Lesson 8: Structure and Applications of Polymers
Overview
Although natural polymers have been used by mankind for many centuries, the use of polymers has exploded with the development of synthetic polymers within the last 100 years. Due to satisfactory properties, ease of production, and lower costs, synthetic polymers have replaced many metal, wood, rubber, and fiber parts in many materials applications. In this lesson, we look at the molecular structures of polymers and the development of numerous polymers that are synthesized from small organic molecules. Several different types of end uses of polymers in materials applications including plastics, fibers, coatings, adhesives, films, foams, and advanced materials will be discussed.
Learning Objectives
By the end of this lesson, you should be able to:
- Describe a typical polymer molecule in terms of its chain structure and, in addition, how the molecule may be generated from repeat units.
- Cite the differences in behavior and molecular structure for thermoplastic and thermosetting polymers.
- Draw repeat units for polyethylene, poly(vinyl chloride), polytetrafluoroethylene, polypropylene, and polystyrene.
- Name and briefly describe the four general types of polymer molecular structures.
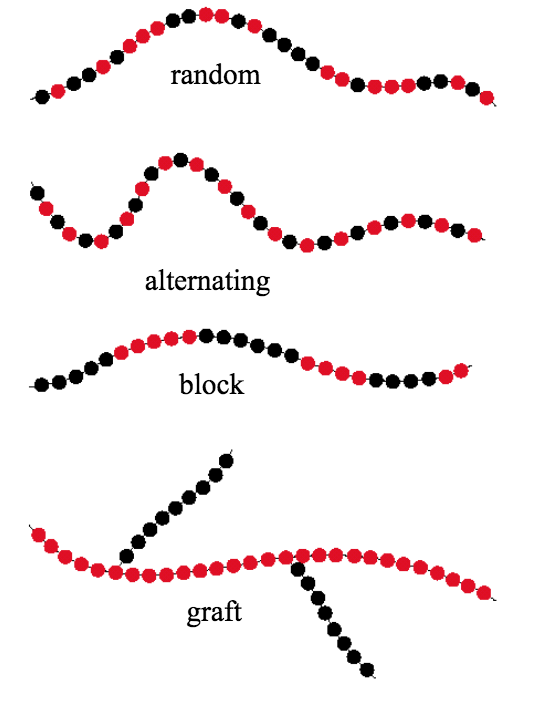
- Name and briefly describe the four types of copolymers.
- Define hydrocarbon, unsaturated hydrocarbon, saturated hydrocarbon, and isomerism.
- Cite the seven different polymer application types and note the general characteristics of each type.
Lesson Roadmap
Lesson 8 will take us 1 week to complete. Please refer to Canvas for specific due dates.
| To Read |
Read pp 215-231 (Ch. 11) in Introduction to Materials ebook Read pp 232-245 (Ch. 12) in Introduction to Materials ebook |
|---|---|
| To Watch | Plastic: The Secret Life of Materials |
| To Do | Lesson 8 Quiz |
Questions?
If you have general questions about the course content or structure, please post them to the General Questions and Discussion forum in Canvas. If your question is of a more personal nature, feel free to send a message to the instructor through Canvas email. The instructor will check daily to respond.
What is a Polymer?
In this lesson, we will introduce the structure, history of development, and properties of polymers. The roots of the word polymer are actually very descriptive of a polymer. The root ‘mer’ means unit, and poly means many. Taken together, the word polymer can be deconstructed as many units. Typically, ‘mer’ is referred to as a monomer. ‘Mono’, which is the root for one, literally translates as one 'mer'. A commonly used definition of polymer is a material that is composed of many monomers (from 10s to 1000s) all linked together to form chains. A monomer can be composed of one to many atoms which form the base unit which is repeated to form a polymer, as represented in the figure below.

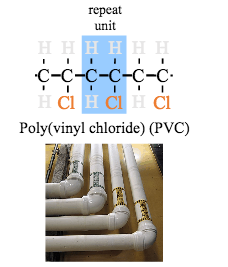
We will also study how chains of polymers are constructed. Polymers can resemble spaghetti noodles (linear), ladders (cross-linked), long chains with smaller chains hanging off the main chain (backbone) known as branched polymers, elaborately complex structures (network), or a mixture of some or all of these basic types. Other polymers, known as copolymers, are constructed from two distinctly different starting monomers and are classified as random, alternating, block, or graft polymers.
