Lesson 2: Economic, Environmental, and Societal Issues in Materials Science
Overview
Overview
In addition to fundamental materials properties, selecting which material to use in an application can be limited by a number of factors. Some of these factors include the cost of production, availability of starting materials (natural resources), level of pollution resulting from the manufacturing process, and amount of waste produced at the end of the lifecycle of the application. In this lesson, I will present relatively brief overviews of economic, environmental, and societal considerations that are important in the materials selection process.
Learning Outcomes
By the end of this lesson, you should be able to:
- Diagram the total materials life cycle, and briefly discuss relevant issues for each stage of the cycle.
- List the inputs and outputs for the materials life cycle analysis/assessment scheme.
- Cite issues that are relevant to the "green design" philosophy of product design.
- Discuss recyclability/disposability issues relative to the three primary classifications of solid materials and composite materials.
- List and briefly discuss three controllable factors that affect the cost of a materials product.
Lesson Roadmap
Lesson 2 will take us one week to complete. Please refer to the course calendar for specific due dates.
| To Read |
Read pp 25-36 (Ch. 2) in Introduction to Materials ebook Reading on course website for Lesson 2 |
|---|---|
| To Watch | Making Stuff: Cleaner |
| To Do | Lesson 2 Quiz |
Questions?
If you have general questions about the course content or structure, please post them to the General Questions and Discussion forum in Canvas. If your question is of a more personal nature, feel free to send a message to all faculty and TA's through Canvas email. We will check daily to respond.
Reading Assignment
Things to consider...
While you read the material for this lesson in your e-book and on the course website, use the following questions to guide your learning. Also, remember to keep the learning objectives listed on the previous page in mind as you learn from this text.
- What are the relevant issues for each stage of the total materials life cycle?
- What are the inputs and outputs for the materials life cycle analysis/assessment scheme?
- What is the "green design" philosophy of product design and how does it differ from the total materials life cycle paradigm?
- What are the recyclability/disposability issues relative to metals, ceramics, polymers, and composites?
- What are the three controllable factors that affect the cost of a materials product and how do they affect the cost?
Reading Assignment
Read pp 25-36 (Ch. 2) in Introduction to Materials ebook
Introduction
In this lesson, we're going to look at the economic, environmental, and societal issues of materials science. The textbook reading for this week will introduce these topics, while the additional text on this website will supplement the reading material and explore further the topics of green design and social justice with regard to materials. The video for this lesson, Making Stuff: Cleaner, explores the science and technology of making energy production and usage cleaner and more efficient. Materials development in generating, storing, and distributing energy towards creating a more sustainable future are highlighted in the video.
To Read
Read sections 20.1 - 20.4 in the customized e-book (answer quiz questions on those sections, and then return to this website).
Economic Considerations
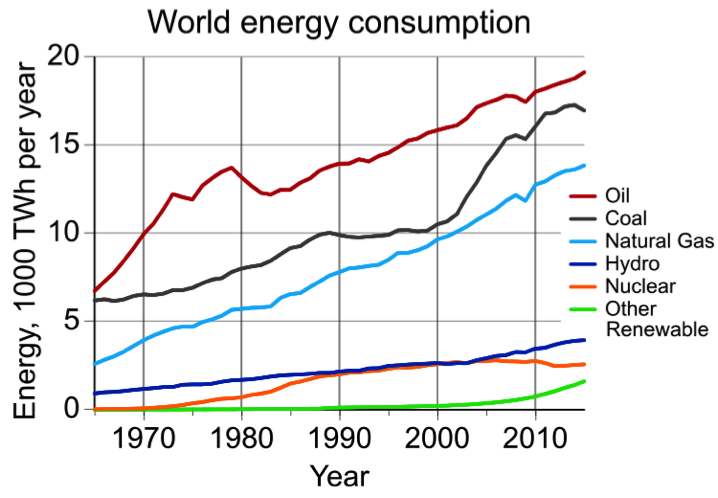
First and foremost, a product must make economic sense. The price of a product must be attractive to customers, and it must return a sustainable profit to the company. To minimize product costs, materials engineers should consider three factors: component design, material selection, and manufacturing technique. Also, there could be other significant costs including labor and fringe benefits, insurance, profit, and costs associated with regulatory compliance. As the world has become more populated and that population is increasing its usage of the earth's natural resources, engineers are increasingly being asked to consider sustainable practices when developing new products. Also, since it is estimated that approximately half the energy consumed by the U.S. manufacturing industry is used to produce and manufacture materials, the efficient use of energy for manufacturing processes and utilization of sustainable energy sources when available is highly desirable.
