heat, or electricity back into coal (or oil, natural gas, or nuclear energy). Fossil fuels are very energy dense, and the molecules and atoms are neatly organized. Once the bonds are broken and the energy is released, there is no way to put it back together. That's the 2nd Law in action.
To Watch Now
The video below provides a very good explanation and animation of how a coal-fired power plant works. Think it's as easy as dumping a bunch of coal into a furnace and turning a turbine? Watch the video to find out. (9:28 minutes)
First Energy operates twenty power plants in the northeast United States that produce nearly seventy million megawatt-hours of electricity each year for over 4 million residential, commercial, and industrial customers. Over half of First Energy's electricity is produced by using coal compared to about 60 percent nationwide. Let's see how electricity is generated from coal by taking a virtual tour of a coal-fired power plant similar to those operated by First Energy.
This plant has three generating units that produce more than 2,000 megawatts of electricity. At full capacity, this one power plant can produce enough electricity to supply the needs of one and a half million homes and businesses. Generating electricity requires a fuel source. At this plant, the fuel source is coal which arrives mainly by a barge but also by rail and truck. Each barge delivers 1500 tons of coal - enough to keep the plant running for a couple of hours. The plant uses about 21,000 tons of coal each day, so over a million tons of coal is stockpiled next to plant. A device called a stacker reclaimer scoops coal onto a quarter mile long conveyor that can transport up to 900 tons of coal into the plant each hour.
Once inside the plant, up to a thirty-hour supply of coal can be stored in bunkers. Coal moves from the bunkers to feeders to be measured and moved to pulverizers. Each generating unit has 16 coal feeders that supply 8 pulverizers. Coal enters the pulverizer and spins in a large drum where hundreds of steel balls grind it into a fine powder.
Now a fine powder, the coal leaves the pulverizers and heads to the boiler. About 335 tons of coal can be pulverized per hour. Large fans add warm air to the powdery coal and blow it into the boiler. The boiler has miles of tubes filled with high quality water. Once inside the boiler, the coal ignites, releasing energy and generating intense heat. This changes the water inside the tubes to hot dry steam. The temperature of the steam is now about 1,000 degrees Fahrenheit. This process generates thermal energy which is transformed to mechanical energy at the turbine which is the next stop on our tour.
High pressure steam, now one thousand degrees Fahrenheit at 3500 pounds per square inch, is piped from the boiler to the first in a series of turbines. Here it expands between layers of turbine blades mounted on the turbine shaft the steam loops back to the furnace, then on to the second turbine. In this process, the steam turns the series of turbines 3600 revolutions per minute, providing power to the generator, the last in the series of machines. The generator continuously creates an electrical charge of 34481 amps at 18000 volts of electricity. From here, the electricity leaves the plant and begins its journey to customers.
After the steam has been used to create electricity, it is exhausted from the turbine and sent to a condenser to be changed back into water. Inside the condenser, steam passes over the outside of pipes filled with chilled water from the cooling towers. The steam condenses to water and returns to the boilers to repeat the steam generating process over again. As the steam condenses, the chilled water inside the condenser pipes becomes warm from the heat of the steam, so it is sent to a cooling tower. These particular towers are called natural draft cooling towers, which are designed with no moving parts. Inside, the water splashes over a series of baffles which breaks up water into small droplets. These droplets mix with air from the open bottom of the tower, evaporating some of the water and cooling the rest by as much as 27 degrees. The newly chilled water returns to the condenser to repeat the process, cooling more steam.
Since 1971, First Energy has spent more than $5 billion dollars on environmental protection. Our plants are equipped with air quality control systems to remove sulfur dioxide and particulates called fly ash. Fly ash is removed by a mechanical process, and sulfur dioxide is removed with a chemical reaction using lime. These processes take place in large ductwork scrubber trains located between a unit's boiler and its chimney. Boiler gases pass through scrubbing vessels and are sprayed with slurry, a mixture of water and lime. The sulfur dioxide particles and other pollutants are absorbed by the slurry and fall to the bottom of the vessels. A fan releases the cleansed gases through the chimney. By the time this happens, more than 99 percent of particulates have been removed, as well as ninety-two percent of sulfur dioxide. A plant similar to this one can remove over 400,000 tons of sulfur dioxide each year.
The scrubbing process creates a huge amount of a byproduct called calcium sulfite. This plant could create more than 3 million gallons of calcium sulfite slurry each day. First Energy created a process that turns that waste product into a valuable building material called gypsum which is used to make wallboard or drywall. The slurry leaving the scrubbers is thickened and pumped through a forced oxidation gypsum or fog system. Oxygen is then added changing it into gypsum which is dried treated and sent to a gypsum manufacturer to make wallboard.
First Energy recycles about a half million tons of gypsum each year - enough to manufacture wallboard for seventy thousand new homes.
Precipitators are another method of extracting coal ash, also called fly ash, which can be recycled or deposited in landfills. This mechanical process extracts fly ash left over from the combustion process. These poles, called rappers, shake fly ash from a vibrating wire. Gas from the furnace containing fly ash flows into the box. The fly ash drops into a storage chamber and is blown into silos to be loaded into a truck.
Fly ash from First Energy plants is used in manufacturing a variety of products, including concrete crowding roofing shingles, grouting, and anti skid road materials. First Energy also use a selective catalytic reduction technology or SCR to reduce nitrogen oxide emissions. SCR systems work much like a catalytic converter on a car. Flue gas containing nitrogen oxide emissions from the combustion process is mixed with ammonia. The mixed gases travel through a series of catalytic layers which causes the nitrogen oxides to react with the ammonia. The reaction converts the nitrogen oxides to water vapor and pure nitrogen, a benign chemical that makes up eighty percent of the air we breathe. Both elements are returned to the environment through the station stacks.
Now that we've taken a look at how electricity is produced and some of the environmental control protection systems used in our plants, let's take a look at how the electricity is delivered to our homes and businesses. Transformers located just outside the plant boost the power from 18,000 volts to 345,000 volts. So it can travel long distances over transmission lines to where it is needed. Substations located along the path reduce the voltage so it can be moved along distribution lines attached to the utility poles. As the electricity nears customer locations, transformers on poles step down the power for usage in homes and businesses. We hope you enjoyed seeing how electricity is produced at First Energy where our energy is working for you.
Optional - Explanation of the Second Law of Thermodynamics
The second law can be confusing, but the narrator in the video below does a pretty good job of explaining some aspects of it. Watch the Second Law of Thermodynamics (12:40 minutes) from Kahn Academy.