Now, watch this TED-Ed video titled, “From DNA to Silly Putty, the Diverse World of Polymers” (4:59), before proceeding on to the next section of our lesson.
To Watch
What do silk, DNA, wood, balloons, and Silly Putty all have in common? They're polymers.
Polymers are such a big part of our lives that it's virtually impossible to imagine a world without them, but what the heck are they? Polymers are large molecules made of small units called monomers linked together like the railroad cars from a train. Poly means many, and mono means one, and mers or mero means parts. Many polymers are made by repeating the same small monomer over and over again while others are made from two monomers linked in a pattern.
All living things are made of polymers. Some of the organic molecules in organisms are small and simple, having only one of a few functional groups. Others, especially those that play structural roles or store genetic information, are macromolecules. In many cases, these macromolecules are polymers. For example, complex carbohydrates are polymers of simple sugars, proteins are polymers of amino acids, and nucleic acids, DNA and RNA, which contain our genetic information, are polymers of nucleotides. Trees and plants are made of the polymer cellulose. It's the tough stuff you find in bark and stems. Feathers, fur, hair, and fingernails are made up of the protein keratin, also a polymer. It doesn't stop there. Did you know that the exoskeletons of the largest phylum in the animal kingdom, the arthropods, are made of the polymer chitin?
Polymers also form the basis for synthetic fibers, rubbers, and plastics. All synthetic polymers are derived from petroleum oil and manufactured through chemical reactions. The two most common types of reactions used to make polymers are addition reactions and condensation reactions. In addition reactions, monomers simply add together to form the polymer. The process starts with a free radical, a species with an unpaired electron. The free radical attacks and breaks the bonds to form new bonds. This process repeats over and over to create a long-chained polymer. In condensation reactions, a small molecule, such as water, is produced with each chain-extending reaction.
The first synthetic polymers were created by accident as by-products of various chemical reactions. Thinking they were useless, chemists mostly discarded them. Finally, one named Leo Baekeland decided maybe his useless by-product wasn't so useless after all. His work resulted in a plastic that could be permanently squished into a shape using pressure and high temperatures. Since the name of this plastic, polyoxybenzylmethylenglycolanhydride, wasn't very catchy, advertisers called it Bakelite. Bakelite was made into telephones, children's toys, and insulators for electrical devices. With its development in 1907, the plastics industry exploded.
One other familiar polymer, Silly Putty, was also invented by accident. During World War II, the United States was in desperate need of synthetic rubber to support the military. A team of chemists at General Electric attempted to create one but ended up with a gooey, soft putty. It wasn't a good rubber substitute, but it did have one strange quality: it appeared to be extremely bouncy. Silly Putty was born!
Synthetic polymers have changed the world. Think about it. Could you imagine getting through a single day without using plastic? But polymers aren't all good. Styrofoam, for example, is made mainly of styrene, which has been identified as a possible carcinogen by the Environmental Protection Agency. As Styrofoam products are being made, or as they slowly deteriorate in landfills or the ocean, they can release toxic styrene into the environment. In addition, plastics that are created by addition polymerization reactions, like Styrofoam, plastic bags, and PVC, are built to be durable and food-safe, but that means that they don't break down in the environment. Millions of tons of plastics are dumped into landfills every year. This plastic doesn't biodegrade, it just breaks down into smaller and smaller pieces, affecting marine life and eventually making their way back to humans.
Polymers can be soft or hard, squishy or solid, fragile or strong. The huge variation between means they can form an incredibly diverse array of substances, from DNA to nylon stockings. Polymers are so useful that we've grown to depend on them every day. But some are littering our oceans, cities, and waterways with effects on our health that we're only beginning to understand.
