Product Blending
Product Blending
Product blending plays a key role in preparing the refinery products for the market to satisfy the product specifications and environmental regulations. The objective of product blending is to assign all available blend components to satisfy the product demand and specifications to minimize cost and maximize overall profit [5]. Almost all refinery products are blended for the optimal use of all of the intermediate product streams for the most efficient and profitable conversion of petroleum to marketable products. For example, typical motor gasolines may consist of straight-run naphtha from distillation, crackate (from FCC), reformate, alkylate, isomerate, and polymerate, in proportions to make the desired grades of gasoline and the specifications.
Basic intermediate streams can be blended into different finished products. For example, naphthas can be blended into gasoline, or jet fuel streams, depending on the demand. Until the 1960s, the blending was performed in batch operations. With computerization and the availability of the required equipment, online blending operations have replaced blending in batch processes. Keeping inventories of the blending stocks along with cost and physical data has increased the flexibility of and profits from online blending through optimization programs. In most cases, the components blend nonlinearly for a given property (e.g., vapor pressure, octane number, cetane number, viscosity, pour point), and correlations and programming are required for reliable predictions of the specified properties in the blends [3].
[3] U.S. Refinery Sulfur Production Capacity [1]
[5] Petroleum Refining, by J. H. Gary, G. E. Handwerk, M. J. Kaiser, 5th Edition, CRC Press NY, 2007, Chapter 12, Product Blending, p.267.
Octane Number Blending
Octane Number Blending
Octane numbers are blended on a volumetric basis using the blending octane numbers of the components. True octane numbers do not blend linearly, thus it is necessary to use blending octane numbers in calculating the octane number of the blend. Blending octane numbers can be estimated from empirical correlations that have been developed over the years. Blending octane numbers, when added on a volumetric average basis, will give the true octane of the blend, as can be obtained from standard test using CFR test engines.
(True Octane Number of a blend) ON = Σ xi∗ ONi
Where xi is the volume fraction of component i in the blend, and ONi is the blending octane number of component i.
For example, if you have
- a light naptha stream with a blending octane number = 75
- a reformate with a blending octane number = 86
and would like to get a gasoline of ON = 83, what would be the volume fraction of reformate (x) in the blend?
- ON = x(86) + (1-x)(75) = 83
- x = 0.73
So, you need 73% by volume of reformate in your blend. Additive concentration may be calculated the same way, and ON for multicomponent blends can be calculated the same way for research or motor octane numbers.
Pour Point Blending
Pour Point Blending
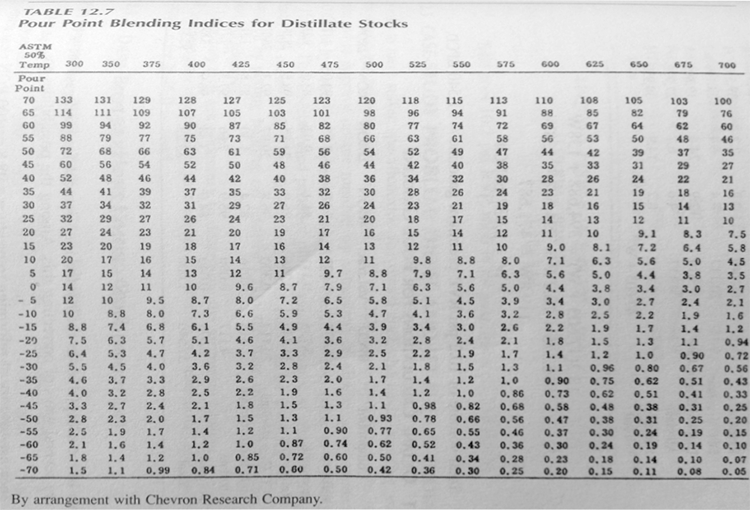
Pour point is an important property for diesel and fuel oil blends. Pour point blending is also non-linear, and pour point blending indices were developed to enable reliable calculation of the pour points of the blends. Pour point blending indices for some distillate fuels are given in Table 12.7 of the textbook, and copied in Figure 9.11, where the blending indices are tabulated as a function of ASTM 50% temperature, °F (first horizontal listing) and Pour Point, °F (first vertical listing). For example, pour point blending index for a distillate that has an ASTM 50% temperature of 500°F and a pour point of 40°F would be 36. This index can be used in calculating the pour point of blends using this distillate as a component.