Let's talk about the Second Law of Thermodynamics. This law is weird. There's about 10 different ways to state it, which is one reason why it's weird. Let's start with one of the most common ways to state it, which is, if you've got a cold object and a hot object, heat will never be seen to flow spontaneously from a colder object to a hotter object. So if you have these two sitting together, maybe an ice cube and a hot piece of metal, and you make them touch, heat's going to flow between them, but we know what's gonna happen. The heat's gonna flow from the hot object to the cold object, and never the other way. At least, not spontaneously. You can force heat from a cold object to a hot object, like we do with a refrigerator or a freezer, but that's using a heat pump. And those refrigerators and freezers are doing work to force that heat from the cold region into the hot region. It won't do it spontaneously by itself. You've got to force it to do it. So what the second law says, or at least one version of it, is that that process will never be seen to happen in reverse. The heat will never be seen to flow from the cold object to the hot object. Now, you might be thinking, "Duh. "Do we really need a law to tell us that?" But it's not so obvious, because you can still conserve energy and momentum and all the other rules of physics and laws of physics by allowing heat to flow from the cold object to the hot object. In other words, let's say the cold object started with 10 Joules of thermal energy and the hot object started with ... It's hotter, so let's just say it has 30 Joules of thermal energy. You could imagine five Joules of energy going from the cold object into the hot object which would leave you with five Joules of energy for the cold object, 35 Joules of thermal energy for the hot object. You still have 40, just like you did before. You didn't break the law of conservation of energy. It's just, energy won't go that way. So why? Why is thermal energy never seen to flow from the cold object to the hot object, even though it satisfies every other known law of physics besides the second law? Well, before we answer that question, I think it'd be useful to talk about an alternate version of the second law, which looks something like this. The total disorder will never be seen to decrease. What do I mean by "disorder"? Imagine you had a room and there were blue spheres. And they're bouncing around wildly. So these all have some velocity and random directions. And when they strike a wall or each other, they lose no energy. So they keep bouncing around like crazy. And then there's another section of the room with red spheres, and these are also bouncing around randomly. They lose no energy. They keep doing their thing. Except, there's a divider in this room that doesn't allow the red spheres to go onto the blue spheres' side, and vice versa. These can't mix up. So right now, this is an ordered state because the reds are separated from the blues. So we say that this state has a certain amount of order to it. But let's imagine we removed the divider. Now what's gonna happen? Well, you'll see these things mix up. This blue sphere will move over here, and it'll bounce onto this side. This red sphere will go over here. They'll just keep getting mixed up. And at some given moment, you might find the spheres in some configuration like this. They're still bouncing around, but now they're all mixed up and we say that this state has a higher amount of disorder. This is not ordered. We say that this is more disordered, which supports the second law. The second law says, if you let things do what they wanna do spontaneously, your system will go from a more ordered state to a more disordered state. And you'll never see it go the other way. We can stand in this room and wait. But you're probably never gonna see the blue spheres line up all on the left side and the right spheres line up on the right side. With 12 total spheres, maybe if you wait long enough, a really long time, you might catch it where all the red spheres are on one side and blues are on the other. But image this. Imagine now, instead of six reds and six blues, there's 100 reds, 1,000 reds, maybe 10 to the 23d and Avogadro's number of reds, and now they're all mixed up. The odds of ever seeing them get back to this ordered state are basically zero. The probability isn't exactly zero, but the probability is very, very low that you would ever see a disordered state with that many number of particles reassemble themselves into an ordered state.
So we kind of just know that from experience and what we've seen in our day-to-day lives. But you still might be wondering, "How come? "How come we never see a disordered state Well, it basically has to do with counting. If you were to count all the possible ways of lining up the reds over here on this side and the blues on the left-hand side, there'd be a lot of combinations that would satisfy that condition. I mean, you could swap this red with that red, and this red with that red, all on the right-hand side. All these reds could get swapped around. And these blues, as well. They can get swapped around on the left-hand side. You get a large number of variations that would satisfy the condition of blues on the left, reds on the right. But now I want you to ask yourself, how many possibilities are there for having blues and reds spread out through the whole room? Well, you could probably convince yourself, there's more. And it turns out, there'll be a lot more. its position on the right-hand side somewhere. This red can get swapped out anywhere over here. I can swap a red with this blue, and this blue with this red, and this red with this red, and this blue with this blue. I can move them all over. Now that these spheres have the whole room through which they can mix, the amount of states that will have blues and reds mixed throughout the whole room will vastly outnumber the amount of states that have just reds on one side and just blues on the other side. And this simple idea is the basis for the Second Law of Thermodynamics. Roughly speaking, the Second Law of Thermodynamics holds because there are so many more disordered states than there are ordered states. Now, I'm gonna tell you something that you might not like. This particular disordered state that I have drawn, this exact one, is just as likely as this exact ordered state. In other words, if I get rid of the barrier over here, if you came in, you'd be just as likely to find the room in this exact configuration as you were to find it in this exact configuration. These two exact states are equally likely, which sounds weird. It makes you think, "Well, you're just as likely But no. This particular state is just as likely as this other particular state. But there are so many more mixed-up states than there are separated states. Even though any particular state is just as likely, since the mixed-up states vastly outnumber the separated states, if you pick one at random, it's gonna be a mixed-up state because there are so many more of them. Imagine putting these all into a hat. Imagine writing down all the possible configurations of states, ordered, disordered, in between. You put them all into a hat, you pull one out randomly, any particular state is just as likely. But since there's so many more disordered states, you pick one out randomly, it's probably gonna be mixed up. And if there's a large number of particles, you're almost certain to find it mixed up. So to help us keep these ideas straight, we need some different terms. Physicists came up with a couple terms. One is a macrostate. And a macrostate is basically saying, okay, the particle are mixed up. That's one possible macrostate. And we could be more precise. We can say, the reds and the blues can be anywhere within the box. Another possible macrostate would be to say that the particles are separated, that is to say, reds are on this side, anywhere on that side, but on the right side, and blues are on the left side, anywhere on the left side. These terms are referring to a macrostate, an overall description of what you would see. Now, there's another term, a microstate. And a microstate is a precise, exact description of the nitty-gritty details of what every particle is doing within there. If I just tell you, "The particles are mixed up," you're not gonna know exactly where they are. Similarly, if I just tell you, "They're separated," you're not gonna know exactly where they are. You'll know they'll be on the right-hand side, the red ones will, but you won't know. Maybe this red ones moves down here, maybe this red one moves up here. The microstate is an exact description. This red one's right here, going a particular speed. This blue one's right here, going a particular speed. If you specify the exact location, blue right here, blue right there, going that fast, red right here, what you're describing to me is a microstate. And so the second law, another way of thinking about it, there are more microstates for a disordered macrostate than there are micorstates for an ordered macrostate. And that's why we see systems go from order to disorder. It's really just a statistical result of counting up the possible number of states. You might be wondering, what does this have to do with heat going from hot to cold, all this talk about microstates and macrostates? Well, it's not just position that can get disordered. It's velocities that can get disordered, energy that can get disordered, and that's more of like what's happening up here. The positions of the hot molecules aren't necessarily moving over into the cold range. But the energy over here is getting dissipated into the cold area. So image it this way. Let's get rid of all this. And imagine you had a room with a gas in it, but this gas was kind of weird. At this particular moment, all the gas molecules on the right-hand side were moving really fast, and all the gas molecules on the left-hand side were moving really slow. So the room was separated into a cold region and a hot region, just like this energy is. This is ordered, or at least, somewhat ordered. It's more ordered than it's going to be. If you wait a while, this is all gonna mix up. You're gonna have some fast-moving particles over here, some slow ones over here. It's all gonna be blended together.