Natural Polymers
DNA (deoxyribonucleic acid), proteins, sugar, starches, and carbohydrates are some examples of natural polymers used by plants and animals. The corresponding monomers for these polymers are listed in the table below. In addition to these important to life polymers, natural polymers derived from plants and animals have been used by humans for many centuries. These include wood, cotton, leather, rubber, wool, and silk. One of the oldest known uses of polymers is depicted in the picture below. The Incas of South America used rubber balls in some of their competitions. In the next sections, we will begin to discuss human-made polymers known as plastics.
| Polymer | Monomer |
|---|---|
| DNA (deoxyribonucleic acid) | nucleotides |
| Proteins | amino acids |
| Sugar, Starches, Carbohydrates | glucose |

Plastics
Plastics are polymers derived from petroleum products. They are a perfect example of designing better, cheaper, and completely human-made materials. Plastics are inspired by nature, i.e., natural polymers, but are completely synthetic.
Most polymers are made up of carbon and hydrogen atoms, and many plastics are as well. Polymers that contain hydrogen and carbon atoms are called hydrocarbons. Carbon atoms can form single bonds to four other atoms. If the carbon atoms in a polymer are bound to four other atoms the polymer is referred to as a saturated hydrocarbon. If on the other hand, the carbon atom is not bound to four other atoms it will typically form double or triple bonds, as needed, with another carbon atom. In this case, the polymer is referred to as an unsaturated hydrocarbon. This distinction is important as unsaturated polymers are generally unstable and more reactive than their saturated cousins.
Around 1850 billiards was becoming increasingly popular, but there was a problem. The balls were made of ivory, which is in very limited supply and is, thus, very expensive. Not to mention it requires the killing of elephants to obtain. In 1856, the first human-made plastic (Parkesine) was patented by Alexander Parkes from Birmingham, England. Often called synthetic ivory it was composed of nitrocellulose – cellulose treated with nitric acid and a solvent. It was the first thermoplastic, but it failed as a commercial product due to poor product quality control. The following 10-minute video discusses the development of polymers to replace ivory billiard balls, the science behind some of the most-used plastics, and some examples of thermoplastic and thermosetting polymers.
To Watch
Charles Darwin was a big fan of billiards. He loved his billiard table. It was one of his prized possessions and one of the most valuable things he owned. Well - not the actual table itself. In fact, it was the balls that were so valuable. Pure ivory carved from the tusks of elephants; only the wealthiest could afford a full set. Luckily, he was married to an heiress.
A full set of billiard balls would require at least one, possibly two, full elephants worth of ivory. The idea that any bar in the world might contain a billiard table available for anyone to play, and not like run out of the bar with your pockets full of valuable ivory would have sounded insane. And the billiard industry was well aware of this problem. Billiard balls were getting more expensive, elephants were getting rarer. It was thus not with an environmental motive, that in the Phelan and Collender Pool Supply Company offered $, to anyone who could come up with a substitute material that worked as well as ivory, but could be produced more quickly and sustainably then dead elephants.
An inventor named John Wesley Hyatt took on that challenge. He used nitrocellulose, a flammable solid, created by mixing cotton with nitric acid to create a hard, shiny, white sphere. The properties were extremely similar to ivory billiard balls. The company never gave him the prize. But he did patent the technique, using it to create billiard balls, piano keys and even teeth, becoming pretty dang wealthy in the process. Also, he pretty much created the industry that made all of the polymer materials that surround you right now, and that we'll be discussing today in Crash Course Chemistry. Also elephants didn't go extinct, so that's a plus.
[Theme Music]
The polymer that John Hyatt worked on was, somewhat unsurprisingly, kinda crummy. It worked well once it was created, but the manufacturing process was dangerous because nitrated cellulose can explode in a warm breeze. So luckily, some replacements started creeping in. Replacements with some names you probably recognize, like polyvinyl chloride or PVC, bakelite, polystyrene, polyester, and nylon.
These are all polymers; huge chains or sometimes D networks of repeating organic units called monomers. Each polymer has a monomer, but they're all relatively simple at that basic one-unit level. The trick is that they bond to each other on each side potentially forever, though in reality the chains are sometimes hundreds, sometimes thousands, sometimes hundreds of thousands of units long. In order to make a polymer all you need is a molecule that can easily bond to another identical molecule at points. And the simplest of those is ethene, also known as ethylene.
