Lesson 5: Structure and Applications of Metals
Overview
The crystal structure of a material can directly affect their properties. For example, gold and silver which share a common crystal structure are much less brittle than the metals beryllium and magnesium which possess a different crystal structure. Also, crystalline and noncrystalline materials of the same composition can possess significantly differing properties. In this lesson, we will discuss how structure can affect materials properties and also introduce imperfections, which can have major impacts on the properties of materials.
Learning Objectives
By the end of this lesson, you should be able to:
- List and explain the contributions to material processing made by the Egyptians.
- Explain the difference between melting and smelting.
- Distinguish between single crystals and polycrystalline materials.
- Describe the difference in atomic/molecular structure between crystalline and non-crystalline materials.
- Draw unit cells for face-centered cubic, body-centered cubic, and hexagonal close-packed crystal structures.
- Define polymorphism and allotropy.
- Sketch the three orthogonal crystal systems (cubic, tetragonal, orthorhombic) and the hexagonal crystal system with proper lattice parameter labels.
- Define isotropy and anisotropy with respect to material properties.
- List and describe the different types of imperfections in a crystal.
Lesson Roadmap
Lesson 5 will take us 1 week to complete. Please refer to Canvas for specific due dates.
| To Read |
Read pp 99-120 (Ch. 5) in Introduction to Materials ebook Webpages on this site for Lesson 5 |
|---|---|
| To Watch | Metal: The Secret Life of Materials |
| To Do | Lesson 5 Quiz |
Questions?
If you have general questions about the course content or structure, please post them to the General Questions and Discussion forum in Canvas. If your question is of a more personal nature, feel free to send a message to the instructor through Canvas email. I will check each of these daily to respond.
Things to Consider...
While you read the material for this lesson in your e-book and on the course website, use the following questions to guide your learning. Also, remember to keep the learning objectives listed on the previous page in mind as you learn.
- What contributions did the Egyptians make in the development of materials processing?
- What is the difference between melting and smelting?
- How do single crystal and polycrystalline material differ in grain structure?
- How do crystal and polycrystalline materials differ in their atomic/molecular structure?
- What is the difference between an amorphous and crystalline material?
- What is the difference between crystal structure and a crystal system?
Copper (Chalcolithic) and Bronze Ages
In the Neolithic Age, which was the period at the end of the Stone Age, the Egyptians were experiencing increasing population along with extensive food production capabilities, several communities in similar stages of development, and an extensive trade network with other civilizations. As the Egyptians entered the Copper and Bronze Ages, their technological advancements in gold, copper, and bronze processing were aided by their access to key natural resources. The figure below from the British Museum shows known natural resources of ancient Egypt.
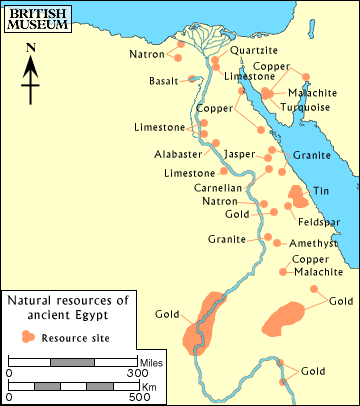
In the following pages, we will discuss gold, copper, and bronze processing as developed by the Egyptians.
Egyptian Gold Processing
It is not surprising that gold was the first metal processed by the Egyptians. Very few metals are found in their native state, i.e., not bound to other elements in a compound such as a mineral. Copper is very rarely found in nature as an element and iron is typically only found as an element in some meteorites. Iron from meteorites was extremely rare in Egypt and was known as metal from the gods. Gold, however, is routinely found in nature as an element unlike copper and iron, and most other metallic elements. Gold, although rare, can be found as flakes or nuggets. As shown in the illustration below from an ancient Egyptian tomb, the Egyptians used charcoal and blow pipes to reach the temperatures needed to melt gold. Also, ‘slag’ (impurities) were skimmed off the molten gold.