And so, what would you say if you were standing in here? At first, you'd feel cold because these particles don't have a lot of energy. Then you start feeling warmer and warmer. You'd say heat is flowing over to the left because you feel faster-moving particles striking your body. And so you'd rightly say that heat is moving from the right of this room to the left of this room. It flows from the hot to the cold. And that's what's happening up here. Heat flows from the hot to the cold. You might object. These are solids, I said, copper and an ice cube. A copper atom's not gonna make it over into the cold ice cube. But the energy is gonna move. So you can make the same argument over here. Don't allow these, let's say these are the copper atoms moving around fast, or at least jiggling in place rapidly. When they bump into the slower-moving water molecules in the ice cube, they're gonna give those water molecules some of their energy. And this energy's gonna become mixed up. The energy will become disordered. It will go from this ordered state, where the high energy is over here and low energy's here, to a disordered state where the energy's distributed somewhat evenly. So essentially what I'm saying is, if you consider the macrostate, where the hot molecules are separated from the cold molecules, there will be less microstates that satisfy that condition than there will be microstates that satisfy the condition for a macrostate where the energy is mixed up and you're just as likely to find a fast-moving particle on the left as you are on the right. This will have vastly more microstates, many more possible ways of making up a mixed-up state than there are microstates that create a separated state. I mean, there's gonna be a lot. I'm talking a lot of microstates that satisfy this condition for this macrostate, separated. But there will be so many more microstates for the mixed-up case, this dominates. That's why you always see heat flow from a hot object to a cold object, just because it's statistically inevitable with the large number of particles that you have here. There are so many more ways of heat flowing from hot to cold than there are from cold to hot, statistically speaking, you just never see it go the other way. Energy will always, at least spontaneously, if you let it do what it wants to, energy's always going to dissipate and evenly distribute. That's why it goes from the hot to the cold. This energy's trying to get mixed up, just because statistically, there are so many more ways for that to happen. Now, I need to tell you that there's actually a scientific term for the amount of disorder, and we call it the entropy. Physicists use the letter S to denote the entropy. And if you wanna know the formula for the entropy, you could look on Bolzmann's grave. This is Ludwig Bolzmann. He's got it on his gravestone. How awesome is that? The entropy S is k, Bolzmann's constant, times log. This is actually natural log of W. And W is the number of microstates for a particular macrostate. So you got some configuration, you wanna know the entropy? Just look at what macrostate it's in, count up how many microstates are there for that macrostate, take log of it, multiply by Bolzmann's constant, that gives you the entropy. And there's a term for this W. It's called the Multiplicity, because it's determining the multitude of microstates that satisfy the conditions for a particular macrostate. Now, entropy is cool. Entropy is weird. Entropy is somewhat mysterious and still, probably, has secrets for us to unlock here. I don't have time to go into all of them here, but if you read up on it, entropy has a role to play in the fate of the universe, the beginning of the universe, the arrow of time, maybe our perception, all kinds of facets of physics that are extremely interesting. And entropy, you always find this guy lurking around. And one place you always find entropy is in the Second Law of Thermodynamics, because it allows us a third way to state the second law, which is that the total entropy of a closed system will always be seen to increase. Technically, if it's a reversible process, the entropy could stay the same. But honestly, for all real-world processes, the entropy's gonna increase for a closed system, which is to say that the disorder increases.
Here is another optional link regarding the 2nd Law [39].
Clearly, a lot of engineering goes into building a power plant. Despite the technical prowess required to convert coal into electricity, the process is extremely inefficient, as are all of the major forms of electricity generation in the U.S. and the world. Take a look at the chart below to see just how inefficient this process is for different fuels.
As you can see, as the most efficient fuel, natural gas-fired power plants are just above 40% efficient on average. Coal is closer to 30%. This, of course, means that around 70% is wasted as heat. 70%! And this does not take into consideration the losses associated with transporting the electricity across long power lines, which in the U.S. averages around 5%.
Power plants are not alone in their inefficiency. The typical internal combustion engine of a car only provides around 20% - 25% of the energy from gas to move the car. New natural gas furnaces are very efficient (95%+), but many older ones operate at lower than 80% or even 70% efficiency. This is all poor energy management in principle - it's just plain wasteful - but it is also important for a couple of other reasons, one in particular. Specifically, there is a limited amount of all of these sources, and yet they are essential for modern society. In other words, coal, oil, natural gas, and nuclear are non-renewable energy sources. (To be fair, all indications are that the world will not run out of coal, natural gas, oil, or nuclear energy terribly soon, but no one knows when it will become too expensive to use. More on that later.)
The "Fifth Fuel" (Or Perhaps the "First Fuel")
One last note before moving on to renewable and non-renewable sources. Energy efficiency is sometimes referred to as the "fifth fuel." Why do you think that is? (Hint: coal, oil, natural gas, and nuclear are the four primary fuels used globally, though that is changing as renewables are used to a greater extent.)
Increasing efficiency reduces the use of other sources of energy. Efficiency is on the demand side of energy use because it affects energy demand (think of this as how much energy is "demanded" for use.) Energy sources are the supply side of energy use because they supply the energy. By reducing demand through energy efficiency, you reduce the need for supply, which is almost like having more supply, to begin with. Hence, it is sometimes referred to as the "fifth fuel." There are tremendous opportunities for energy efficiency improvements worldwide.
Some energy efficiency advocates refer to efficiency as the "first fuel," because they feel that it should be the top priority in terms of energy management. There is some strong validity to this. Consider that a report [42] from the American Council for an Energy Efficient Economy found that it is cheaper to reduce energy use through efficiency than it is to supply energy by any other source. Very interesting reading, if you are so inclined (and only a few pages long).
Check Your Understanding - Efficiency
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Renewable and Non-Renewable Energy
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
What is the difference between renewable and non-renewable energy?
Knowing whether a source of energy is renewable or non-renewable is important when considering energy and/or sustainability. Renewable energy is defined by the U.S. Environmental Protection Agency thus: “Renewable energy includes resources that rely on fuel sources that restore themselves over short periods of time and do not diminish” (Source: U.S. EPA [43]). Non-renewable energy is energy that cannot restore itself over a short period of time and does diminish. It is usually easy to distinguish between renewable and non-renewable, but there are some exceptions (more on that in a minute).
To Read Now
Once again, we will go to the US EIA for a description of renewable energy sources. Please read through the following links (quickly, if nothing else). You are welcome to read the sub-headings for each source, but that is not necessary. The key points are summarized below.
- Solar [44]
- Wind [45]
- Wind Turbines (video) [46] (Transcript [47])
- Hydropower [48]
- Biomass [49]
- Biofuels [50]
Optional Readings/Videos
- Solar Photovoltaics video [51]
- Geothermal [52]
Please note that these readings only scratch the surface of the world of renewable energy sources! The DOE's "Energy 101" YouTube channel [53] has a bunch of good, short videos about different energy sources, note that there are a lot more energy-related links on the EIA websites that you read just now. You could spend your whole career learning about them (as some people do), and still have more to learn. I encourage you to learn as much as you can about these and other sources. It may sound like hyperbole, but you can never know too much about energy. It figuratively and literally makes the world go 'round.
Renewable Energy
It should be clear how most of these sources fit the definition of renewable energy ("resources that rely on fuel sources that restore themselves over short periods of time and do not diminish") and have various benefits and drawbacks. Please note that this does not provide a comprehensive list of pros and cons, but will give you a solid idea of many of them:
- Solar energy comes directly from the sun, which comes every day in most locations and does not diminish appreciably over time. Yes, the intensity does ebb and flow on short and long timescales, but it is hopefully not going away anytime soon. If the sun burns out and stops shining, we have bigger problems than solar panels not working!
- Pros: A few benefits of solar energy are that it is relatively predictable and reliable, it is effectively limitless, and that it does not create any emissions/pollution when generating energy.
- Cons: The main drawback is that it is intermittent, both in terms of the sun only being in the sky 50% of the time, and that weather can impact it significantly. Solar is also very diffuse, meaning that it is not very concentrated, and so, usually a large area is required to provide a lot of useful energy. Solar PV used to be very expensive but is now cost-competitive.
- The wind gets its energy from the sun - it is caused mostly by differential heating across the surface of the earth - so cannot be "used up" either.