It's polymer, you'll be unsurprised to hear is polyethylene, which you've probably heard about. We’ll talk more about the specifics in a second, but basically because pi bounds in the double bond are weaker than sigma bonds they can be broken and new monomers can be added. Just to note to avoid confusion polyethylene has that "-ene" sound in it, right
but it's not an alkene because all those double bonds get broken to form new sigma bonds. It's a polymerized alkene, but the molecule itself is an alkane. It's confusing so I thought it's worth pointing out. Now chemists might want a bunch of different things out of their polymers; maybe they want it to be stretchy, maybe strong, maybe transparent, maybe recyclable. Polyethylene is transparent and thermoplastic, meaning it can be melted and reformed. Making it recyclable.
Some other polymers like polyurethane or Bakelite are thermoset. Which means that they change chemically during some kind of curing process and cannot be melted down and reformed. Polyethylene can actually be converted into a thermoset polymer by introducing cross-links, basically molecular bridges between those polymer chains. Any plumbers out there probably have heard of cross-linked ethylene or PEX pipe. Which is what this is. It's extra, super strong because of those cross-links. Polyethylene is also nice because it's strength can be varied by changing the size of the molecules. If they are allowed to polymerize until they are tens of thousands of monomers long, the plastic they will form will be all knotted up in these ultra-long chains and it will be extremely strong. That's why this HDPE, high density polyethylene, is a strong bottle. Whereas this is much squishier, this is low density polyethylene. However those ultra-long chains also make it much more viscous when heated and thus more difficult to process. It also loses some of its opacity and becomes more of that milky white color. Now polyethylene is great. It's really great. So great that it's the most common plastic in the world. We produce over a million tons of it per year. But we want a lot out of our plastics - strength, color, elasticity, resilience, recyclability - we need everything from saran wrap to car tires. All of these are traits that chemists work tirelessly to create in the early to middle 20th century and continue to work on even today.
One of the earliest techniques they used to try and bring out new properties was to change the substituents on the ethylene monomer. Just, see what would happen. Like, what if we swapped out one of the hydrogens for chlorine? Well you get polychloroethene, kinda, that's not what we call it.
OK, so remember how benzene when attached to a chain is a phenyl group, and how those two words have nothing to do with each other? Well the same thing goes for the ethene functional group, which is called a vinyl group. It's an old word, super old, it comes from the Greek word for wine. And that is why chloroethene is more commonly called vinyl chloride. And polychloroethene is more commonly called polyvinyl chloride, or PVC. Which is what this little ducky is made out of and also what records are made out of, which is why we call them vinyl.
Now what happens when we change out a hydrogen for a methyl group?
Well then suddenly this molecule is a propene or, if you are using ye old ways, a propylene. And, yes, if you polymerize it, it becomes polypropylene. If one of the hydrogens is replaced with a phenyl group, well that chemical was first derived from trees in the Styrax family, so it's called styrene. Polymerize it, polystyrene. Make a foam out of it, Styrofoam. Now if you change all four of the hydrogens on the base ethylene with fluorine it becomes tetrafluoroethylene. Polymerize that and instead of hydrogens that polymer is bound entirely to fluorines. Fluorine as we could guess from its spot on the periodic table, love electrons. It is extremely electronegative. But because it holds onto its electrons so tightly, and is so satisfied in this polymerized chain, the electrons are unavailable for even minimal interactions with any other molecules. I'm not just saying this stuff is super difficult to difficult to react with, or it's really stable. It's more than that the electrons aren't even available for the sort of interactions that make things stick to each other, or cause friction. Which is why you have heard of polymerized tetrafluoroethylene, because it's super useful, either by its abbreviation, PTFE, or by its brand name Teflon.