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 10 |
| 1980 | 13 |
| 1990 | 14 |
| 2000 | 16 |
| 2010 | 18 |
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 6 |
| 1980 | 8 |
| 1990 | 10 |
| 2000 | 11 |
| 2010 | 16 |
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 4 |
| 1980 | 6 |
| 1990 | 7 |
| 2000 | 9 |
| 2010 | 13 |
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 1 |
| 1980 | 2 |
| 1990 | 2.5 |
| 2000 | 3 |
| 2010 | 4 |
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 0 |
| 1980 | 1 |
| 1990 | 2.5 |
| 2000 | 3 |
| 2010 | 3 |
| Year | Energy, 1000 TWh per year |
|---|---|
| 1970 | 0 |
| 1980 | 0 |
| 1990 | ~.1 |
| 2000 | ~.1 |
| 2010 | 1 |
Sustainability represents the ability to maintain an acceptable lifestyle at the current level and into the future while preserving the existing environment. Your textbook discusses one approach to achieving sustainability: green product design. In the next section, we will look at some green design principles and examples of their application. Before moving on to that section, please watch the following short video. This (1:53) video on using renewable feedstock to replace nonrenewable starting raw materials highlights a green design principle used to make processes more sustainable.
To Watch
So far, many of our plastic products are derived from crude oil, which is a non-renewable source. We cannot grow or produce crude oil. This means we do not have an infinite supply of it. Instead, we must wait millions of years in order for dead carbon-based living organisms to be compressed by layers and layers of sediment before we get crude oil.
The term renewable feedstock the raw material that can be grown or produced by humans. The usage of renewable feedstock is attractive because it reduces the amount of harmful waste produced by the crude oil refinery and distillation processes.
Most printer inks are made from crude oil-derived pigments. If you think about the amount of printing that is done on a global scale this can be a problem in the long term. Currently in development are soy-based inks, which are derived from the oil of the soybean plant. As a plant, soybeans are a renewable resource.
The production process of these inks is overall more environmentally friendly than their petroleum-based counterparts. Also, the soy-based inks are much brighter than the petroleum-based inks. The recycling process of paper products printed with soy-based inks is also considerably more environmentally friendly. When paper products are recycled the inks need to be removed. Petroleum inks can be difficult to remove but soy-based inks can be removed with relative ease.
The term renewable feedstock refers to raw material that can be grown or produced by humans. The usage of renewable feedstock is attractive because it reduces the amount of harmful waste produced from the crude oil refinery and distillation processes. Most print inks are made from crude oil derived pigments. If you think about the amount of printing that is done on a global scale, this can be a problem in the long term.
Currently in development are soy-based inks which are derived from the oil of the soybean plum. As a plum, soybeans are a renewable resource. The production process of these inks is overall more environmentally friendly then their petroleum-based counterparts. Also, these soy-based inks are much brighter than the petroleum-based inks.
The recycling process of paper products printed with soy-based inks is also considerably more environmentally friendly. When paper products are recycled, the ink needs to be removed. Petroleum inks can be difficult to remove, but soy-based inks can be removed with relative ease.
Components of Green Design

There are three primary components of green design: reduce, reuse, and recycle. The reduce concept means to redesign a product to use less material. The reuse concept means to fabricate a product using material that can be used again. Recycling refers to the concept of reprocessing a product at the end of its lifecycle into new raw material that can be processed into new products.
One green design principle is that if there is less waste produced, then there is less to clean up. Please watch the following short (2:23) video that highlights this principle.
To Watch
When we say green chemistry we mean environmentally friendly chemical processes and doing chemistry with personal safety and the environment in mind. There are 12 principles designed to help make chemistry greener. We will discuss each of these 12 principles with an example.
Let's start with a simple concept. If you keep your room neat and tidy you won't have to clean up as much. If your room gets really messy then you will have a lot to clean up. The same concept is applied in green chemistry. If we prevent waste from being produced then there would be less to clean up. Now this is almost impossible to have zero waste for any given chemical reaction. On a global scale, The beauty of this principle is that it can be applied to almost every single chemical reaction conducted. For example, Polystyrene is a fossil fuel derived polymer used to make all disposable cups and packaging materials. The polymer itself needs to be expanded before it can be used in packaging. Traditionally this was done with CFCs or chlorofluorocarbons which deplete the ozone layer. Now this is done with carbon dioxide waste from other industrial processes. Instead of releasing carbon dioxide into the atmosphere and contributing to the greenhouse effect, this is contained and used to expand polystyrene. So, there is no additional carbon dioxide being released into the atmosphere.
Another green design principle related to producing less waste is to produce waste that is biodegradable. Please watch the following short (2:06) video that highlights this green design principle.
To Watch
A lot of waste is produced on a global scale. Unless the waste is recycled it fills up in our landfills, destroys habitats, and will be a very serious health hazard. Imagine if one day the waste that we produce can be naturally be broken down by microbes in the environment or dissolve into safer materials. This principle explores such a concept.