- Pros: More good news is that the wind will never disappear as long as the sun shines and the earth is spherical, and like solar, wind does not generate emissions. Well-sited onshore wind is actually the least expensive form of electricity.
- Cons: However, the wind is also variable - more in some locations than others - and is less predictable than solar energy in most locations.
- Hydropower is the power in moving water and gets its energy from the sun as well and is even more consistent in most locations than the wind.
- I want you to think for a moment how the energy in moving water started out as solar energy. (This is a good thought experiment in energy conversion.) Answer: Remember that water flows downhill, and so the motion energy in flowing water started out as gravitational potential energy. How does water get this potential energy, i.e., how does it get uphill? Mostly from evaporation caused by the sun!
- Pros: In terms of other benefits, like solar and wind, hydropower does not generate emissions, and is very consistent and reliable in most locations. Though it should be noted that some methane emissions result when organic material behind dams decomposes.
- Cons: There are some drawbacks associated with large hydropower installations (see the EIA's Hydropower and the Environment website [54] for some examples), and in some cases, very big environmental and social drawbacks (e.g., in the Three Gorges Dam [55] in China). All of these factors are important to keep in mind. Hydroelectricity is the single biggest source of renewable electricity in the world.
- One additional drawback of all of the above sources is that they are each location-specific. In other words, some locations may have a lot of sun, wind, and/or hydro, while others may have very little. (This will be addressed in more detail in a future lesson.) This problem can be at least partially solved by transporting electricity, but that is not always easy, and often expensive.

All of these sources renew themselves over short periods of time and do not diminish. And though intermittent, none of these sources are going to disappear in the foreseeable future. They are textbook renewable energy sources.
Good to Know: Agrivoltaics
Agrivoltaics are a burgeoning systems-thinking application. Agrivotaics combines - you guessed it - agriculture and photovoltaics. Ground-mounted solar arrays are a great application of solar PV technology, but they do take up a lot of space relative to their energy output. So why not find a way to use all of this space? Enter agrivoltaics! With some careful design considerations (e.g. knowing which plants are shade-tolerant or even prefer some shade), crops can not only be successful but in some cases more successful in terms of production than when planted in an open field. This is particularly helpful in hot, dry climates, such as the eastern part of Colorado, which is pictured below. But it can be successful in more humid and cooler climates as well.
Agrivoltaics are becoming increasingly recognized and researched throughout the U.S. and internationally. Feel free to browse through the National Renewable Energy Laboratory's (NREL) article about agrivoltaics [57] for more information.

Okay, so what about biomass and biofuels? They are both derived from living or recently living things (trees, corn, algae, sugarcane, etc.) They also get their energy from the sun (anyone sensing a pattern here?), and plants are usually pretty good at regenerating themselves. But I want you to take a minute to try to think about examples of biomass and/or biofuels that might not be "renewable," in the sense of the definition above. Can you think of any examples of non-renewable biomass?
Nearly all forms of biomass and biofuels are renewable. Corn-based ethanol is the most-used source of bio-based energy in the U.S. Corn can be grown in the same field year after year, so it is renewable. Whether or not it is sustainable is another question, which will be addressed later. The primary source of bioenergy in Brazil is sugarcane. Nearly all of Brazil's vehicles are able to use 100% sugarcane ethanol for fuel. (Contrast this with the U.S., where most automobile engines are only required to be able to handle up to 10% ethanol.) Sugarcane grows year-round in Brazil, so is definitely renewable.
There are many other biomass sources that fit our definition of renewable, including animal dung, algae (for biodiesel), jatropha nut, soybean, switchgrass, and more. Wood is used around the world as a source of heat, particularly for cooking. Most trees and shrubs regrow relatively quickly, so they are generally considered renewable. But even a fast-growing tree like an oak (up to two feet per year, according to the National Arbor Day Foundation [58]) has limits. Though most biomass sources are considered renewable, keep this in mind: if you harvest a renewable resource faster than it regenerates, it will not be able to renew itself over time. We will revisit this point in a later lesson, but it is important to remember.
Not all Renewables Are Created Equal
Most renewable energy sources are carbon-free. This means that they do not emit any carbon dioxide when they generate energy. Solar, wind, and hydroelectric are carbon-free. Nuclear, though not renewable, is also considered a carbon-free energy source, because unlike coal and natural gas, it does not burn. As noted in a previous reading, nuclear energy generates heat through fission, not combustion. Biomass and biofuels are often considered carbon-neutral because they emit carbon dioxide when they are burned. So, why are they carbon neutral?

Good to Know
The International Energy Association (IEA) is a good source of information for international energy data. The US EIA (not to be confused with the IEA) publishes some international data as well, but IEA is usually the first place I look. The chart below shows the estimated energy use by type worldwide in 2016, and the data are from their 2018 Key World Energy Statistics [59] document. Note that the energy unit they use is Mtoe, which stands for million tons of oil equivalent. A Mtoe is equivalent to about 0.04 quads. In other words, there are about 25 Mtoe in a quad. The amount of energy is not important for our purposes, but please take note of the percent of the total that each energy source provides.
FYI, another good, reliable source of global energy data is BP's annual "Statistical Review of World Energy [60]." It is worth browsing if you are interested!
There are a few interesting things to point out from the chart above.
- First of all, Total Primary Energy Supply (TPES) refers to all original or primary energy consumed. For example, if your electricity is supplied by a power plant, the energy your electronic device is using right now is not primary energy because the electricity was converted from an original source (e.g., coal, oil, natural gas, nuclear). Given that electricity generation is always less than 100% efficient (sometimes much less, per the previous section), the primary energy used by your device is greater than what shows up on your electric bill. Incidentally, the "energy sources" on the left-hand side of the sankey diagram that you looked at earlier this lesson are primary energy.
- Another interesting thing to point out is that biofuels are lumped together with "waste." In many parts of the world, including many states in the U.S., if you burn garbage to produce heat and/or electricity, it is considered a biofuel, and thus renewable. I'll leave it to you to think about whether or not that is reasonable. But note that biofuels (and biomass) constitute the majority of that "slice" of the global energy pie.
- Hydro is at only 2.5%. But where are wind and solar? We hear about them all the time in the U.S., and in other parts of the world. If you read the fine print at the bottom, you'll see that wind and solar's contribution was so small that it was lumped into the "other" category, at a measly 1.7%. This has improved since 2015 (and was 1.1% of the total in 2012), and in fact has been growing at an all-time high rate, but there is still a long way to go before wind and solar make a major dent in the global energy regime.
Non-Renewable Energy
Non-renewable energy sources diminish over time and are not able to replenish themselves. In other words, they are finite, and once they are used, they are effectively gone because they take so long to reform.
You have already read about the four non-renewable energy sources: coal, oil, natural gas, and nuclear. Let's start with coal, oil, and natural gas, which (as you read earlier) are referred to as fossil fuels. Fossil fuels were created from the remains of dead plants and animals. The source material is renewable (it's biomass!), but since they take millions of years to form, they are not replenished over a "short" period of time, so are non-renewable. Fossil fuels are forming somewhere under your feet right now, but don't hold your breath waiting for them to finish.
The nuclear energy we use comes from an isotope of uranium called U-235. Unlike fossil fuels, U-235 has cosmic origins: it was formed by one or more supernovae around 6 billion years ago, about 1.5 billion years before the Earth was formed (a supernova is a collapsing star, "supernovae" is the plural form of supernova) (source: World Nuclear Association [62]). Again, this is not renewable on a human timescale.