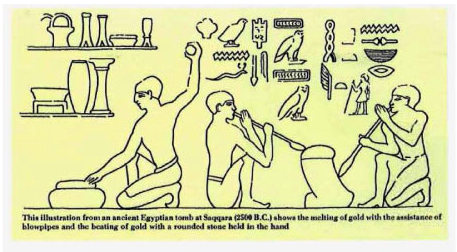
The molten gold was poured into molds to form jewelry and other items. In addition, the Egyptians were able to hammer gold into very thin (5 µm) leafs. Gold is a malleable material. Malleability is a material’s ability to be deformed under compressive stress, i.e., to form a thin sheet by hammering or rolling. A ductile material (ability to be deformed under tensile stress, i.e. can be pulled into a wire) has to be a malleable material as well, but malleable materials do not have to be a ductile material. An example of this is lead. Lead is malleable but when pulled to form a wire it pulls apart. As you can see malleability and ductility are closely related but do not possess the same definition in material science.
The Egyptians believed gold to be a divine material which held magical powers. Electrum is an alloy of gold which is approximately 80% gold mixed with 20% silver. An alloy is a mixture of metals or a mixture of a metal with small amounts of non-metals. We will discuss metal alloys in more detail in the next lesson. In the next section, we will discuss Egyptian copper processing.
Egyptian Copper Processing
Native copper occurs in a very limited supply, so the start of the Copper Age is marked by the discovery of smelting copper from its ores which allows for a ready supply of copper. The two basic naturally occurring copper (II) carbonate minerals are pictured below.
Copper is a very malleable material, unlike flint, which at the beginning of the Copper Age was the dominant weapon and tool material. Although copper is soft it does have a significant advantage over flint. It can be repaired. Native Americans used native copper beginning circa 6000 BCE. As mentioned, the supply of native copper is very limited and its supply was easily exhausted. Copper ores, on the other hand, were readily available. However, to extract the copper from copper ores smelting was required.
What is smelting? Smelting is a process that uses heat and chemistry to drive off other elements such as gases or slag, leaving behind only the metal. Typically, ores are impure and require a flux to separate the metal from the slag. Flux is an additive used to change the impurities from a form that is inseparable from the metal to a form that is separable. For example, adding iron ore as a flux during the smelting of copper can transform the impurity solid silicon dioxide into an iron-silicon oxide. Unlike the solid silicon dioxide which remains in the liquid copper, the iron-silicon oxide floats to the top and can be skimmed off. Smelting is different than melting in that in melting you have to be able to raise the temperature to the melting point of the material. The Egyptians did not have the ability to reach the temperatures needed to melt the copper minerals outright.
How Did Egyptians Discover that Malachite and Azurite Contain Copper?
It is unknown exactly how the Egyptians discovered that malachite and azurite contain copper. Here are a couple of possibilities.
Egyptians used malachite as a pigment and cosmetic, including as a distinctive eyeliner. While a normal open fire would not reach the temperatures required to melt bulk malachite, in powder form it is possible that accidentally putting Malachite powder on the coals of the fire could produce small balls of copper.
A second, and more likely possibility centers around the Egyptians using malachite as a pottery glaze. Small balls of copper could have been formed in the pottery kiln during firing, and then noticed by kiln workers after cooling.
Egyptian Copper Smelting Process

The Egyptian copper smelting process utilized a ’bowl furnace’ which was supplied additional air, to raise the temperature of the fire, through the usage of foot bellows. Malachite and azurite were used as a source ore for the copper, charcoal was used as the reducing agent to separate oxygen from the copper, and iron ore was used as the ‘flux’ to bind and float away impurities. The advantage that copper has over bone, wood, or flint, as a tool or weapon, is that when damaged it can be repaired. However, like most pure metals, i.e., metals with a low level of impurities, copper is soft. It turns out that intentionally or unintentionally adding another soft metal in small amounts can make the host material stronger and harder. This is a process known as alloying, which we will be discussing further in the next lesson.
Now let’s go to the next section and look at the soft metal, tin, that the Egyptians added to copper to produce a more durable and harder metal.