Ideally, whatever we use and throw away would present no hazard to the environment and would not accumulate in landfills. If you've ever had to get stitches because of an injury, the stitches slowly dissolve over time. This is in fact a polymer called Polyglycolic acid which is broken down into its respective monomers by enzymes in our body. This is then either respired as carbon dioxide or excreted in our urine. There is a new class of plastics known as the bioplastics. These polymers are made from natural monomers such as cellulose and lactic acid and can be broken down in the environment. For more information on these, please see our Synthetic Polymers video.
As a result of these bioplastics, carrier bags can be broken apart by microbes in the environment. This is important because it reduces the amount of waste that accumulates in landfills. Egg cartons once made of polystyrene, which is derived from petroleum-based products, are now usually packaged in newspaper material which can be recycled and therefore, do not accumulate as waste.
Some processes result in waste that is toxic or hazardous. The following video (2:04) showcases a genetically modified bacteria that has been developed to produce an enzyme that, when used with glucose, can replace a known carcinogen in a widely used synthesis process. In addition, the replaced chemical is derived from nonrenewable fossil fuels, while glucose is readily available, non-toxic, and renewable.
To Watch
The next time you look at tights, certain paints, or even plastics used in window frames you may want to think about the raw materials from which they are made. Do you know any of these materials? Pause the video, think, and resume when ready.
If you said Nylon for tights, you'd be right. If you said Polyurethane you'd be right for paints and coatings. Finally, plastic window frames are made of PVC. These contain a plasticizer allowing the frames to be easily molded. All of these materials require Adipic acid in their production. Usually, Benzene is used to make this compound, but unfortunately Benzene is carcinogenic or cancer-causing. Instead, glucose and a special enzyme extracted from genetically modified bacteria can be used to produce Adipic acid. What do you think the advantage is in doing the synthesis in this way? Pause the video, think about this, and resume when ready.
An answer is that glucose is harmless and is essentially sugar. So conducting the synthesis in this way protects the workers who would produce this compound by coming into contact with carcinogenic Benzene.
For more efficient use of energy, synthesis processes should be designed to occur near room temperature and at atmospheric pressure to reduce the amount of energy used when possible. Heating, cooling, and increasing or decreasing pressure, requires energy. The following green chemistry principle video (1:26) discusses the advantages of designing your synthesis process to occur near room temperature and at atmospheric pressure.
To Watch
Many chemical processes are designed to be as efficient as possible. There are many reasons why this is done.
Firstly, it is very costly if the chemical synthesis is not designed to be efficient. In industry, chemists are aiming to modify or develop reactions so that they'll proceed an ambient temperature and pressure. This is because it costs energy to produce both high and very low temperatures.
The 2005 Noble Prize for chemistry was jointly awarded to a group of chemists who developed an energy efficient catalyst for a high atom economy reaction so that it was able to proceed its standard ambient temperate and pressure. Recall that a high atom economy reaction is one that uses all the atoms in the reactant to create the product or products. The reaction is called the Metathesis method and is responsible for the synthesis of many polymers with special properties, additive to polymers and fuels, and biologically active compounds such as insect pheromones, herbicides, and drugs.
Recycling
Recycling of used products rather than disposing of them as waste is a desirable approach for several reasons. Recycled material replaces the need to extract raw materials from the earth. The energy requirements to process recycled materials are normally less, and in the case of aluminum much less, than the energy required to process extracted raw materials from the earth. In addition, recycling conserves natural resources and eliminates the ecological impact from the extraction of raw materials from the earth. Proper product design facilitates recycling, which reduces pollution emissions and landfill deposits.
Some issues surrounding recycling include that products must be disassembled or shredded to recover materials, and collection and transportation costs are significant factors in the economics surrounding recycling. The following video examines the anatomy of a properly designed landfill. After watching the video (4:39), proceed to your textbook and read section 20.5.
To Watch
Whenever you throw away your rubbish, do you ever wonder where it goes, what happens to it, and the effect this can have on the environment? The answer is that everything has not been recycled or reused ends up in a landfill site. What do you think this means? If you think that a landfill is an open hole in the ground where we bury the rubbish you'd be wrong. In fact, it is a carefully designed structure built into or on top of the ground to store your rubbish in such a way that it will be isolated from groundwater. Our freshwater supply will be kept dry preventing leaking into this groundwater and will not come into contact with the air causing horrible smells. This isolation is achieved with a bottom liner typically made from very thick PVC and a daily covering of soil. However, in many countries, open rubbish tips still exist causing environmental and health problems. If our rubbish is isolated from the surrounding environment it doesn't decompose. Landfill sites are not like a compost pile where the purpose is to bury rubbish in such a way that it will decompose quickly. Rather, landfill sites are simply a way that we cope with the vast amounts of rubbish that we produce.