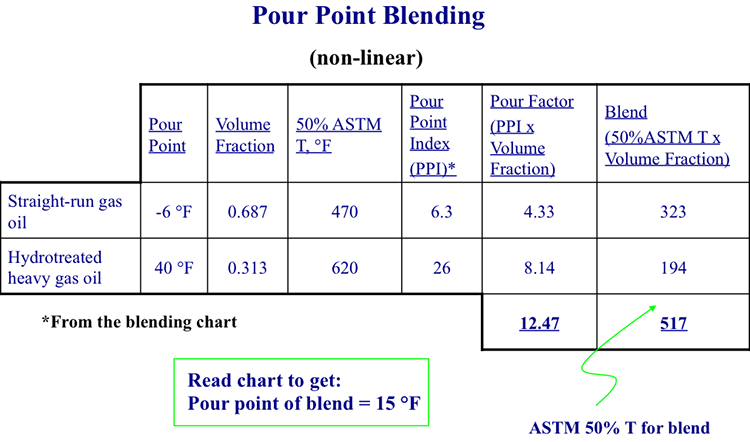
As an example of using blending indices to calculate the pour point of a blend, consider blending a straight-run gas oil (50% ASTM T = 470°F and pour point = -6°F) and a hydrotreated heavy gas oil (50% ASTM T = 620°F and pour point = 40°F). What would be the pour point of a binary blend that consists of 68.7 % vol of straight-run gas oil and 31.3% vol of hydrotreated heavy gas oil?
The procedure used to calculate the pour point of the blend (shown Figure 9.12) can be summarized as follows:
- Read pour point blending indices for the two distillates in the table (PPI column) shown in Figure 9.11, and list it in the table (Figure 9.12). Straight- run gas oil PPBI =6.3 (using double interpolation), and Hydrotreated heavy gas oil PPBI = 26.
- Multiply PPBI for individual distillates with their respective volume fraction to calculate the Pour Factor. For example, the Pour Factor for straight-run gas oil = 0.687 x 6.3 = 4.33. Add the pour factors for each component, to calculate the blending index for the blend = 12.47.
- Calculate blend 50% ASTM temperature (linearly additive) of the blend by multiplying the volume fraction with ASTM 50% T of each component and adding them together (0.687x470 +0.313x620 = 517).
- Using the blend ASTM 50% just calculated (517) and the blending index for the blend from the pour factors (12.47), interpolate/read the pour point of the blend from the table in Figure 9.11, as 15°F.
Note that if you would assume linear addition of the pour points, you would calculate a blend pour point as = 0.687x(-6) +0.313x40 = 8.4°F. A serious underestimation of the pour point! Thinking that your diesel fuel has a pour point 8.4 °F, you may try to start your diesel truck on a 12°F day, to no avail, not knowing that your fuel tank has a gel, and not a liquid that can be easily pumped to the combustion cylinder.
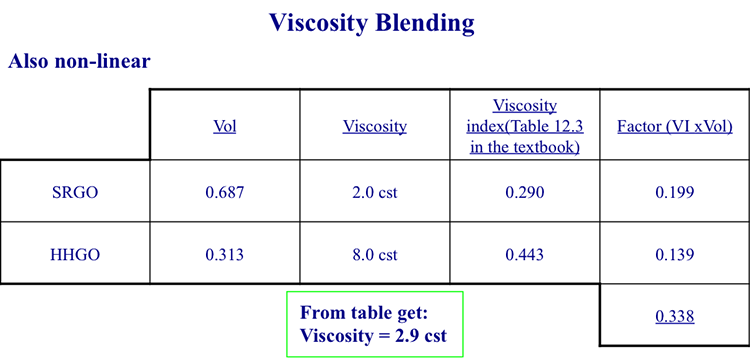
Similar to pour point, the viscosity of blends can also be calculates using blending index numbers, or plots developed for this purpose. For the same blend, we can use the Viscosity Blending Index Numbers in Table 12. 3. of your textbook [6], and the procedure shown in Figure 9.13 to calculate the viscosity of the blend. Try to verify the numbers given in Figure 9.13, and calculate a viscosity of the blend if viscosities were linearly additive to compare with the value calculated in Figure 9.13. Post your questions, if any, and comments in the Help Discussion Forum.
| Pour Point | Volume Fraction |
50% ASTM T, ºF |
Pour Point Index (PPI) |
Pour Factor (PPI x Volume Fraction) |
Blend (50% ASTM T x Volume Fraction |
|
| Straight-run gas oil | -6ºF | 0.687 | 470 | 6.3 | 4.33 | 323 |
| Hydrotreated heavy gas oil | 40ºF | 0.313 | 620 | 26 | 8.14 | 194 |
| Total | - | - | - | - | 12.47 | 517 |
Read chart using this information to get a pour point of 15ºF
| Volume | Viscosity | Viscosity Index (Table 12.3 in the textbook) | Factor (VI x Vol) | |
| SRGO | 0.687 | 2.0 cst | 0.290 | 0.199 |
| HHGO | 0.313 | 8.0 cst | 0.443 | 0.139 |
| Total | - | - | - | 0.338 |
From the table, you get: Viscosity = 2.9 cst
[6] Petroleum Refining, by J. H. Gary, G. E. Handwerk, M. J. Kaiser, 5th Edition, CRC Press NY, 2007, Chapter 12, Product Blending, p.263.