All fossil fuels emit carbon dioxide (CO2) and other emissions when they are used to generate energy. Recall that they are made mostly of hydrogen and carbon, and the carbon mostly ends up as CO2. Nuclear is considered carbon-free, because it is not burned, and it is not made of carbon. Remember that energy is extracted through fission or splitting of atoms. This generates heat, but no emissions. (It is important to note that it does result in very dangerous and long-lasting radioactive waste, but that will be addressed in a future lesson.)
To summarize:
Non-renewables
- Coal, oil, and natural gas are fossil fuels. Even though they all get their energy from the sun, none of them are renewable. They all emit CO2 and other emissions when burned.
- Nuclear is also non-renewable, but not a fossil fuel. It is carbon-free but causes radioactive waste.
- Most importantly, for all intents and purposes, whatever coal, oil, natural gas, and nuclear exists today is all that we will ever have.
Renewables
- Solar, wind, and hydro are renewable and carbon-free, and effectively inexhaustible.
- Bioenergy is renewable and carbon-neutral. It emits CO2, but no more CO2 than was originally pulled from the atmosphere. Even though it is considered renewable, it is possible to use bioenergy unsustainably by harvesting it more quickly than it can be replenished.
Check Your Understanding
All of the carbon dioxide emitted from coal, oil, and natural gas was originally pulled from the atmosphere to make the plants from which is was derived grow. In other words, the amount of carbon dioxide emitted is no more than the amount of carbon dioxide it originally removed from the air. Why are they not carbon-neutral energy sources?
Good to Know
We hear a lot about renewables and natural gas in the U.S., as their use has been growing rapidly for some time now. But as you can see in this chart from the EIA, coal and nuclear still constitute over 40% of all electricity generation in the U.S. Solar, despite its massive growth and growth potential, is only 1.8%! We have a long way to go, people!
 [63]
[63]
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Sustainability
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
Hopefully, you now have a reasonably good grasp of what energy is, how it is used, where we get it from, whether or not it is renewable, as well as some good resources for finding energy information. I do not expect you to be energy experts, but it is important that you possess a good baseline knowledge of energy basics if you are going to critically analyze material that has energy information in it. There are many free information sources available, some of which I listed in the previous pages. If you have suggestions for other sources, feel free to share them on one of the course discussion boards.
Sustainability and Sustainable Development
Okay, time to shift gears and address sustainability. Unlike energy, sustainability (and “sustainable”) does not have a universally accepted definition. The phrase “sustainable development” is usually used to describe the goal of sustainability planning, and is often used interchangeably with the term “sustainability.” For the purposes of this course, the terms are effectively the same. Before we start really digging into the term, it’s good to start with the root word “sustain.” Dictonary.com's most relevant definition of “sustain” [65] is:
“to keep up or keep going, as an action or process”
This lies at the core of the term, and is a good place to start. If something is being done that cannot continue to be done for the foreseeable future, then it is not sustainable. The devil is in the details, though, as we will see.
Suggested Reading
The reading in this box is not required. I summarize the key points below. But it will help you understand the content in more depth.
There is almost an unfathomable number of books, articles, and websites that address sustainability. I just Googled "sustainability" and got 437,000,000 results in 0.65 seconds! There is no shortage of information out there, nor is there any shortage of definitions of sustainability. Robert Engelman, President of the Worldwatch Institute, does a very good job of cutting through some of the "sustainable" and provides some cogent thoughts on the state of sustainability and how it can be framed in the book Is Sustainability Still Possible, by the Worldwatch Institute. You are welcome (but not required) to read his entire "Beyond Sustainababble" chapter [66]. I have provided key excerpts below, which I suggest you read before moving on. I have emphasized some key text in bold lettering:
We could spend weeks analyzing the content of Engelman's chapter, but I would like to focus on a few key points.
1. The Overuse of the Term "Sustainability" and "Green"
First of all, what it means to be sustainable (and it's even fuzzier substitute "green") is open to interpretation at best, and misuse at worst. (Greenwashing is an example of such misuse, and will be addressed in more detail later in the course.) Since there is no single definition of sustainability, anyone is free to use the term to describe whatever they want, regardless of whether or not it is truly sustainable. Sustainable travel, sustainable consumption, sustainable underwear, sustainable food, green growth, green cars, greenhouses, green energy - as Engelman puts it, "frequent and inappropriate use lulls us into dreamy belief that all of us - and everything we do, everything we buy, everything we are - are now able to go on forever, world without end, amen" (p. 4).
How often do people stop and think about what it really means to be sustainable or green? Engelman points out, and I must say I agree, that too often it means "better than the alternative." But simply doing "better" is almost certainly not going to be enough to achieve a sustainable world. Hopefully, the content in this course will help you find out why!
2. The Brundtland Commission and Intergenerational Equity
Engelman also mentions the Brundtland Commission's definition of sustainable development:
Sustainable development "meets the needs of the present without compromising the ability of future generations to meet their own needs." (Source: Is Sustainability Still Possible?, p. 3. Original source: Our Common Future, World Commission on Environment and Development. Full text available here. [67])
This is the most commonly cited definition of sustainability/sustainable development, in part because it appeared in a book - Our Common Future, published in 1987 - that was the first organized international attempt (in this case, by the United Nations) to address what was widely seen as a global problem. Namely, the commission was tasked with analyzing and proposing solutions for the unsustainable course on which the world's societies were on. But it is also a good, concise way to sum up some primary goals of sustainability. Perhaps most importantly, it acknowledges the need to focus on the world that we leave to future generations. As Engelman puts it, we need to ask ourselves "whether or not civilization can continue on its current path without undermining prospects for future well-being" (p. 4). It is important to point out that not only does society need to simply "last" or "continue" for sustainability to happen, but that we need to consider the quality of life of people living in future societies. This concern is often referred to as intergenerational equity. We will investigate the quality of life in more depth in future lessons.
On paper, the goals indicated by this definition may seem pretty straightforward:
- allow the current generations to continue to thrive,
- improve the lots of those that are currently suffering, and
- make sure future generations are able to meet their "needs."
But what is a "need," exactly? Is it meeting the bare essentials of survival, e.g., food, shelter, and clothing? Do I need to have a car? Do you need to have 3+ solid meals a day? Does your neighbor's family need that guest bedroom for when family visits? Do working Germans need to have four weeks of paid vacation each year? Does the mother or father in rural Kenya need a cell phone if there are no landlines? Does India need to update its outdated electricity infrastructure? It's hard to argue that any of these things are true needs, but if you asked each person in this situation, they would all probably say that they are, or at least that they are an important aspect of their lives.
Further, as Engelman brings up, to what degree do we sacrifice the needs and wants of the current generation in order to maximize the chances of future generations to live a good quality of life? Are you willing to impact your quality of life by buying fewer things, not traveling by airplane, not eating meat, living in a smaller house, not owning a car, and growing your own food, just so people in the future can live a better life? I would argue that some of these things actually improve the quality of life for you right now, but who has the right to decide what quality of life means? And how can we guarantee that any of this will work? None of these questions have easy, obvious, or even objectively correct answers, but they are all important to ask if we are to address sustainability.
3. Environmental Concern
There is something explicitly missing from the Brundtland Commission's definition (though it is implied) and from any part of the discussion so far, though it is mentioned in the book chapter. What about the natural environment? There are a few ways to approach this question - nature-centric (ecocentric) and human-centric (anthropocentric) - but for now, let's focus on the anthropocentric approach.