Let's now take a look at the parts of make up the landfill cell and how these cells make up organized landfill sites. Groundwater has to be protected from chemicals found in our waste and so compacted clay is placed on top to act as a physical barrier. On top of this is a very thick waterproof plastic liner. This stops water that contains waste chemicals called leachate from contaminating this groundwater. A geotextile mat, typically made from polypropylene or polyesters are like a woven fabric, and this too prevents plastic sheeting from being damaged when a shark gravel layer as the rub on top is compacted. The gravel layer on top of the geotextile map filters large pieces of debris allowing only water through. this improves the drainage of the landfill cell. The gravel layer is connected to a leachate pipe. The leachate is collected through pipes where it goes into a leachate pond. This leachate can't be used as drinking water and needs to be treated like sewage before it can be moved on the sites. This must continue even when the landfill site is full. A drainage layer and the soil layer prevent firming from entering landfill cells. Finally, on top of the landfill cells old-new is where buried rubbish is compacted by heavy diggers of machinery. Bacteria in a landfill break down the trash under anaerobic conditions meaning in the absence of oxygen and so a byproduct of this breakdown is landfill gas. This presents a hazard because this gas contains methane which could explode therefore, it has to be removed.
To do this the series of pipes are embedded within the landfill to collect the gas and some landfills this gas is vented or burned. What do you think might be a better use for this gas? Pause the video and continue when you're ready.
If you said that the methane in landfill gas could be used as an energy source you'd be right. This means it could be collected and burning boilers to generate electricity.
In summary, a landfill is carefully constructed and allows waste only to be stored. It may never decompose and is not really a viable solution for dealing with waste. In addition extensive monitoring is required to ensure that no leachate escapes into the groundwater. This has to happen even once a land that is no longer in use. There aren't really any upsides to landfill where the gas produced can be collected and balance of fuel, it can also cause explosions if not vented properly. So we need ways to recycle, reuse, and reduce.
To Read
Read section 20.5 in the customized e-book.
In the next sections, we will be discussing the recycling of metals, glass, polymers, paper, and limits of recycling.
Recycling of Metals
As mentioned in the e-book, aluminum is the most commonly recycled nonferrous metal. (Ferrous is Latin for iron, so a nonferrous metal is a metal which does not contain iron.) Aluminum is recycled because it takes a lot less energy to recycle aluminum than it takes to extract aluminum from bauxite ore, which requires heating and electrolysis. In addition, aluminum readily forms an oxide that forms a protective surface. This protective surface protects the bulk of the aluminum from oxidizing further. This results in most of the aluminum being recovered every time it goes to the recycling phase, in contrast to iron.
In the case of iron, oxidation, i.e., rust, does not protect iron from oxygen and water, and significant amounts of iron are not recyclable because the iron has been converted to rust. Please watch the following video (5:04) which summarizes the points about recycling of metals emphasized in your e-book and this website.
To Watch
Certain metals can be extracted from their ores. For example, iron is extracted from hematite in a blast furnace and copper can be extracted from malachite. The process of extracting metals from their ores can be time-consuming, costly, and harmful to the environment. Other less reactive metals such as gold and platinum can be found naturally as native metal. The issue here is that there is a finite or limited supply of these metals on earth. In this lesson we will learn about recycling metals and the advantages and disadvantages to these industrial processes.
Aluminum is the most commonly recycled metal. Aluminum is used to make soft drink cans, aluminum foil, certain food cans, and even certain packaging materials. Aluminum is also used to make the outer bodies of some cars and airplanes due to the fact that it is less dense than steel. The extraction of aluminum from its ore is done by electrolysis. It may not seem like a lot of electrical energy is needed, but keep in mind that this is done on an industrial scale worldwide. The recycling of aluminum uses only 5% of the energy used in its extraction from its ore which uses a small voltage but a large current. In fact some countries really encourage recycling by having separate sections in their public waste containers. One for waste, one for cans and other metal products, and another for paper products. Some countries also have mandatory household recycling in a sense that if aluminum cans are found in regular waste bins for pickup the fine can be issued. In fact recycling aluminum is so common that any aluminum material that you encounter today has some if not more than 50% recycled aluminum content.
Steel can be recycled and most steel mills primarily use recycled scrap steel instead of caste or pig iron from blast furnaces. Copper is also recycled. Once again this is a more energy efficient process than the extraction from its ore as well the copper ore supply on earth is being depleted at a very fast rate. As a result other methods for the extraction of copper such as phytomining and bioleaching are being used instead.