Egyptian Smelting of Tin
It is not known how exactly the Egyptians discovered the smelting of tin. In some ways, it is a bit surprising. Cassiterite, SnO2, the mineral used as the ore for extracting tin, is extremely difficult to find and not particularly noteworthy, i.e., it does not stand out in the field. It is hard and, like gold, has a high specific gravity. Having a high specific gravity means that flakes or nuggets of cassiterite, like gold, would settle to the bottom of a slurry if panning for gold. So, while it is difficult to find sources of cassiterite it might have been possible to backtrack upstream by finding cassiterite flakes downstream of sources.
While it might be counterintuitive to think that the Egyptians added an even softer metal, tin, to copper to make it harder, it is possible that the Egyptians thought that whatever made cassiterite hard would be transferred to the copper. The smelting of tin is very similar to the smelting of copper. Charcoal is also used as the reducing agent. Tin, unlike copper, is too soft for practical purposes. However, when it’s mixed with copper in small amounts, typically 5 – 10% tin, it can produce a much harder metal than unalloyed copper or tin. Please proceed to the next section to learn more about this new harder metal, bronze.
Egyptian Bronze Processing

Bronze is an alloy of copper and tin. Tin is a slightly bigger atom than copper. In bronze, typically 5 – 10% is tin and the rest is copper. The slightly larger tin atoms replace copper atoms in the copper crystalline structure as shown in the figure below. We will learn more about metal alloys in this lesson and the next. Although copper and tin are both soft metals and not ideal for tools or weapons, the combination that produces bronze is much harder than copper or tin. As we will learn later, this is due to the larger tin atoms making it harder for rows of copper atoms to move. This results in bronze being harder.
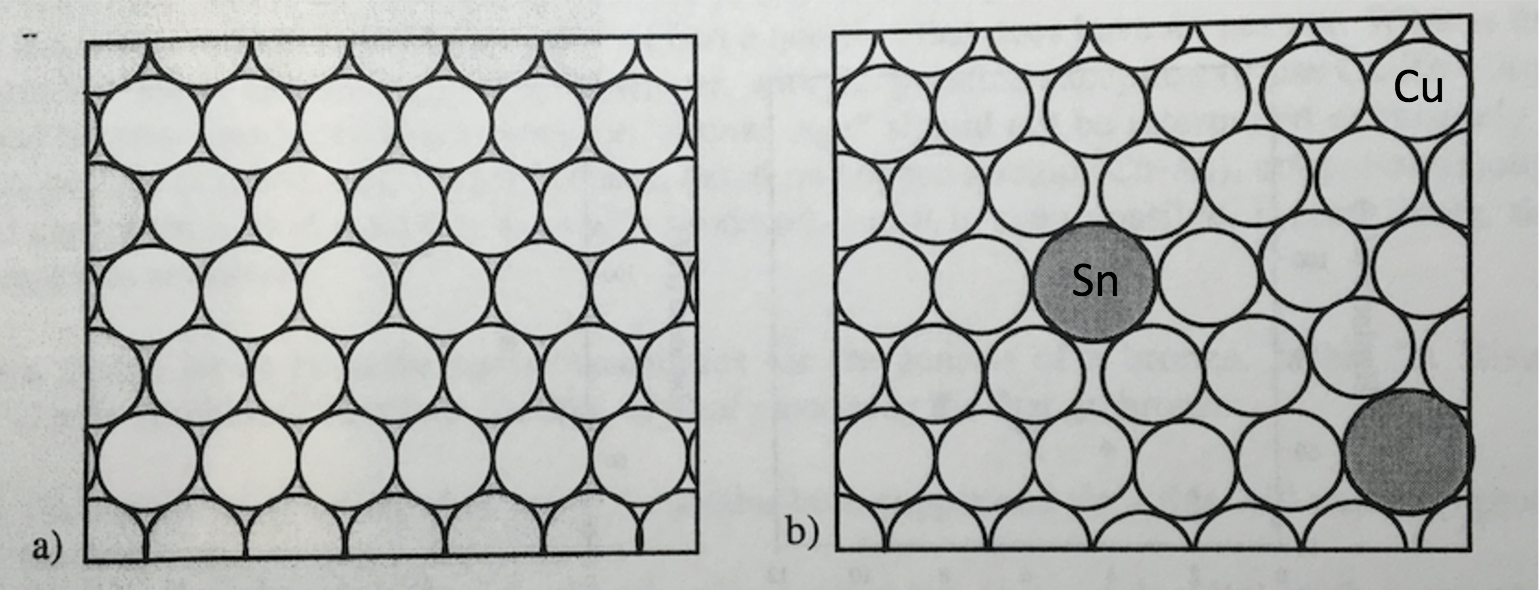
In the practice of producing bronze, the Egyptians placed tin with copper ingots into clay crucibles. The clay crucibles were lowered into a charcoal fire which could exceed 1100 °C through the use of blowing air using foot bellows. The Egyptians would then stir, remove the slag, and pour the melt into a mold.