The anthropocentric sustainability implications of human concern for nature are concisely summarized by the US EPA when they note that "everything that we need for our survival and well-being depends, either directly or indirectly, on our natural environment" (Source: US EPA [68]). We will investigate this further through ecosystem services in a future lesson, but the logic is impossible to argue against: If we destroy nature, we destroy ourselves. At the very least, the oxygen we breathe is generated by plants and other organisms like phytoplankton, and the food we eat is reliant upon soil and water, though there are many more things we currently depend on nature for. Many would argue that nature has value in and of itself (this is generally referred to as deep ecology or ecocentrism), but that goes beyond the scope of this course.
4. The Importance of Metrics
As Engelman stresses throughout his chapter, if we are to know whether or not we are living sustainably, we must measure it. In his words, sustainability "must be tied to clear and rigorous definitions, metrics, and mileage markers." If we do not define and measure it, how can we know whether or not we are closer or farther away from achieving it? These are often called metrics or indicators, and there are many of them, including levels of biodiversity, pollution levels, quality of life metrics, economic indicators, percentage access to clean water and energy, and more. Engelman mentions concentrations of carbon dioxide (CO2) in the atmosphere, which the best science indicates is very likely the major cause of global warming trends, as a very important metric. This will be addressed in more detail later in the course, but suffice to say the trend is pointing in the wrong direction, and possibly already at dangerous levels. There are many other indicators that are at a varying level of (non-)concern, some of which will be addressed later. Unfortunately, Engelman is mostly right when he writes that "the basic trends themselves remain clearly, measurably unsustainable."
5. Economics and Systems Thinking
Finally, Engelman addresses the fraught relationship between economic prosperity and sustainability, and the difficulty in satisfying both present and future needs. Ridding the world of abject poverty is at the forefront of sustainability goals, and is addressed in future lessons. But unfortunately economic growth and sustainability - particularly environmental sustainability - are often at odds. For example, increasing access to fossil fuels generally helps facilitate improving economic conditions, but causes unsustainable emissions. Even current and future sustainability can be at odds, e.g., when Engelman notes that: "Safe water may be reaching more people, but potentially at the expense of maintaining stable supplies of renewable freshwater in rivers or underground aquifers for future generations."
This all indicates the importance of systems thinking. There is a lot of literature about systems thinking, and it does not have a single definition. (If only the world of sustainability were so simple!) It can be thought of as analyzing the world around us as a collection of interrelated systems, and considering phenomena as related to other phenomena. In other words, systems thinking requires consideration of connections. There is an old saying that "the biggest cause of problems is solutions," which is important to keep in mind when analyzing sustainability issues. Examples of unintended (sustainability) consequences abound. For example:
- The so-called Green Revolution instituted in Pakistan and India in the 1960's and 1970's probably saved [70] millions, or even hundreds of millions of lives, but has also contributed to [71] soil loss, debt, and farmer suicides due to the unsustainable farming practices it uses.
- Forest fire prevention and suppression in the U.S. has led to more severe forest fires (an example of a Penn State led study [72] can be found on Penn State News, December 17, 2019).) As it turns out, low-grade forest fires naturally reduce understory fuel sources (shrubs, fallen branches, etc.), which help prevent more intense fires from occurring.
- Many invasive species were purposefully introduced by humans, only to inflict lasting damage on native plant and/or animal populations. Kudzu [73]is a vining plant that has proved to be a major menace wherever it grows in the U.S., yet was promoted first as an ornamental plant, then a tool for preventing soil erosion. Cane toads were released [74] into Australia in the 1930's in an effort to control the beetle population. Not only have they not controlled beetles, but they are now major nuisances to humans and native species and habitats.
From a sustainability perspective, systems thinking means that you should at least always a) consider the short- and long-term impacts of actions, both in space and time, and b) consider the possible causes of issues. It is unwise to address a problem or situation without thinking about the possible causes and consequences. More on this below.
The Three E's of Sustainability
The EPA offers a definition of sustainability that encompasses a lot of the concepts described above: "To pursue sustainability is to create and maintain the conditions under which humans and nature can exist in productive harmony to support present and future generations" (source: US EPA [68]). Note that this definition changed slightly in early 2017. It used to be: "Sustainability creates and maintains the conditions under which humans and nature can exist in productive harmony, that permit fulfilling the social, economic and other requirements of present and future generations." Read into that change what you will. This is a more thorough definition than the Brundtland Commission's and provides a more actionable list of goals. (Though it should be noted that there is still a lot of room for debate on how to achieve them or what they really mean.) It also brings to mind what is commonly referred to as the "three E's of sustainability."Optional Reading
Box 2.1 on pp. 7 - 8 of the document below provides a helpful primer to the three E's (3 E's). This is a chapter from The Post Carbon Reader, an edited volume by Post Carbon Institute [76]. You are welcome to read the rest of the chapter as well.
"What is Sustainability," p. 7 - 8 [77] by Dillard, Dujon, and King.
Sustainability and sustainable development are often thought of as having three core components: environment, economy, and equity. These are commonly referred to as the "3 E's" of sustainability. The 3 E's are a useful way to provide an analytical framework for sustainability. This 3E framework is useful because it provides questions that can be asked when investigating whether or not something is sustainable. While even these terms can be defined in various ways, we will use the following definitions from the reading when analyzing the sustainability implications of something:
- Is it "environmentally sustainable, or viable over the very long term"? (environment)
- Is it "economically sustainable, maintaining [and/or improving] living standards over the long term"? (economy)
- Is it "socially sustainable [and just], now and in the future"? I would add "Are the benefits and burdens shared be everyone equally?" (social equity)
As Dillard and Dujan note, if a business is attempting to address these criteria, it is often called the triple bottom line. If it meets all three criteria, and will likely continue to do so into the foreseeable future, then that is a pretty strong case for sustainability.
The details of how to maintain environmental sustainability are not without controversy, but at some point, we will have to maintain a steady-state of natural resources if we are to survive (this will be addressed later). As Engelman and others say, this may come at the expense of quality of life for some/many people now. No one said it will be easy.
But through my own personal experience and the experience of others, it is clear that social equity is the most confusing of these concepts. Dillard, Dujon, and King do a good job of outlining what it means. Contrary to what some believe, equity does not mean equal distribution of resources. There will always be inequality, whether we want it or not. What it does refer to is the fairness of opportunity and access to resources like education, health care, a clean environment, political participation, social standing, food, shelter, and others. In a socially equitable society, everyone has reasonable access to things that provide a good quality of life. Social equity is about equality of opportunity. Whether or not they take advantage of this opportunity is another story. There is an important difference between being uneducated because of laziness and because of a lack of access to good schools. Making this happen is easier said than done, but the distinction is important to make.
One reason that addressing equity can be controversial is illustrated in the image below. What do you think it is?

As indicated in the caption, equity often requires providing more resources to those that are at some disadvantage. Why they are disadvantaged, who decides they deserve help, the amount of help they are given, and more aspects can be controversial. Which is understandable, given that individual and group circumstances are rarely black and white and oftentimes public resources such as tax dollars are involved. Generally, those that advocate for equity err on the side of "too much" equity rather than "too little."
Economy can also be a point of confusion. It is very important to keep in mind that "economy" from a 3E perspective does not refer to just having and/or making money. It refers both to engaging in actions that are economically sustainable (if businesses do not make enough money to continue, they will not be in business for long) and having enough money to provide and maintain& a reasonably high quality of life over the long term. Yes, money is often an important - if not the most important - factor in achieving a high quality of life, particularly at lower income levels. But please keep in mind as we move forward that, from a sustainability perspective, the true "economic" goal is quality of life, not high income. Money often does contribute to a high(er) quality of life, but not always, as we will see later. Money is a means to an end. For sustainability purposes, that economic "end" is providing adequate living standards for people now and in the future. (After all, if you are incredibly happy, healthy, safe, and have everything you need, does it matter if you do not have a lot of money? More on this later.)
