If there is magic on this planet, it is contained in water.
The Immense Journey by Loren Eiseley (1907-1977), anthropologist and author.
Loren Eiseley's quote says much about the amazing attraction of watery environments for humans, and much about the unique physical properties of water, too. The surface tension of liquid water allows water striders to dance across the film. The crystalline structure of ice, solid water, is less dense that of water as a liquid, so those of us in cold, wintry climatic regions can ice skate on the surface of a pond. The mysterious approach of fog, condensing water in a gaseous form, fascinates us in the dim light of dawn or dusk; and in some parts of the world, fog provides the only source of nourishing moisture (e.g., coastal reaches of the Atacama and Namib deserts in Chile and Namibia, respectively, both geographically located in the southern hemisphere near cold ocean currents flowing northward to the equator).
The common occurrence of water on Earth in three distinct physical states – liquid, solid, and gas – is fundamentally important to most life on our Blue Planet, including humans. This simple molecule consisting of two positively-charged hydrogen atoms bonded with one negatively-charged atom of oxygen creates the many forms of water we experience. This type of molecular attraction allows the cohesion of water molecules, whereas another type of chemical bond attraction leads to adhesion, the property that makes water a so-called “universal solvent” capable of dissolving many more substances than other liquids.

Our dependency on water in daily life – making coffee or tea, storing frozen foods in a freezer, negotiating constantly changing weather conditions when we travel – has made us all familiar with the key temperatures at which water freezes (0°C, 32°F) or boils (100°C, 212°F) at the benchmark pressure of sea level. Perhaps less known is the temperature at which water becomes most dense (4°C, 39°F), yielding ice-covered lakes and ponds, floating icebergs and iceflows, and even floating ice cubes in a chilled beverage. Another confounding, but influential, characteristic is water's high specific heat whereby much heat is absorbed before the temperature rises, and the slow release of that absorbed heat to the surrounding environment. Those dwelling in coastal regions or by large lakes will recognize this “marine or coastal influence” that moderates temperature fluctuations on adjacent lands.
In Lessons 4 and 5, we will delve further into how aquatic ecosystems are classified, and where and why they are distributed unevenly around the planet, respectively. As can be seen in Figure 1.2, freshwater comprises a small proportion, less than 3%, of all water on Earth -- the great majority of that, about 69% of the freshwater proportion, existing as a solid in ice caps, glaciers, and permafrost. About 30% of the freshwater occurs as liquid groundwater. Only 1% of the total amount of freshwater exists as a liquid in our rivers, lakes, and wetlands, soil moisture, and living organisms, leaving only a tiny bit in gaseous form as part of the atmosphere.
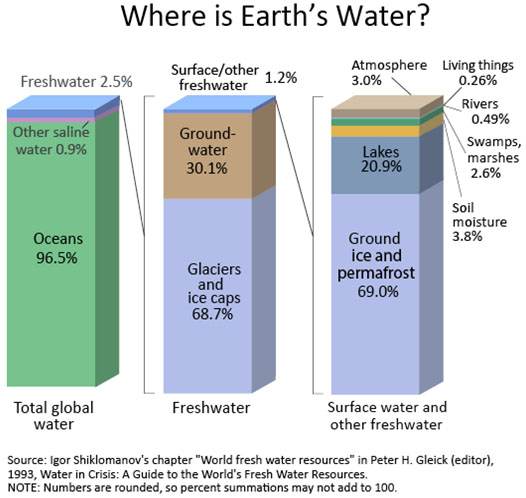
Total global water
-
Oceans 96.5%
-
Other saline water 0.9%
-
Freshwater 2.5%
-
Glaciers and ice caps 68.7%
-
Groundwater 30.1%
-
Surface/other freshwater 1.2%
-
Ground ice and permafrost 69.0%
-
Lakes 20.9% Soil moisture 3.8%
-
Atmosphere 3.0%
-
Swamps, marshes 2.6%
-
Rivers 0.49%
-
Living things 0.26%
-
-
“Not man apart” – a quotation from John Robinson Jeffers (1887-1962), an American poet. This brief expression is taken from his poem, The Answer (1936), and was adopted by environmental leaders and organizations, particularly the Sierra Club and Friends of the Earth in the 1960s and 1970s, as a way to stimulate action on environmental issues. David Brower, a founder of the Sierra Club and Friends of the Earth, published a Sierra Club book of photographs and Jeffers' quotes about Big Sur along the California coast. It makes the point that “we” have been dependent upon water throughout our existence. At a minimum, we use the oceans for food, transportation, and recreation, but our links to freshwater are essential.
From the beginning of time, animals, including humans, that depend on freshwater have searched for, gathered by, and managed water resources for their benefit. These primary ecosystem services, as we now refer to them, involved drinking, bathing, cooking, and irrigating crops, for our basic needs. Over time, as the principles of physical science and engineering knowledge have accumulated, management and manipulation of water resources by human societies have become nothing short of amazing. In this exercise, we will investigate and share examples of how humans have supplied freshwater to meet their basic needs. Each student gets to choose the time period and the specific example to describe, thus providing all of us with a series of relevant, successful (or perhaps unsuccessful) stories of how cultures around the world and through time have managed water resources.