Why Did it Take So Long Between the Bronze Age and the Iron Age?
The beginning of the Bronze Age occurred around 3500 BCE and the beginning of the Iron Age began around 1000 BCE. Why did it take 2000 years for bronze to be replaced by iron? Looking around us we see structural steel and concrete seemingly everywhere in our modern cities. However, the processing of iron is not a trivial process.
Due to limitations in furnace designs, i.e., the maximum obtainable temperatures, the availability and quality of iron varied greatly. As we’ll see in the next lesson’s video, Secrets of the Viking Sword, throughout history there have been legendary quality swords, i.e., Damascus and Samurai to name just a couple. These swords were produced using time-intensive and, many times, ritualistic processes. These blades were produced in areas known in the modern day as Iran, Japan, and China. Most of the iron used in weapons during the Iron Age, i.e., Roman swords, was a low-density iron sponge-like material. This sponge-like iron was then pounded to shape, densify, and remove impurities. Bronze was superior to the iron produced commonly, so why did iron ultimately replace bronze?
Bronze weapons were indeed of higher quality than the common iron weapons typically produced. However, tin, which is required for the production of bronze, is not abundantly available. As a consequence, bronze weapons were the weapons utilized by nobles, royalty, pharaohs, etc. The common foot soldier was not going to possess bronze weapons; there were not enough to go around.
Unlike tin, iron ore is readily available. So, although inferior to bronze, an army of hundreds or thousands could be equipped with iron weapons, which was not practical with bronze weapons. So, the ability to produce large numbers of iron weapons overcame the advantages of bronze. Eventually, time and further development allowed for the production of these so-called legendary swords which supplanted bronze as the weapon material of choice for the nobility. But it wasn’t until much later, during the advent of the Industrial Revolution, that advancements in furnace design and process control enabled the reliable and massive production of the iron alloy known as steel. In this lesson’s video, the importance of steel and how the production of steel was changed during the beginning of the Industrial Revolution will be showcased. We will return to this topic at the beginning of the next lesson on metal alloys.
Now, let’s take a step back from our discussion on the historical development of metal processing and begin an introduction to the structure of metals.
Structure and Application of Metals
When you mention crystal to most people, they think of fine glassware. Metal is not the first thing that comes to mind. But, in fact, most metals are crystalline, and it is rather difficult to make noncrystalline metals. Crystalline materials have their atoms arranged in a periodic, ordered 3D array. Typically, all of the metals, many ceramics, and some polymers are crystalline. Noncrystalline materials have atoms with no periodic arrangement, i.e., a random order. Noncrystalline material can result when you have complex structures or you rapidly cool from the liquid state to the solid state. Amorphous material is another name for noncrystalline material.
Why do metals form crystals? It turns out that the lowest energy for metal atoms occurs when the atoms are packed together as tightly as possible. If you’ve ever tried to put many small pieces into a large box, you know that if you put the pieces in the box in an ordered fashion you can fit much more in the box than if you just throw things into the box in a disorderly fashion. So, for metals, ordered structures tend to be nearer the minimum energy and are more stable. In addition, since metallic bonds are nondirectional it is much simpler for metal atoms to densely pack than it is for ceramics and polymers. So how do metal atoms pack together? In the next section, we will look at one of the ways that metal atoms pack together.