Silver and gold from jewelry are also recycled. As these metals are found as native metals the issue here is not the energy cost but rather the rarity of these metals. The main source of gold is recycled electronic goods such as computers which have their electrical contacts plated with gold so there is no corrosion and a perfect electrical connection. The main issue with recycling metals is the separation process in the waste containers. Most often not all metal products are clearly labeled as recyclable. As well, many metal products are alloys. Can you remember what an alloy is? Please pause the lesson and continue when you are ready.
An alloy is a mixture of two or more elements with at least one of the elements in the mixture as a metal. Many metal products are made into alloys for increased strength and other desirable properties, but not all alloys can be recycled such as manganese aluminum alloys. Some alloys which can be recycled need to be molten and separated into their constituent metals thereby making it a more energy costly process. As well, we have to take into consideration the fact that these metal products must first be collected and then transported to recycling facilities. All depending on the distances covered this could involve quite a bit of fuel usage, and therefore increasing CO2 and noxious gas emissions.
In summary, aluminum steel and copper are the most commonly recycled metals and the recycling process usually uses significantly less energy than extraction from their ores.
In the next section, we will discuss recycling of ceramics, in particular, the recycling of glass which is the most common commercial ceramic.
Recycling of Glass
Glasses are the most common commercial ceramics, however, there is little economic incentive to recycle glass. The raw materials for producing glass are inexpensive and readily available. Glass is relatively dense, which makes it expensive to transport which adds to the costs of recycling. Glass must be sorted before being processed during recycling, usually done manually which adds to costs. Not all glass is recyclable, and the glass comes in many different forms. Please watch the following video (3:29) which summarizes the points about recycling of glass emphasized in your e-book and this website.
To Watch
In this lesson we will learn about recycling glass. Although the usage of plastics is more common as it is lighter and less prone to breakage, glass is still widely used. Glass is used to make soft drink bottles, food containers such as plates and glasses, and vases. Glass is primarily silicon dioxide with a small percentage of calcium oxide and sodium oxide. The beauty about glass is that it is a hundred percent recyclable and unlike paper it does not break down into smaller components with each recycling process. For the moment, only glass bottles can be recycled not crystal glass, window glass, and windscreen glass on cars. This is because other materials are added to this type of glass and the recycling process at the moment cannot separate those materials from glass.
First, glass bottles and containers need to be collected and separated according to their color. This is important because the different colors are made due to many different substances that are added. Ideally, green glass will be recycled into green glass and brown glass into brown glass. Any the labels present must be removed and the bottles must be thoroughly washed to ensure that any residual contents and adhesives are removed. The bottles are then crushed into smaller pieces before they are melted. Why is the step important? Please pause the lesson to think about this and resume when you are ready.
This is an important step because the smaller pieces mean that melting will occur quicker due to the increased surface area. Following on the same idea if you want it to fully dissolve sugar in water, powdered sugar would dissolve faster than a cube of sugar. The smaller pieces of glass are then melted, poured into a mold, and allowed to cool and harden. The bottles are now fully recycled and ready for reuse. This recycling process uses a lot less heat energy than glassblowing and it's therefore, a more environmentally friendly option. As well as being recycled into bottles, mixed glass can be crushed to form glass aggregate and used as a cheaper substitute for gravel.
In summary, glass is fully recyclable and can be recycled a virtually infinite number of times without being degraded into other products.
In the next section, we will discuss some of the limitations of recycling.
Limits of Recycling
Recycling has a number of advantages. Properly done, it reduces the usage of raw materials, energy usage, air pollution, water pollution, and greenhouse gas emissions. There are, however, a number of limits to the effective implementation of recycling. Recycling can involve energy usage, hazards, labor costs, and practices by individuals and countries, which can hamper the efficient implementation of recycling plans. The biggest limit to recycling is that not all materials can be recycled and so materials can only be recycled a limited number of times due to degradation each time through the process. This degradation is referred to as downcycling.
In addition, recycling poses a number of societal and ethical issues. As highlighted in the e-book, e-waste recycling has led to electronic waste from developed countries being shipped to undeveloped countries for recycling. In many cases, this leads to low wages and terrible conditions for workers involved in the recycling process and the release of toxins which are environmental and health risks for the individuals and their surrounding communities. Please watch the following video (5:26) which summarizes the limits of recycling as discussed in your e-book and this website.
To Watch
A common definition is that recycling is a process which allows waste materials to be turned into new products and prevents the waste of potentially useful materials. Recycling reduces the use of fresh materials. It acts to reduce energy usage, reduces air pollution from incineration, reduces water pollution from land filling by reducing the need for conventional waste disposal, and has lower greenhouse gas emissions as compared to fresh material production. So, with all of these benefits you must be wondering if there are any limits to a process that can have such a positive effect on the environment. Limits to recycling are often considered in terms of energy, hazards, costs, and practices by individuals and countries. The biggest limits of recycling however, is that not all materials can be recycled or can only be recycled a number of times before they lose their quality. Some materials once used are always going to have to be dumped as we have no method for making them useful again. What sorts of materials do you think can be recycled? See if you can come up with five. Pause the video and continue when ready.