Food for Thought
Engelman's chapter brings up some very tough questions that (probably) need to be answered if we are going to achieve a sustainable world. I would like you to think about these moving forward this semester:
- Is there any way to know how much sacrifice is needed now to allow for future generations' quality of life?
- How can considerations for future generations be successfully integrated into today's actions?
- Is there a way to account for this economically? If so, what kind of controversies would it cause?
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Energy, Sustainability, and Society
Learning Objectives Self-Check
Read through the following statements/questions. You should be able to answer all of these after reading through the content on this page. After going through the content, check the boxes next to the questions/statements that you feel at least somewhat confident answering. I suggest writing or typing out your answers, but if nothing else, say them out loud to yourself. This is to help you reflect on important content, and will help you prepare for this week's quiz. It will also help lay the foundation for future course content.
It may be helpful to summarize some of the key points from the previous page (though more will be addressed in this week's homework questions):
- Sustainability/sustainable development has no single definition, but the most commonly cited one is by the Brundtland Commission ("meeting the needs of the current generation without compromising the ability of future generations to meet their needs").
- We must consider quality of life when addressing sustainability and may need to make some sacrifices to achieve abroad-based quality of life.
- We must utilize systems thinking when addressing sustainability problems, recognizing that actions taken will likely have far-reaching consequences.
- If we are to achieve sustainability, we must at least consider the environmental, economic, and social equity impacts on current and future generations.
- In order to know how well we are achieving sustainability, we must find a way to measure it or at least find ways to know if things are (not) going well.
Sustainability is some heavy, complex stuff! Most would argue that the future of civilization depends on how we address sustainability, starting yesterday <raising hand>. As Asher Miller phrases it in his introduction to The Post Carbon Reader [80], "The success or failure of the human experiment may well be judged by how we manage the next ten to twenty years" (p. xv). (For better or worse, and unfortunately I'd say "worse," that was written about 10 years ago.) Sustainability is a very important topic, but it is an even more complex and broad topic than energy. I don't expect you to be an expert (yet), but I hope that this course helps you think critically about sustainability-related and other claims.
Energy, Sustainability, and Society
I have a challenge for you: think of something that you did in the past week that did not involve energy.
Okay, so that's not really a fair challenge. Everything we do, even thinking about things that we might do, require energy. Here's a more reasonable challenge: think of something that you did in the past week that did not involve the use of non-renewable energy.
Any food you eat almost certainly required non-renewable energy. There are obvious connections like farm machinery, artificial fertilizers, and herbicides, transporting food, refrigerating food, cooking food, and packaging food. But even if you grow your own [81], you likely used a tool or fencing that was manufactured using non-renewables, seeds that were processed and shipped with fossil fuel-using machines, packaging that was made using non-renewable energy, or maybe even plastic row markers made with petroleum-based plastics. Almost all transportation uses non-renewables, most businesses run on non-renewable energy sources (either directly or indirectly through electricity generation), almost all of the products you buy contain materials either made of or that are processed with fossil fuels. The electronic device you are looking at right now is partially made of and manufactured using fossil fuels. In short, modern society is very dependent upon access to non-renewable energy, particularly fossil fuels. As Asher Miller notes in The Post Carbon Reader:
Look around and you'll see that the very fabric of our lives - where we live, what we eat, how we move, what we buy, what we do, and what we value - was woven with cheap, abundant energy. (p. xiv)
Watch the video below for an interesting 5-minute journey through the last 300 years of fossil fuels in society.
It all started with a big bang. Wait, we don't have to go back that far. The earth was formed 4.5 billion years ago. No, still too far. Try this. It's the middle ages. People in Britain run out of firewood. They start burning coal, but they use up the coal on the ground. Miners dig deep, Coal mines fill with water. Samuel Newcomen invents a coal-burning steam engine to pump out water so miners can keep digging. James Watt makes it practical for other uses. Now we have ingredients for the industrial revolution. Fossil fuels and a way to put them to work. All hell breaks loose. Coal miners bog down lugging coal. Rails make it easier rails and with steam engines combined make a railroad. Michael Faraday makes the first electric motor. Nicola Tesla invents alternating current. Soon, utility companies start burning coal to generate electricity. Meanwhile, Edwin Drake drills the first rock oil well in Pennsylvania and Gottlieb Daimler builds an automobile running on petroleum. Coal tar and oil are turned into industrial chemicals and pharmaceuticals that prolong life. More population growth. The Wright brothers start oil fueled Aviation. Fritz Haber and Carl Bosch make fertilizer from fossil fuels. Fertilizer and oil-powered tractors expand food production, feeding more people. World War One is the first fossil fueled conflict. Then comes World War Two, giving us guided missiles and atom bombs. In between is a great depression partly caused by overproduction. Powered assembly lines make products faster than people need them. Advertising executives invent consumerism to soak up overproduction. It's the 1950s. Advertisers use television to hook new generations of consumers. In the 70s, there's an oil shock. Everyone's shocked to realize how dependent they are on oil. With the energy crisis, the environmental movement is born. But oil prices fall, and everyone forgets energy shortages. There's a showdown between market and planned economies. Market wins. Goodbye evil Soviet empire. Politicians decide the market will solve everything. Personal computers arrive.
Globalization takes over when the market notices labor is cheaper in China. Suddenly everyone has a cell phone, but world oil production stalls out. China's now burning half the world's coal to make export products, but where will China get more coal and oil to fuel more growth? Environmental problems everywhere. Rising CO2 levels lead to record heat waves, floods, droughts. Oceans acidify. Topsoil erodes by 25 billion tons a year from industrial agriculture. Ancient forests disappear. Species go extinct at a thousand times normal rates. Freshwater is scarce or polluted. Oil companies drill in miles of sea water because the easy oil is gone, but a deep water oil platform explodes and fouls the Gulf of Mexico. Manufacturing moves to polluting countries where labor is cheap, while the U.S. becomes a casino. The financial sector is forty percent of the economy. But wall street is over leveraged. Banks fail, unemployment soars, credit evaporates. The economy is on the verge of collapse! Okay. Present time. It's amazing how far we've come in 200 years. Just three human lifetimes from the beginning of industrialism until now. But where are we headed? We can't keep doubling human population. We can't keep dumping carbon in the atmosphere. We can't keep ruining topsoil. We can't keep growing population and consumption or basing our economy on depleting fossil fuels. We can't just print more money to solve the debt crisis. It's been an exhilarating ride but there are limits. Now, it's not the end of the world, but we have to do four things fast: Learn to live without fossil fuels. Adapt to the end of economic growth as we've known it. Support seven billion humans and stabilize population at a sustainable level. And deal with our legacy of environmental destruction. In short, we have to live within nature's budget of renewable resources at rates of natural replenishment. Can we do it? We have no choice. Alternative energy sources are important, but none can fully replace fossil fuels in the time we have. Also, we've designed and built our infrastructure for transport, electricity, and farming to suit oil, coal, and gas. Changing to different energy sources will require us to redesign cities, manufacturing processes, health care and more. We'll also have to rethink some of our cultural values. None of our global problems can be tackled in isolation, and many cannot be fully solved. We'll have to prepare for business as unusual. Our best goal is resilience: The ability to absorb shocks and keep going. If we do nothing we still get to a post carbon future, but it will be bleak. However, if we plan the transition we can have a world that supports robust communities of healthy creative people and ecosystems with millions of other species. One way or the other we are in for the ride of a lifetime. Understand the issues and pitch in. It's all hands on deck!