Simple Cubic Crystal Structure
Start by taking four atoms and arranging them in a square. Then take four more atoms and arrange them in a square. Then put the first square on the second square to form a cube with eight atoms, one at each corner. This structure is the simple cubic crystal structure. It turns out that only the metal Polonium (Po) has this crystal structure. The reason this crystal structure is so rare is that packing atoms in this way does not lead to a very high packing density. In the next section, we will add one atom to the simple crystal structure and produce a crystal structure that is much more common.
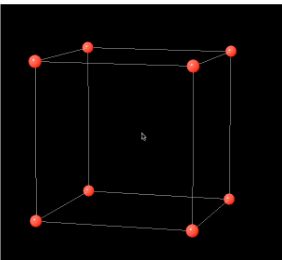
Body Centered Cubic Structure (BCC)
Let's take our simple cubic crystal structure of eight atoms from the last section and insert another atom in the center of the cube. This new structure, shown in the figure below, is referred to as body-centered cubic since it has an atom centered in the body of the cube. Some examples of metals that possess this crystalline structure include the α phase of iron, chromium, tungsten, tantalum, and molybdenum.
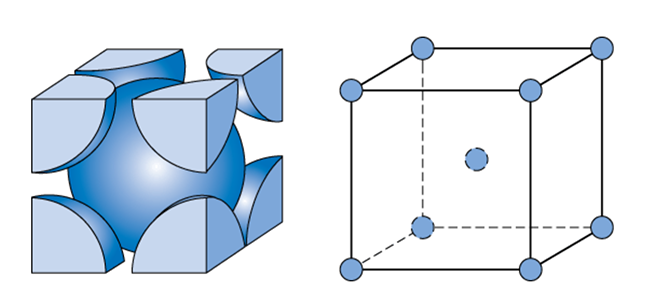
Face Centered Cubic Structure (FCC)
If, instead of starting with a square, we start with a triangle and continue to add atoms, packing as tightly as we can, we will end up with a layer of atoms as shown in the figure below.
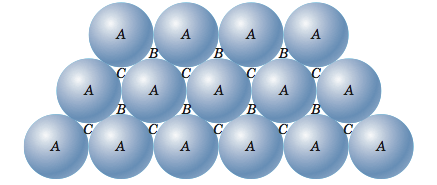
Now let me put an atom on top of that first layer over one of the 'B' positions and let it rest down into one of the valleys. I can now place two more atoms in nearby 'B' positions so that each will rest in their own valley in such a way that all three atoms will touch and form a triangle. Now let me add more atoms to the second layer, packing them in as tightly as possible. These two layers are shown in the figure below. If you look closely, you should be able to see that the second layer only covers half of the valleys produced by the first layer. The 'C' valleys are left uncovered. In fact, half of the valleys of the second layer lineup with the unoccupied 'C' valleys of the first layer.
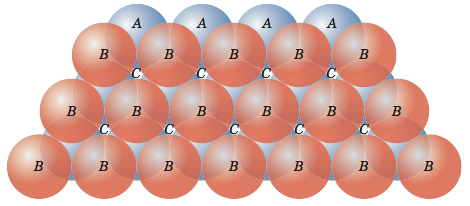
Now let’s put a third layer where the atoms are placed where the unoccupied valleys of the first two layers lineup, the 'C' valleys. It is a little difficult to visualize, but if one of the top layer atoms is one corner of our cube and that corner is pointing out then we obtain the cube shown in the figure below.
This crystal structure is known as face-centered cubic and has atoms at each corner of the cube and six atoms at each face of the cube. It is shown in the figure below. This structure, as well as the next structure we are going to discuss, has the atoms packed as tightly as theoretically possible. Metals that possess face-centered cubic structure include copper, aluminum, silver, and gold.