Commonly, these are the materials that are recycled: glass, paper, metal, plastic, textile, and electronics. How many did you get?
Now let's consider the different limitations. First up is the energy aspect. Put simply some materials like paper and aluminum metal agree to have lower processing costs when it comes to recycling them rather than using fresh materials to make new products. However, the recycling of materials like plastics is extremely energy intensive. Before the plastics can be melted and mixed together, they require sorting usually by hand as there are many different types of plastics usually indicated by special marking a number. If a mixture of plastic recycled together now ever contaminate the melt or you get a lower grade of plastic than the originals which is therefore less valuable. This reduction in the quality of recycling product is called downcycling. In addition to this there is the added complication that devices often use mixed materials. Think of a car. There are wide range of recyclable materials the copper wire the aluminum in engine some of the plastics, the glass, and the iron however, what about the alloys? These are mixtures of metals energy will be needed to separate these and so one of the most difficult problems of recycling is the separation of randomly intermixed particles.
Secondly, there are hazards to recycling including the recycling of dangerous metals. Can you think of any dangerous metals? Pause the video and name some. Continue when you are ready.
Some of the metals associated with recycling are lead and mercury. Often these metals can come from the recycling of waste electrical and electronic equipment. In India and China, a significant amount of pollution is generated where informal recycling in an underground economy of these countries has generated an environmental and health disaster. High levels of lead polybrominated diphenyl ethers, polychlorinated dioxins and furans, as well as polybrominated dioxins, have become concentrated in the air, bottom ash, dust, soil, water, and sediments in areas surrounding these underground recycling sites. Many of these chemicals become dissolved in the water that we drink called leachates. Also, plants can take up these chemicals allowing toxic chemicals to enter the food chain. Finally, if the chemicals are airborne there are own problems with us inhaling them.
There are also social issues connected to recycling whilst it may create jobs they are often jobs with low wages and terrible working conditions in developing countries. In areas without many environmental regulations or worker protections, job like ship braking can result in deplorable conditions for both workers the surrounding communities.
Thirdly, is a challenge for you what do you think the cost of recycling depends on? Pause, think, and continue when ready.
A good answer might be that the costs of recycling depend on the efficiency of the recycling program. Governments or local authorities may not recycle because it's cheaper to use landfill. Also, consumers are encouraged to recycle, but this depends on people being involved in pre-sorting their recycling. Some countries or local governments impose charges when this is not done.
In the next section, we will discuss the recycling of polymers, in particular, plastics.
Recycling Polymers
One way of classifying polymers is to break them up into two classes. The two classes of polymers are thermoplastic polymers and thermosetting polymers. The basic property that separates a thermoplastic polymer from a thermosetting polymer is the polymer’s response to being heated. When the thermoplastic polymer is heated, it melts, softens, and can be reformed when cooled. When the thermosetting polymer is heated, it hardens and cannot be reformed and stays hard when cooled. We will learn much more about each of these two classes of polymers and the reasons for their defining properties later in our lesson on polymer structures.
Since thermoplastic polymers can be melted and reformed, they are easily recycled. However, their properties do degrade with each reuse. Thermosetting polymers are much more difficult to recycle. Some of them can be ground up and used as filler for other processes, and, on a case-by-case basis, some can be processed to be broken down into their underlying base units which can be reused. Another approach to reducing the amount of plastic that ends up in our landfills is the development of biodegradable plastic. The idea here is that plastic can be made to breakdown (be compostable). In addition, bioplastics often come from renewable raw materials. But this leads to an ethical issue: do you use the available arable land for plastic or food production?
Now, please watch the following video (4:38) on plastics and biodegradable plastics which summarizes some of the issues around plastic recycling and bioplastics as discussed in your e-book and this website.
To Watch
In the previous video, Limits to Recycling, we looked at the overall limits to recycling of materials. We touched on plastics in that video. You may be aware that the biggest problem in recycling plastics is they are not biodegradable so that soft drinks bottle or your wrappers that you just throw away and not biodegradable. This means the fate of most plastics is to go into landfill sites or our oceans where it will take hundreds possibly thousands of years for them to degrade. You may say what about recycling? Briefly, the problems with recycling plastics are they have to be manually sorted which is a labor and energy-intensive process. Mixed polymers are found in a range of materials so cannot be recycled and are often dumped. Once a plastic has been recycled once it can become downcycled whereby its quality is decreased. The fact is that plastic recycling rates are far below other recycling rates, but the industry has improved in recent years.