The charts below provide rather dramatic evidence of how important non-renewable energy is to the U.S. All charts are from the EIA's Annual Energy Outlook (AEO) series, which are published on a yearly basis. I have provided a series of charts to provide some indication of how difficult it is to predict future trends. But, these serve as official (and generally pretty accurate) guides to future energy use.
The first chart is from the 2015 version of the AEO. Though a bit outdated, I put it here because the chart style makes it very easy to see the dominance of non-renewable energy sources. The second chart is from a more recent report (2019) that has total energy consumption, and the third from the most recent (2022) report. The second and third charts are obviously more recent, but is not quite as easy to interpret. Another nice feature of these charts is that they include both historical use and projected future use.
Any way you slice it, the charts make clear that non-renewable energy - particularly fossil fuels - have played and will continue to play a dominant role in society. At this point, our society simply cannot function at its current capacity without them.
Another aspect worth noting is that aside from recessions (e.g., early 1980's and 2007-8), energy use continues to increase over time. Despite consistent increases in energy efficiency, the U.S. can't seem to level off, never mind reduce overall consumption. This is also something that will have to be addressed if we are going to have a sustainable energy future.
Finally, Figure 1.15 shows which energy sources are most responsible for carbon dioxide emissions in the U.S. Oil i the current leader, but as more and more natural gas is used (particularly to generate electricity), it will likely come close to catching up to oil-based emissions by 2050, according to the EIA.
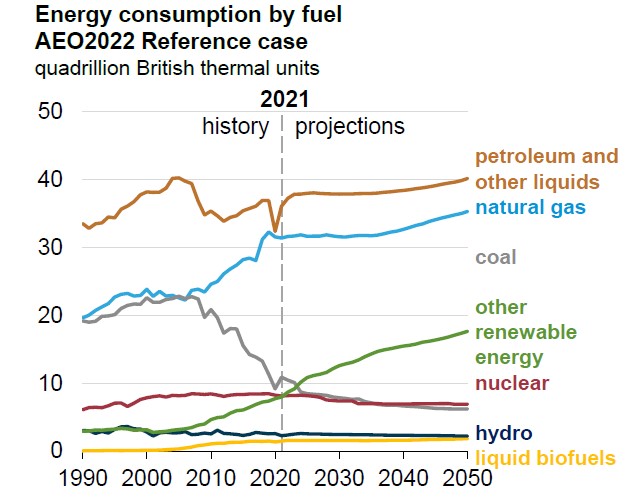

Non-renewable energy is extremely useful - it has played an essential role in human society developing to the point that it has. It is energy dense, generally easy to transport and control, and is used for a variety of purposes. Non-renewable energy will continue to play a starring role, for at least the short term future. I enjoy the freedom of the open road in my car [88]. I like to have a house in which I have some control over the temperature and humidity. I like to buy new things from time to time. I enjoy the occasional air travel. I eat food that was shipped from countries on the other side of the world. If we are all to enjoy such things (and more) in the way society and our economy is currently structured, we need access at least to fossil fuels. But given our understanding of the nature of sustainability and non-renewable energy, this cannot go on forever. In fact, it will probably need to change dramatically within the next 10-15 years.
If nothing else, since non-renewable energy is finite, we will reach limits at some point in the future - exactly when is open to debate. But even before that eventuality, it is becoming apparent that the results of unsustainable energy (and resource) use is making it difficult for current generations to meet their needs, never mind future generations. The topics in the next lessons illustrate some of the reasons that scientists and others are worried about the sustainability of our society, some of which are directly related to energy, others not.
Food for Thought
Richard Heinberg mentions four things that must be done to achieve a sustainable society with an adequate quality of life. Think about how difficult each of these is. Which do you think is the most difficult to achieve? Do you think they are even feasible? Can you envision a society that achieves these, and if so, is it good or bad? I don't have the answers (I wish I did!), but I think they are important questions to ask. Heinberg is not alone in thinking these are important.
- "Learn to live without fossil fuels"
- "Adapt to the end of economic growth as we’ve known it"
- "Support 7 billion humans and stabilize population"
- "Deal with our legacy of environmental destruction"
Optional (But Strongly Suggested)
Now that you have completed the content, I suggest going through the Learning Objectives Self-Check list at the top of the page.
Summary and Final Tasks
All right, that does it for the content for this week. Before you relax, make sure you complete the assignments listed at the beginning of this lesson.
This week, we went over some of the core considerations for energy and sustainability.
You should be able to do the following. The Lesson 1 quiz will help you solidify these skills:
- define energy, energy efficiency, and the First Law of Thermodynamics;
- identify and describe types of energy and energy conversions:
- identify and define fossil fuels, non-renewable energy sources, and renewable energy sources, and their origins and characteristics;
- analyze the energy data provided in charts and graphs;
- identify reliable sources of energy data;
- evaluate the implications of sustainability definitions; and
- define the "3 E's" of sustainability and use the 3E framework to evaluate the sustainability of given actions.
The Language of Energy and Sustainability
At the end of each lesson, I will provide a list of all of the key terms from the lesson. These terms are easy to find because most of them are in bold throughout the lesson, or appear in headings. This is designed to help you review the content, both before you take the quiz, and later. Many of these terms will be used in other parts of the course, in future courses in the Energy and Sustainability Policy curriculum, and in the sustainability and energy literature. They are mostly listed in the order they appear in the text.
- Energy, work
- Kinetic energy, potential energy, electromagnetic energy, sound energy, radiant energy, mechanical energy, electrical energy, chemical energy, gravitational energy, nuclear energy, thermal energy, First Law of Thermodynamics
- Coal, oil, natural gas, nuclear, fossil fuels, hydrocarbons
- British Thermal Unit, Btu, Joule, kilowatt hour (kWh), 100 cubic feet (ccf), quads, national labs, energy sources, end-use sectors, Sankey diagram
- Energy efficiency, Second Law of Thermodynamics, the "fifth fuel"
- Renewable energy, non-renewable energy, solar photovoltaics, wind turbines, hydropower, biomass, biofuels, primary energy, carbon free, carbon neutral
- Energy Information Administration (EIA), International Energy Agency (IEA)
- Sustainability, sustainable development, Brundtland Commission, ecocentric, anthropocentric, 3 E's, environment, intergenerational equity, equity, economy, triple bottom line, sustainability indicators/metrics
Time for the Lesson 1 Quiz!
This quiz is based on the material this week. Unless otherwise indicated, all of the answers to the quiz questions are in the required readings, videos, and website text from this week. You have unlimited time to take this quiz, but it must be completed by the due date to receive credit. You get one try. Refer to one of the course calendars for due date. Note that you can start the quiz and save your progress, and pick up where you left off later. The quiz answers automatically save as you complete the quiz, and if you stop taking it you will pick up right where you left off if you start again. If the quiz is partially completed, it will automatically submit at the due date/time if you do not submit it yourself. Please note that students in the past have had some issues saving and resuming with Internet Explorer. I suggest using Firefox or Google Chrome. To take the quiz:
- Go to the Modules tab.
- Locate the Lesson 1 Module.
- Click on Lesson 1 Content Quiz.
- Begin!
- When you are done, click "Submit." You cannot undo a submission once it is submitted.
Don't forget to start commenting on the Yellowdig discussion board!