So how do we actually make these things? Well, ethene based polymers form through a process called addition polymerization. The monomers are simply added together and no by products are formed. In order to get the process kicked off you have to introduce a free radical. To me, that always sounded like some crazy freedom-fighter diving into battle without much thought for what would come after he was consumed in the firefight. And, that's kinda what they are. Free radicals are atoms or ions that have a single unpaired electron. This is crazy unstable. It's basically like having half of covalent bond dangling off the atom. Anywhere this can form a bond, it's going to form a bond. And, in the case of addition polymerization it attacks the double bond and joins one of the carbons, while the other carbon is itself left with an unpaired electron.
The molecule itself is now a newly formed free radical, and it attack another nearby pi bond, joining with another molecule of ethene, forming another radical. This process continues until two radicals meet each other consuming both free radicals without producing any more, thus, ending polymerization. There are, of course, other sorts of polymerization, as well. Sometimes a hydroxyl group from one molecule is happy to join up with a hydrogen from another, forming water. The water will break away as a byproduct, leaving the two molecules bound together. This often occurs when an amine group, with its loosely held hydrogen, meets a carboxylic acid, with its loosely held -OH group. This is just what happens when hexamethylenediamine meets adipic acid forming another branded polymer, nylon. By dissolving hexamethylenediamine and adipic acid into two different immiscible, or un-mixable, solvents, we actually create nylon right here. The nylon forms at the interface between the two immiscible liquids. And, we can literally grab it and pull it out of the vial, twisting and spooling it until we get a nice glob of nylon. This works because hexamethylenediamine has an amine group on each end and adipic acid has a carboxylic acid on both ends. Thus, when the two monomer unit, called a dimer, is formed, there is still a carboxylic acid on one end and an amine group on the other, allowing for further polymerization. These amine acid condensation polymerizations also allow for the creation of possibly the most important polymers on the planet; natural polymers being created inside of you right now out of monomers that we call amino acids.
Did you see that one coming?
Amino acids polymerize through condensation reactions guide by the code in your DNA and some very complicated enzymes to form basically you. Other important polymers in your body include polysaccharides, which we use to store energy; and yeah DNA and RNA, which we use to encode information for the formation of proteins.
But that would be back to biology, which is a whole other Crash Course. Which to be clear is available if you'd like to watch it.
And thank you for watching this episode of Crash Course Chemistry, in it you learned: that the first commercial polymer ever saved the lives of a lot of elephants. That ethene is sometimes called ethylene. And, that a huge variety of polymers is based on the addition reaction of ethene based monomers, including Teflon. Which so friction-less because of fluorine's extreme electronegativity. You also learned how addition polymerization reactions work. And, that other polymers are formed by condensation reactions. Including the polymerization of amino acids monomers, which along with other polymers like DNA and RNA, and glycogen, make up a lot of the stuff that is you.
This episode of Crash Course was written by me, Hank Green, edited by Blake de Pastino, and our chemistry consultant was Dr. Heiko Langner. It was filmed, edited and directed by Nicholas Jenkins. Our script supervisor was Stefan Chin. Our sound designer was Michael Aranda. And, our graphics team is Thought Café.
After watching this video, please proceed to the first (of two) reading assignments for this lesson.
Reading Assignment 1
Things to consider...
As you do the first reading for this lesson, use the following questions to guide your learning. Remember to keep the learning objectives listed on the overview page for this lesson in mind as you learn from this text.
- How is the basic structure of a polymer made up of monomers?
- What are the different chain structures and how do they define polymer behavior?
- What are the four general polymer molecular structures?
- How important are organic polymers, in particular hydrocarbons?
- What are the differences in behavior and structure for thermoplastic and thermosetting polymers?
- What determines whether a polymer material is a good or bad candidate for recyclability or repurposing?
Reading Assignment
Read pp 215-231 (Ch. 11) in Introduction to Materials ebook
Polymer Formation
Polymers are formed by two main ways called addition and condensation polymerization. In addition, polymerization, an initiator (or catalyst) reacts with a starting monomer. The result of this initiation reaction is a monomer attached to the initiator with an unsatisfied bond. The unsatisfied bond is free to react with another monomer, thus adding to the chain. The process repeats over and over again until two chains combine or another initiator binds to the end of the chain, both of which will terminate the chain. In condensation polymerization, a monomer with an exposed H (hydrogen) atom binds with a monomer with exposed OH (oxygen-hydrogen) atoms. During the reaction, water is released (compensated) as the H and OH combine to form H2O (water). The following 4-minute video discusses addition and condensation polymerization.