Worldwide seven groups of plastic polymers have been given a plastic identification code that people use for recycling. You can find these on most packaging or plastic materials. Different types of plastics will be recycled by different municipalities even by different countries in some instances. Generally, it's difficult to recycle plastics with numbers one and two. Plastics in these higher numbers are known as rigid plastics. So, the disposal of plastics is a tricky one. Other than reducing their use of recycling or reusing them what else do you think might be done to dispose of them? Pause the video and continue when you think you have an answer.
One way is that the plastic can be incinerated or buried. Whilst this produces useful energy, some plastics contain polymers that are halogenated When these polymers are burnt. For instance, PVC, toxic fumes like hydrochloric acid are released. This can cause severe respiratory distress and so is hugely problematic. In addition, burning of polymers produces CO2. The greenhouse gas also contributes to global warming. This is because about 10% of the oil is used as petrochemicals. If we then use the polymers again as a fuel for energy production then at least we're using the oil twice. You may have heard the idea of using bioplastics as a solution, but these two can have problems. What do you think bioplastic is? Pause the video and resume when you have an answer.
Bioplastic is a biodegradable plastic. This means it's compostable and it can be broken down by the result of bacterial action when it is disposed of. Whilst this is a benefit, there are still issues with bioplastics that need solving. Bioplastics often come from renewable raw materials like starch, maize, cellulose, and polylactic acids. These are plant-derived materials and so the ethical question arises is where the land should be used to grow crops or bioplastic manufacture instead of crops for food? Originally we said that bioplastics are readily compostable, however, bioplastics are not nearly as readily compostable as regular plant material. If you toss a bioplastic fork into your compost and assume it will be dirt in a few months you'll be disappointed. Whilst a bioplastic fork is compostable it requires high-intensity high heat commercial composting for that to happen quickly.
In summary, plastic disposal has a number of issues with regards to landfills, recycling, and incineration. However, bioplastics so slightly better do not present themselves as the better alternative because there are issues of land usage, the quality of the bioplastic, and how it is composted. The best alternative is to reduce our usage and disposal of plastics altogether.
In the previous video, the incineration of waste was discussed. Incineration leads to a huge volume reduction of waste, which results in less waste ending up in the landfill. Waste in the landfill is the least environmentally friendly option. However, incineration typically results in less recycling, which would be a more efficient use of recyclable material than incinerating it. This reduction of recycling due to incineration is considered the major disadvantage of incineration. Although an important concern with incineration is the production of toxins, with proper technology these toxins can be managed. A segment of the video for this week, Making Stuff: Cleaner, discusses burning waste to create electricity. Please watch the following short video (4:40) which discusses burning waste to create electricity as well as the issues regarding incineration discussed above.
To Watch
In this video, we're going to consider the advantages and the disadvantages to waste incineration. What do you think are the advantages of burning waste instead of committing it to the landfill? Pause the video and continue when you have an idea or some ideas.
If the waste is not burned, then it is likely to end up in a landfill site which is considered to be the least environmentally friendly option. Incineration could result in a reduction in the waste volume of around ninety percent and this could be particularly important for small cities where space is scarce and landfill is not an option. Odors and rodents are present in other methods, are not a problem when using incineration as a garbage disposal method. Nearly all of the waste that is burned could be used to generate electricity in what is called energy-from-waste schemes where households and industries could benefit from the electricity or heat produced. The electricity generated could help to pay for the startup costs of the incinerator. Additionally, the steam produced from incineration presents itself as a cost-saving energy source if recycled. Ash produced from these incinerators could be used in the construction and road-building industries. In addition, metals could be extracted from the ash and they could also be used in steel industries. The key advantage, however, is that the production of electricity and thermal energy from waste enables us to conserve conventional sources of energy such as fossil fuels? Now, what about the disadvantages of incineration? Again pause the video and think about what these could be. Resume when you have an idea or some ideas.
The main disadvantage surrounds potential pollutants found in the ash left in the incinerator and those that could be emitted from the chimney. These include dioxins, acid gases, nitrogen oxide, heavy metals, and particulates. As you may have heard from other videos, these are airborne particles that are small enough to get into the lungs of humans. They cause and aggravate respiratory problems such as asthma; however, it is the dioxins resulting from incomplete combustion contained in the gases from the chimneys that attract the most concern. This is because they are suspected of causing cancer. In addition, these emissions could be distributed through the food chains and accumulate over a long period of time impacting both ecosystems and human health. As a result, many people are opposed to having incinerators built in or near their communities.
Another disadvantage is the initial startup costs to build a waste incinerator for the production of electricity and may not be seen as a viable alternative. Where cheaper waste disposal methods exist, once built the maintenance of the machinery could be costly. The biggest disadvantage, however, is that if waste is incinerated without first being sorted this means we are incinerating waste that could have been recycled.
