Deasphalting Process
Deasphalting Process
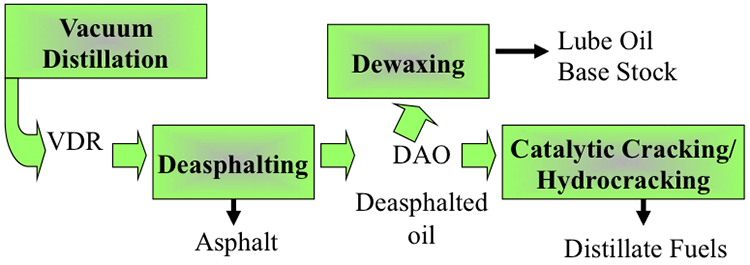
Figure 5.8 places the deasphalting process in a refinery flow scheme as an intermediate process between vacuum distillation and dewaxing processes, for producing the refinery output streams as asphalt deasphalted oil (DAO), which can be directed to another separation process, dewaxing, to produce lubricating oil base stock and wax, or can be sent to conversion units such as hydrocracking to produce light and middle distillate fuels. It is important to note that the deasphalting process is an upgrading process to transform VDR into marketable products, and/or convert it to distillate fuels that command high demands.
The two objectives of the deasphalting process are:
- Produce asphalt - as final product.
- Remove asphaltenes to prevent coke, or metal buildup on catalyst in further processing of DAO.
Depending on the properties of the VDR and prevailing markets, the emphasis could be placed on one of these objectives. Remember that aromatic asphaltic crudes are more expensive to convert into distillate fuels. Such crudes could be processed readily into making high yields of asphalt and serve the asphalt market. With lighter crudes, the principal focus could be on removing the asphaltenes from VDR so that DAO produced can be used in conversion processes with a lower extent of problems caused by asphaltenes such as coke buildup, or metals buildup on catalysts in, for example, hydrotreating or hydrocracking reactions.
Simplified Flow Diagram of a Deasphalting Process
Simplified Flow Diagram of a Deasphalting Process
Figure 5.9 shows a simplified flow diagram of a propane deasphalting process. See a more detailed flow diagram in your textbook, along with a description of the operating conditions in the commercial process. As the first step in deasphalting, residue (feedstock) is mixed with four to eight times the volume of liquid propane. Heavier residua require a higher solvent to residue (S/R ) ratio for effective separation of asphalt. Following the precipitation of asphalt, DAO, and asphalt are separated, and each stream is purified and flashed to recover and recycle the propane solvent, as shown in Figure 5.9.
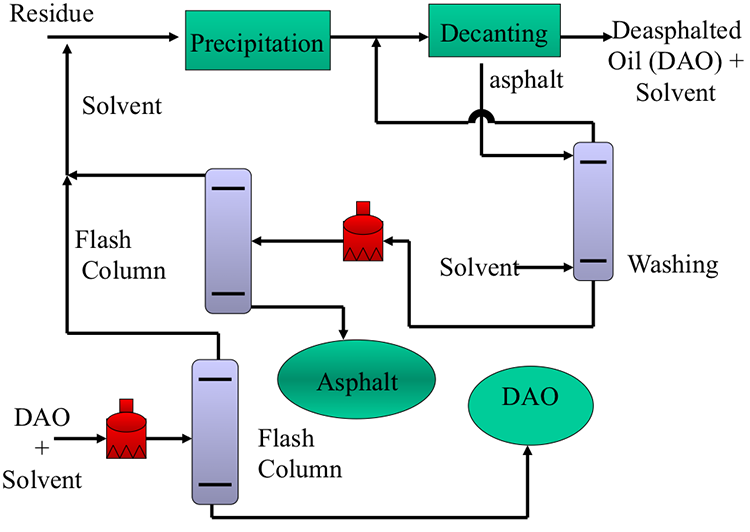
The four-unit operations of deasphalting illustrated in the process flow diagram are:
- Flocculation (& precipitation) of asphaltenes
- Asphalt decanting - separation
- Asphalt washing - to remove entrained oil
- Solvent recovery and recycle
[1] Petroleum Refining, by J. H. Gary, G. E. Handwerk, M. J. Kaiser, 5th Edition, CRC Press NY, 2007, Chapter 15, pp.311-312.