To Watch
In this video, you will learn how condensation polymers form, some examples of condensation polymers and the uses of these polymers.
Unlike addition polymers where monomers react to form a single product, in a condensation polymerization reaction not only is the polymer formed but also a small molecule is eliminated or lost, normally water. Polyesters and polyamides are the two types of condensation polymer we will look at.
We will first look at polyamides. This is known as the amide link. It is formed when a carboxylic acid reacts with an amine. In the formation of nylon 6,6, we react a molecule with an amine group on each side known as hexane-1, 6-diamine and a molecule with a carboxylic acid at each end, hexanedioic acid. Since both these molecules have long chain carbons, they are only complicated the visual structure. Let us remove these and replace them with rectangular blocks. You can now see the functional groups at the ends of each molecule. A carboxylic acid and amine group positioned next to each other. These groups react to form water, which is eliminated. And a large polymer molecule is formed, held together by amide links. Hence the name Polyamide. Nylon has many uses in the textile industry but this is also structurally sound material used in engineering. Especially where low friction is required, such as in bearings or bushes.
We can now look at polyesters similarly. An ester is formed in the reaction of an alcohol with a carboxylic acid. In this example, we will show the formation of polyethylene terephthalate, more commonly known as PET. Using the carboxylic acid Benzene-1, 4-dicarboxylic acid and the alcohol ethane-1, 2-diol. Once again since those these molecules have a long carbon chain that may confuse the overall structure we will replace them with rectangular blocks. As you can see when we align the two molecules, a carboxylic acid and alcohol group can react between the molecules causing Ester links to hold the large polymer molecule together. And once again water is released. PET is commonly used as a plastic for drinks bottles and polyester is used to produce fabric for clothing.
Now at the end of this tutorial, you should understand what condensation polymers are and be able to give examples of polyesters and polyamides along with their uses.
Now that we have reviewed how polymers are formed, let’s discuss one of the possible ways to classify polymers, as thermoplastic or thermosetting.
Thermoplastic and Thermosetting Polymers
In the last lesson on ceramics, we saw that one way to classify ceramics is by their uses (refractories, glass, clay products, abrasives, etc.). Other possible classification categories might include crystal structure and whether they are crystalline or non-crystalline. For polymers, one useful classification is whether they are thermoplastic or thermosetting polymers. As you read in the last reading assignment, thermoplastics soften when heated and harden when cooled. This is totally reversible and repeatable. Most linear polymers and branched structure polymers with flexible chains are thermoplastics. This is in contrast to thermosetting polymers, which do not soften when heated due to strong covalent crosslinks. Thermoset polymers are generally harder and stronger than thermoplastics and have better dimensional stability.
To Watch
For more information about thermoplastic (here referred to as thermo-softening) and thermosetting polymers watch this video (4:40):
The term polymer is used to describe a macromolecule made of many monomers or repeating units. The properties of these polymers all depend on a variety of factors. The monomer unit, the linkages between each monomer, and the intermolecular and intramolecular forces that exist between polymers. In this lesson, we will learn about two classes of polymers thermo-softening polymers and thermosetting polymers. We will also learn about their properties and how these properties arise. The term plastics is used to describe a wide range of polymers made of monomers all derived from the products obtained from the fractional distillation of crude oil. You may be familiar with polyethylene, polypropylene, and even polyvinylchloride. You can learn about the structure of these polymers, how they are made, and their real-life applications from other videos on our channel. Here we will focus on how these polymers respond to heat and why they respond the way they do.
