Lesson 1: What Is Fresh Water?
The links below provide an outline of the material for this lesson. Be sure to carefully read through the entire lesson before returning to Canvas to submit your assignments.
Overview
This lesson will give you an overview of the physical and chemical properties of water, and some insight into how those properties define aquatic ecosystems and influence society's use of water. Read through Lesson 1 material, then review the Lecture 1.1 Microsoft PowerPoint. This Microsoft PowerPoint provides an introduction to Lesson 1 and the overall course. It includes definitions, a brief course outline, an introduction of water uses through time, and some visual impressions of water in art and aquatic ecosystems.
Learning Outcomes
After completing this lesson, you should be able to:
- compare the physical and chemical properties of water in three different phases;
- explain how the physical and chemical properties of water define aquatic ecosystems;
- identify how the properties of water influence society's use of water.
Lesson Roadmap
This lesson is one week in length. Please refer to the Course Calendar in Canvas for specific time frames and due dates. To finish this lesson, you must complete the activities listed below.
| Requirements | Assignment Details | Access/Directions |
|---|---|---|
| To Watch |
Watch/Read through the Lecture 1.1 - Introduction PowerPoint file. |
Registered students can access the Microsoft PowerPoint file under Lesson 1 in Canvas. |
| To Read |
|
|
| To Do |
Complete and submit your synopsis of a historic use of water (2 pages). |
Submit your synopsis in the Assignment 1.1 - What is freshwater? dropbox under the Lesson 1 module in Canvas. |
Questions?
If you have any questions, please post them to our Questions? discussion forum located under Orientation and Resources in Canvas. While you are there, feel free to post your own responses if you, too, are able to help out a classmate.
Background
If there is magic on this planet, it is contained in water.
The Immense Journey by Loren Eiseley (1907-1977), anthropologist and author.
Loren Eiseley's quote says much about the amazing attraction of watery environments for humans, and much about the unique physical properties of water, too. The surface tension of liquid water allows water striders to dance across the film. The crystalline structure of ice, solid water, is less dense that of water as a liquid, so those of us in cold, wintry climatic regions can ice skate on the surface of a pond. The mysterious approach of fog, condensing water in a gaseous form, fascinates us in the dim light of dawn or dusk; and in some parts of the world, fog provides the only source of nourishing moisture (e.g., coastal reaches of the Atacama and Namib deserts in Chile and Namibia, respectively, both geographically located in the southern hemisphere near cold ocean currents flowing northward to the equator).
The common occurrence of water on Earth in three distinct physical states – liquid, solid, and gas – is fundamentally important to most life on our Blue Planet, including humans. This simple molecule consisting of two positively-charged hydrogen atoms bonded with one negatively-charged atom of oxygen creates the many forms of water we experience. This type of molecular attraction allows the cohesion of water molecules, whereas another type of chemical bond attraction leads to adhesion, the property that makes water a so-called “universal solvent” capable of dissolving many more substances than other liquids.

Our dependency on water in daily life – making coffee or tea, storing frozen foods in a freezer, negotiating constantly changing weather conditions when we travel – has made us all familiar with the key temperatures at which water freezes (0°C, 32°F) or boils (100°C, 212°F) at the benchmark pressure of sea level. Perhaps less known is the temperature at which water becomes most dense (4°C, 39°F), yielding ice-covered lakes and ponds, floating icebergs and iceflows, and even floating ice cubes in a chilled beverage. Another confounding, but influential, characteristic is water's high specific heat whereby much heat is absorbed before the temperature rises, and the slow release of that absorbed heat to the surrounding environment. Those dwelling in coastal regions or by large lakes will recognize this “marine or coastal influence” that moderates temperature fluctuations on adjacent lands.
In Lessons 4 and 5, we will delve further into how aquatic ecosystems are classified, and where and why they are distributed unevenly around the planet, respectively. As can be seen in Figure 1.2, freshwater comprises a small proportion, less than 3%, of all water on Earth -- the great majority of that, about 69% of the freshwater proportion, existing as a solid in ice caps, glaciers, and permafrost. About 30% of the freshwater occurs as liquid groundwater. Only 1% of the total amount of freshwater exists as a liquid in our rivers, lakes, and wetlands, soil moisture, and living organisms, leaving only a tiny bit in gaseous form as part of the atmosphere.
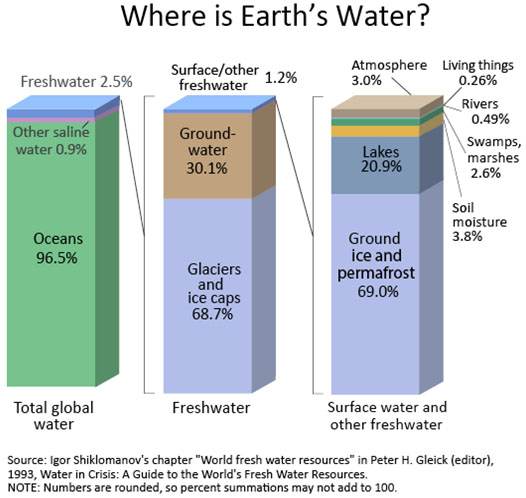
Total global water
-
Oceans 96.5%
-
Other saline water 0.9%
-
Freshwater 2.5%
-
Glaciers and ice caps 68.7%
-
Groundwater 30.1%
-
Surface/other freshwater 1.2%
-
Ground ice and permafrost 69.0%
-
Lakes 20.9% Soil moisture 3.8%
-
Atmosphere 3.0%
-
Swamps, marshes 2.6%
-
Rivers 0.49%
-
Living things 0.26%
-
-
“Not man apart” – a quotation from John Robinson Jeffers (1887-1962), an American poet. This brief expression is taken from his poem, The Answer (1936), and was adopted by environmental leaders and organizations, particularly the Sierra Club and Friends of the Earth in the 1960s and 1970s, as a way to stimulate action on environmental issues. David Brower, a founder of the Sierra Club and Friends of the Earth, published a Sierra Club book of photographs and Jeffers' quotes about Big Sur along the California coast. It makes the point that “we” have been dependent upon water throughout our existence. At a minimum, we use the oceans for food, transportation, and recreation, but our links to freshwater are essential.