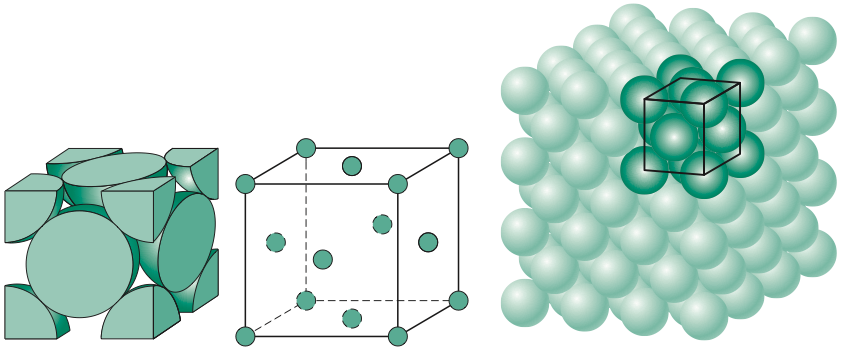
In the next section, we will discuss our fourth and last crystal structure.
Hexagonal Close Packed Crystal Structure (HCP)
If you look at the figure below, you might think that hexagon close-packed crystal structure is more complicated than face-centered cubic crystal structure. In fact, it is a simpler structure.
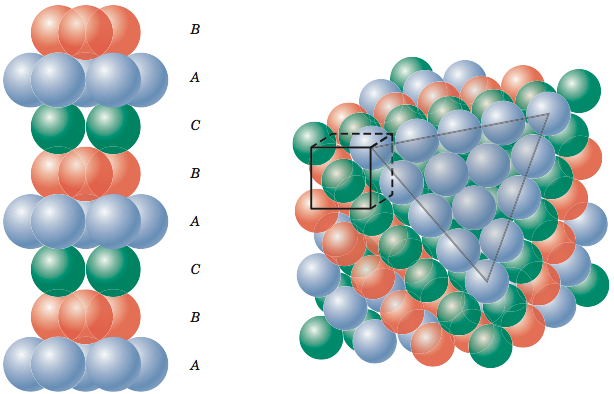
Think back to the last section where we constructed first one layer of atoms and then a second layer of atoms for face-centered cubic structure. Now, for hexagonal close-packed crystal structure, we do not construct a third layer. Instead, the third layer is simply the first layer repeated, the fourth layer is the second layer repeated, and so on and so on as shown in the figure below.
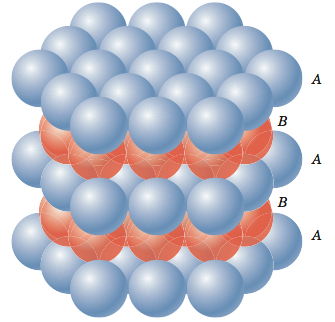
It turns out that face-centered cubic and hexagonal close-packed crystal structures pack atoms equally tightly. Some metals with hexagonal close-packed crystal structures include cobalt, cadmium, zinc, and the α phase of titanium. A more typical representation of the hexagonal close-packed structure is shown in the figure below. In this representation a hexagon on the top and on the bottom sandwich a triangle in between the two hexagons.
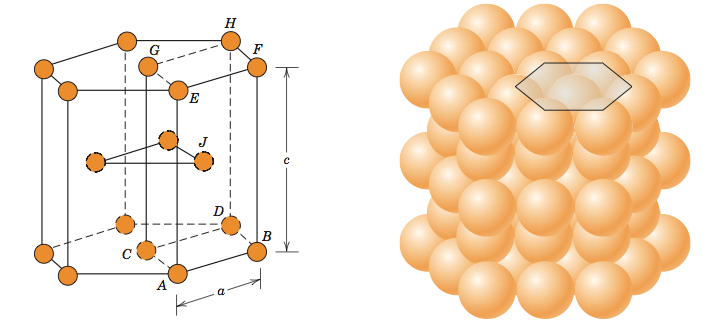
Please proceed to your e-textbook and read the first chapter of this lesson’s assigned reading. Please return back to this website after completing that reading.
Reading Assignment 1
Now please proceed to the first reading assignment (shown below) from your e-book. After you have completed that reading please return to this page and continue the web reading.