Polyethylene, polypropylene, and polyvinyl chloride our thermo-softening polymers. This means that they soften when heated. When soft and in liquid form they can be molded into many different shapes. These plastics are used to make many everyday items such as window and door frames, pipes, wiring insulation, and waterproof clothing items just to name a few. This is made possible because polymers are not linked together. We can think of it like a bowl of noodles. Although the noodles are coiled and tangle with one another they are not linked. Like the noodles, these polymers can slide over one another making these items made from them soft and flexible. In fact, these polymers can only interact by weak intermolecular forces and can, therefore, be separated rather easily when heated giving them relatively low melting points. Some other thermo-softening polymers include polystyrene and polytetrafluoroethylene. Thermosetting polymers, on the other hand, do not soften when heated. Unlike thermo-softening polymers these thermosetting polymers are cross-linked to one another can you think about how this might affect the properties of these polymers? Pause, think, and continue when ready.
The presence of crosslinks hardens the overall structure. A good example of a thermosetting polymer is vulcanized rubber. Rubber tapped from para rubber trees is a polymer of isoprene monomers. It is a runny liquid that can be processed to make latex gloves, erasers, and party balloons. It can also be used to make car and bicycle tires though it has to be vulcanized first. For the vulcanization process, sulfur is added so the disulfide bridges link the polymers together. The presence of these cross-linkages greatly increases its strength and therefore does not soften easily when heated. Let's think about it. No matter how fast you ride your bike the tires do not change shape. Some other examples of thermosetting polymers include a substance used to make old TV sets and certain types of strong glue.
In summary, thermo-softening plastics are soft and melt when heated, whereas thermosetting plastics are hard and do not soften or change their shape when heated.
Now that we have discussed thermoplastic and thermosetting polymers let us review the different basic structures that polymers form and how that structure can determine whether the polymers are thermoplastic or thermosetting.
Basic Polymer Structure
There are four basic polymer structures which are shown in the figure below. In practice, some polymers might contain a mixture of the various basic structures. The four basic polymer structures are linear, branched, crosslinked, and networked.
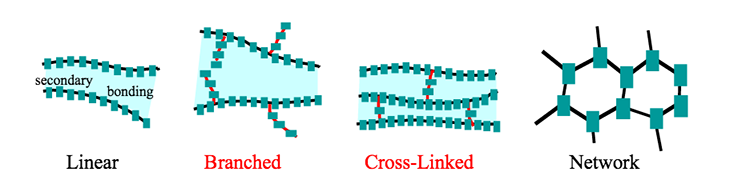
Linear polymers resemble ‘spaghetti’ with long chains. The long chains are typically held together by the weaker van der Waals or hydrogen bonding. Since these bonding types are relatively easy to break with heat, linear polymers are typically thermoplastic. Heat breaks the bonds between the long chains allowing the chains to flow past each other, allowing the material to be remolded. Upon cooling the bonds between the long chains reform, i.e., the polymer hardens.
Branched polymers resemble linear polymers with the addition of shorter chains hanging from the spaghetti backbone. Since these shorter chains can interfere with efficient packing of the polymers, branched polymers tend to be less dense than similar linear polymers. Since the short chains do not bridge from one longer backbone to another, heat will typically break the bonds between the branched polymer chains and allow the polymer to be a thermoplastic, although there are some very complex branched polymers that resist this ‘melting’ and thus break up (becoming hard in the process) before softening, i.e., they are thermosetting.
Crosslinked polymers resemble ladders. The chains link from one backbone to another. So, unlike linear polymers which are held together by weaker van der Waals forces, crosslinked polymers are tied together via covalent bonding. This much stronger bond makes most crosslinked polymers thermosetting, with only a few exceptions to the rule: crosslinked polymers that happen to break their crosslinks at relatively low temperatures.
Networked polymers are complex polymers that are heavily linked to form a complex network of three-dimensional linkages. These polymers are nearly impossible to soften when heating without degrading the underlying polymer structure and are thus thermosetting polymers.
Monomers do not have to be of a single atom type, but when referring to a specific monomer it is understood to be of the same composition structure. When building a polymer from two distinct monomers, those polymers are referred to as copolymers. Next, we will look at how copolymers are classified.