So in summary, the incineration of wastes presents numerous advantages. One, the energy is used the heat or to produce electricity for homes in the industry. Two, this method reduces the need for fossil fuel consumption. Three, the ash produce can be recycled for metals used in the steel industry, and finally four, this method reduces the volume of waste produced. There are also disadvantages. One, incinerators are expensive to build and maintain. Two, pollutants in the ash and emissions from incinerators are harmful to health and ecosystems and finally, three, waste that could be recycled will instead be incinerated if not properly sorted.
Lastly, please watch the following video (5:40) on the recycling of paper, which touches on several themes of this lesson including sustainability, downcycling, and green design principles.
To Watch
Look around you. There are so many paper products from the sheets of paper that you write on to the pages of your textbook - to egg cartons, to newspapers, to certain grocery bags - cardboard boxes, and the list continues. The important fact here is that paper is an essential part of our lives. You may know that paper usually comes from trees. They must first be chopped down and then subjected to an industrial process called pulping which removes the lignin, the polymer that gives trees their strong hard structure. Obviously the paper production process has led to mass deforestation. The main issue here is that the trees are not being replanted, or if they are being replanted they will not grow fast enough to replace the trees that are being cut. This is a serious environmental issue because it destroys habitats, disrupts food chains and food webs, is a severe eyesore, and indirectly leads to increased carbon dioxide content in the atmosphere. It could also lead to erosion of hillsides with the consequence of flooding. An obvious solution is to recycle these paper products. In this lesson, we will learn about recycling paper and the advantages and disadvantages of this industrial process.
Paper is in fact layers of cross-linked cellulose fibers. Recall that cellulose is a polymer of glucose monomer units. If you tear a piece of paper and look closely, you will see fine hairs sticking out. These fine hairs are cellulose fibers. When paper products are recycled the cellulose furs are merely pulled apart and rearranged into new paper products. In some countries, paper recycling is highly encouraged by having specialized waste containers just for paper waste. The paper waste is then collected and separated according to their properties. Sheets of paper are recycled with other sheets of paper and cardboard boxes are recycled with other cardboard boxes.
Now, right before the paper can be recycled, any ink needs to be removed. This process is called de-inking. Most inks are petroleum-based inks which are derived from crude oil products and can be difficult to remove. Currently in development are soy-based inks which are derived from a renewable resource and makes it easier for the paper to be de-inked. Metal parts such as staples and paper clips need to be removed with a magnet and glues are removed with a chemical wash. Bleach is sometimes added to whiten the slurry and therefore, the resulting paper product will be brighter. The cellulose fibers in the slurry are mixed with new pulp and then allowed to settle on gauze and rolled to form the new sheets of paper. This step also removes any excess water from the slurry before it is air-dried.
The main disadvantage with recycling paper is that it can only be done a few times. Recycled paper is usually used to make kitchen rolls, toilet paper, and packaging materials. Each time a paper product is recycled the cellulose fibers become shorter thereby rendering the recycled paper product a little bit less useful than the original. However, this can be used for another purpose. Can you remember what cellulase is? Hint: it is an enzyme. Please pause the lesson to think about this and resume when you are ready.
Cellulase is the enzyme that breaks cellulose apart into its glucose monomers. Currently, the industrial usage of cellulase is to break down cellulose into glucose. The glucose obtained can then be fermented by certain bacteria to produce ethanol, which can then be used as a biofuel.
In summary, recycling paper products help to reduce the rate of deforestation and consumes a lot less energy. Although paper can only be recycled a few times, the cellulose fibers can be broken apart into glucose monomers which can then be used for other environmentally friendly applications.
Video Assignment: Making Stuff Cleaner
Now that you have read the text and thought about the questions I posed, go to Lesson 2 in Canvas and watch the Making Stuff: Cleaner video (55 minutes). This video highlights some innovations in materials science that can potentially help make our technology use cleaner in the future. In "Making Stuff: Cleaner," in contrast to the readings of this lesson, the rapidly developing science and business of clean energy is explored. Some of the latest materials developments in generating, storing, and distributing energy are investigated in the hope of creating a sustainable future.
Video Assignment
Go to Lesson 2 in Canvas and watch the Making Stuff: Cleaner Video (55 minutes). You will be quizzed on the content of this video.
Summary and Final Tasks
Summary
Producing a sustainable society is one of the greatest challenges facing our society. The supply of natural resources, the creation of pollution during the manufacture of materials, recycling issues, and materials waste all issues of concern towards creating a sustainable society. By considering a material's total life cycle, utilizing materials life cycle analysis, and implementing a ‘green design’ philosophy, engineers can work towards alleviating some of these issues.
Reminder - Complete all of the Lesson 2 tasks!
You have reached the end of Lesson 2! Double-check the to-do list on the Overview page to make sure you have completed all of the activities listed there before you begin Lesson 3.