Reading Assignment
Read pp 99-120 (Ch. 5) in Introduction to Materials ebook
Now you should be able to distinguish between single-crystal and polycrystalline materials. If you cannot draw unit cells for face-centered cubic, body-centered cubic, and hexagonal close-packed crystal structures, you should review those.
Metals routinely form crystals. However, sometimes metal is formed from many grains rather than a single crystal. A grain is a region of single crystallinity and material with many grains is a material with many crystals (grains) that are misaligned to each other. This would be termed a polycrystalline material.
Many materials, e.g. iron, titanium, and carbon, possess two or more distinct crystal structures, which is referred to as allotropy or polymorphism. We have discussed metals as though they form perfect crystals, but it turns out that in real life a perfect crystal is not possible.
In the next section, we will introduce crystal imperfections, which in many cases lead to desirable materials properties.
Imperfections in Solids
There is no such thing as a perfect crystal. Crystalline imperfections (or defects) are always present. In addition, impurity atoms are always present. Many of the properties of materials are sensitive to the presence of imperfections, and not necessarily in an adverse way.
So, what kind of imperfections exist in solids? One way to classify imperfections is by their dimensionality. Point defects exist by definition as a point (0 – dimensional) and include vacancies, interstitial atoms, and substitutional impurity atoms. These point defects are shown in the two figures below and will be discussed further in the reading.
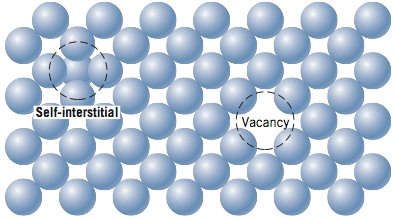
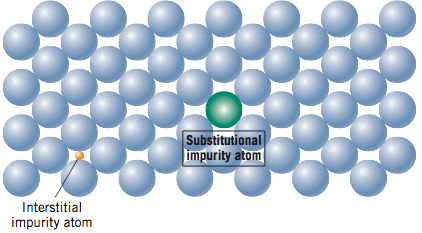
One-dimensional or linear defects are called dislocations. An edge dislocation is when a half plane of atoms disrupts the overall crystal structure. A screw dislocation is when a half twist disrupts the overall crystal structure. A mixed dislocation is a dislocation that combines both an edge and screw dislocation together.
Grain boundaries are regions between different grains within a material. They are classified as an interfacial defect and are two-dimensional.
Now proceed to the second chapter of the Lesson 5 reading assignment and complete the reading.
Reading Assignment 2
As you do the following reading, here are some questions to keep in mind.
- Is there really a 'pure' metal?
- What are point defects?
- What are edge dislocations?
- What are screw dislocations?
- Can you name a two-dimensional imperfection in a crystal?
Reading Assignment
Read pp 121-135 (Ch. 6) in Introduction to Materials ebook
Video Assignment: Metal: The Secret Life of Material
Now that you have read the text and thought about the questions I posed, go to Lesson 5 in Canvas and watch "Metal: The Secret Life of Materials" (51 minutes) about how science has unraveled the secrets of metal at the atomic level. In "Metal: The Secret Life of Materials," materials scientist Dr. Mark Miodownik explains the history, production, and uses of metals. Metals can be strong enough to build modern cities but soft enough to be crumbled in hand.
Video Assignment
Go to Lesson 5 in Canvas and watch the Metal: The Secret Life of Material video. You will be quizzed on the content of this video.
Summary and Final Tasks
Summary
Lesson 3 was concerned primarily with the various types of atomic bonding and how bonding is determined by the electron structures of the individual atoms. In this lesson, the structure of materials was discussed beginning with how metal atoms arrange to form solids. Within this framework, concepts of single crystal (highly ordered), polycrystalline (many unaligned regions of crystalline material), and non-crystalline (little to no order, also known as amorphous) materials were introduced. For crystalline solids, the notion of crystal structure was presented, and specified in terms of a unit cell.
Reminder - Complete all of the Lesson 5 tasks!
You have reached the end of Lesson 5! Double-check the to-do list on the Overview page to make sure you have completed all of the activities listed there before you begin Lesson 6.