Asphaltene Yield
Asphaltene Yield
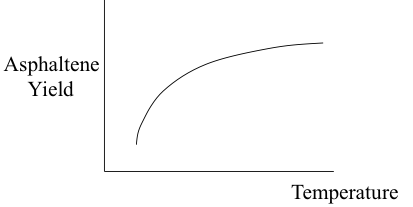
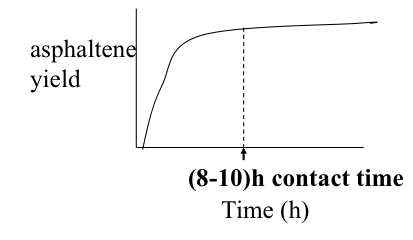
Independent variables in the deasphalting process include the solvent used, pressure, temperature, S/R, and contact time. These variables can be controlled to obtain the optimum conditions for the desired separation in deasphalting. One of the important dependent variables in the process is the asphaltene yield. Figure 5.10 shows, in qualitative plots, how asphaltene yield varies as a function of the process parameters.
Solubility Parameter of the Solvent
Solubility Parameter of the Solvent
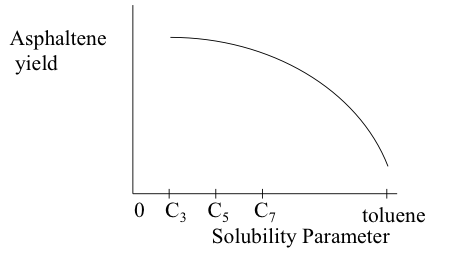
Solubility parameter of the solvent is a key a variable that affects asphaltene yield. Asphaltene is completely soluble in toluene, which has a high solubility parameter. Therefore, the asphaltene yield with toluene will be zero (Figure 5.10a). Propane (C3,) which has the lowest solubility parameter among the solvents given in the plot, will give the highest asphaltene yield.
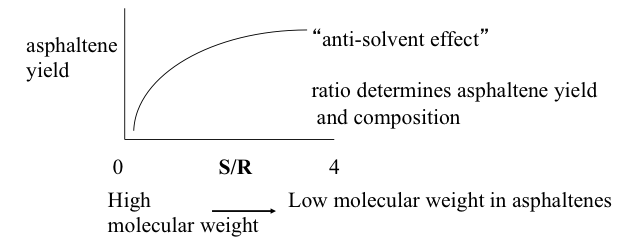
Figure 5.10b shows that asphaltene yield increases with the increasing solvent/residue ratio used in the process. This is an unusual behavior for solvent extraction because, typically, the yield of an insoluble fraction would decrease with the increasing quantity of solvent used in the extraction behavior. The unusual behavior seen in Fig. 5.10b is termed as the “anti-solvent effect.” This relates to the gradient theory of asphaltene solubility in crude oil (or in VDR) such that larger quantities of paraffin solvent used in the process more effectively disrupt the gradient solubility by removing more oils from VDR, forcing the asphalt fraction (asphaltene+resin) to separate out. One should point out here that the molecular composition of the asphaltenes would vary significantly along with the yield of asphaltenes in the plot shown in Figure 5.10b. As the yield of asphaltenes increases with the increasing S/R, lower molecular weight asphaltenes would be progressively included in the separated asphaltenes. In other words, for a given process, low yields of asphaltenes (obtained at low S/R) would contain the highest molecular weight and highly aromatic compounds, whereas asphaltenes obtained at high yield with high SR ratio would include a broader range of molecular weight and aromaticity in the separated asphaltenes.
The consideration of variable asphaltene composition would also apply to the asphaltenes separated with different paraffin solvents, as shown in Figure 5.4b. In this case, lower yields of asphaltenes obtained with C7 (n-heptane) will be associated with heavier and more aromatic asphaltenes than those obtained with propane (C3). These observations relate to the absence of a precise molecular definition to express the complex of asphaltenes, but the need to use an operational definition on the basis of solubility/insolubility in a given solvent. It should, therefore, be remembered that the composition of the asphaltenes, or asphalt produced in a refinery would not only depend on the properties and composition of the parent crude oil, but also on the solvent, as well as the S/R ratio used in the process among other factors, such as temperature and pressure as they affect the solubility parameters of the solvent. Figure 5.10c shows the dependence of asphaltene yield on temperature, as explained by the decreasing solubility parameter of the solvent used in this case with the increasing temperature. A sufficient amount of contact time with paraffin is necessary for good asphaltene separation. The 8-10 h contact time refers to batch experiments in the laboratory. In flow systems of commercial deasphalting, the contact time necessary for the desired separation of asphalt is much shorter.