From the beginning of time, animals, including humans, that depend on freshwater have searched for, gathered by, and managed water resources for their benefit. These primary ecosystem services, as we now refer to them, involved drinking, bathing, cooking, and irrigating crops, for our basic needs. Over time, as the principles of physical science and engineering knowledge have accumulated, management and manipulation of water resources by human societies have become nothing short of amazing. In this exercise, we will investigate and share examples of how humans have supplied freshwater to meet their basic needs. Each student gets to choose the time period and the specific example to describe, thus providing all of us with a series of relevant, successful (or perhaps unsuccessful) stories of how cultures around the world and through time have managed water resources.
Readings
Now that you have read the background material for the lesson, you should watch my Lesson 1 Microsoft PowerPoint, read Chapter 1 from the course textbook, and familiarize yourself with the material on the USGS Water Science website.
The Lecture 1.1 Microsoft PowerPoint is designed to give an overview of water resources and review topics/assignments for GEOG 431. We begin with What is water? and Where is water? using a variety of images to supplement Chapter 1 of the text. The latter sections cover uses of water - past and present - as a way to stimulate your thinking about which historic period you'd like to research for Assignment 1.1. We finish with water themes in art, and a few of my photographic images on water and aquatic ecosystems.
Required Viewing
GEOG 431 Lecture 1.1 What is freshwater? The Pennsylvania State University, University Park, PA, USA.
Registered students can access the GEOG 431 Lesson 1 Lecture 1.1 PowerPoint file located under Lesson 1 in Canvas.
Note: The PowerPoint files for this course are not annotated voice-over video lectures. They are a series of slides that contain important information to supplement the readings.
The first reading is Chapter 1 from the Holden text, where he describes the physical and chemical properties of water in more detail than the Lecture 1.1, and introduces us to water issues throughout the world. A few new terms are introduced here - they are highlighted - but many more will be covered in Lesson 2 and Chapter 2 of the text.
Required Reading
Chapter 1 – Water Basics (p.1-19)
This reading is from the required course text (Holden 2020).
The U.S. Geological Survey is the federal agency in the USA most responsible for understanding and assessing water quantity. They are a science-based agency with little or no regulatory authority. Their websites and publications offer a wealth of information about the basic characteristics and measurement of water that is applicable to freshwater anywhere is the world. Become familiar with their educational website now, and I'm sure you will revisit it during the course when you either need a bit more information about a term or topic or you are just curious about some aspect of freshwater.
Required Reading
USGS Water Science School website [1] is a tremendous source of information.
Lesson 1, Activity
In the Lecture 1.1 Microsoft PowerPoint, you were introduced to a few achievements and uses of freshwater. The text also covers various historic and current water uses. Your assignment for Lesson 1 is to investigate a historic provisioning of freshwater by earlier civilizations, preferably well before 1800, but absolutely before 1900. Thus, you have a 4,000-year “window” from which to gaze upon the past! Choose a place and time period (e.g., a particular phase in a civilization’s history) and document and synthesize their approach to water management. Prepare a 2-page paper, including graphics and citations about your chosen moment in time. Be sure to adequately cite all resources used, including images and maps.
| Description | Full points | Partial points | No points | Points total | |
|---|---|---|---|---|---|
| Topic selection | Appropriate time period selected (pre-1900) and focused on water management. | Appropriate time period (pre-1900) selected but lacks focus on water management. | Inappropriate time period selected and no acknowledgement of water management. | 5 | |
| Content | Thoroughly provides details on 2+ aspects of water management. Demonstrates an understanding of its role in the civilization’s history. | Talks about 2+ aspects of water management. However, details are vague and do not demonstrate an understanding of its role in the civilization’s history. | Only 1 aspect of water management discussed. However, details are vague and do not demonstrate an understanding of its role in the civilization’s history. | No discussion of water management. | 15 |
| College-level writing | No grammar or spelling mistakes. Content is organized well. | 2-3 grammar or spelling mistakes. Flow and organization of content could be improved. | 3+ grammar or spelling mistakes. Content does not flow and is unorganized. | 5 | |
| Citations | Included in-text citations and a complete works cited page. All information is in the student’s own words and appropriately cited. | Missing 2-3 in-text citations or works cited page is incomplete. Some information is not written in the student’s own words. | Missing 3+ in-text citations or works cited page is incomplete. Most information is not written in the student’s own words. | 5 | |
Deliverable
After you have gathered enough information, compose a 2-page paper about historic provisioning of freshwater by earlier civilizations. Please submit your paper using the Assignment 1.1 - What is freshwater? drop box under Lesson 1 in Canvas. (See the Calendar in Canvas for specific due dates.)
Summary and Final Tasks
Summary
You have now completed Lesson 1 and should have gained further understanding about the physical and chemical properties of freshwater that make it essential to our existence. You need to have basic knowledge of the three phases of water - what are they?! In the next several lessons, we will explore surface water and groundwater in much more detail, and then use that information to look at how we classify freshwater and how water of all phases and types strongly influences the Earth's weather and climate.
Reminder - Complete all of the Lesson 1 tasks!
You have reached the end of Lesson 1! Double check the to-do list on the Lesson 1 Overview page [3] to make sure you have completed all of the activities listed.