Copolymers
If a chemist is synthesizing a polymer utilizing two distinct starting monomers there are several possible structures, as shown in the figure below. The four basic structures are random, alternating, block, and graft. If the two monomers are randomly ordered then the copolymer is, not surprisingly, referred to as a random copolymer. In an alternating copolymer, each monomer is alternated with the other to form an ABABABA… pattern. In block copolymers, more complex repeating structures are possible, for example AAABBBAAABBBAAA… Graft copolymers are created by attaching chains of a second type of monomer on the backbone chain of a first monomer type.
Before we move on to the many uses of polymers, watch this four-minute video which will introduce the uses of polymers.
To Watch
In our previous videos we have explored how polymers are formed and equations for polymerization reactions. In this video, we'll explore in more detail some different polymers and their specific uses as well as the problems associated with polymers. As you now know, polymers are a long chain of organic molecules made by repeating monetary units. There are a number of natural polymers in life such as rubber, and even in our own body we have natural polymers such as proteins, carbohydrates, and DNA to name just a few. We'll focus the rest of this tutorial on synthetic polymers. The common name to synthetically made polymers is plastics which are used very frequently in our day-to-day lives. From simple packaging to complex structural building materials. However, the increased use of plastics in our homes leads to nearly one-quarter of all the solid waste being plastic. Some of this can be recycled to minimize the effects on our environment. It's a long-term goal for many chemists is to develop more biodegradable plastics which would naturally break down in our environment. Here are some specific examples of polymers and their common uses.
Polythene used for carrier bags and plastics. High-density polyethylene is used for drain pipes, water bottles, and containers. Polystyrene is used in packaging. Polypropylene is used for bottle caps, plastic bottles, and plastic pipes. Polychloroprene Etaene, often known as PVC, is used for windows and door frames, plastic hinges, and bottles. Polly 1122 tetrafluoroethylene also known as PTFE, which is a nonstick coating on frying pans as well as being used in bearings another low friction surfaces. Kevlar is a unique polymer in that it is used for bulletproof vests and jackets. Nylon is used in textiles, clothing, and carpets. As you can see polymers play a huge role in our day-to-day lives and their use is wide and varied owing to their unique individual properties. It is important to understand the most of the alkene monomers used to make polymers are obtained in some part from crude oil and therefore, it is critical that we recycle plastics to conserve our natural resources for the future manufacture of these polymers.
There are also big problems associated with the disposal of polymers. The biggest problem as mentioned above is that polymers are non-biodegradable which means that microorganisms cannot naturally break them down. Disposal of polymers by burning or incineration is a possibility, as this generates heat which can be used to generate electricity. However, the burning of polymers produces many toxic gasses which themselves can damage the environment and cause pollution.
Now at the end of this lesson, you should have an appreciation of the importance of polymers, be able to name some key polymers along with their uses, and also describe the problems associated with polymers.
Now that you have watched this video, please proceed to the second (of two) reading assignments for this lesson.
Reading Assignment 2
Things to consider...
As you read the second chapter for this lesson, use the following question to guide your learning. Remember to keep the learning objectives listed on the overview page for this lesson in mind as you learn from this text.
- Polymers can be used in a wide variety of applications, what are the seven different polymer application types?
Reading Assignment
Read pp 232-245 (Ch. 12) in Introduction to Materials ebook
What are Scientists Doing Now to Improve Polymers?
Now that you have read about the classical usages of polymers let us take a look at two short videos that discuss two areas that scientists are working in to improve or increase the usage of polymers in our daily lives.
To Watch
The first is a video about designer polymers (3:45).
Polymers have been around for a long time. Some of the commonly named examples are found in clothes. Things like nylon, polyesters, and acrylic. Others are plastics like PVC, polyethylene, and polycarbonates. Whereas some act as coatings on saucepans like PTFE, more commonly known as Teflon. The key thing is that different polymers have different properties.
More recently, chemists have developed a branch of polymers called designer polymers. A designer polymer is one that has been designed to respond to a change in environment or uses properties that are better than traditional polymers. Nylon, a traditional polymer used to make some clothes, has desirable properties he may even wear a jacket that's made from nylon. Nylon is tough lightweight and waterproof sadly it's missing a desirable property in that it doesn't allow sweat to pass through so when the person is wearing a Garmin they can become quite uncomfortable. If only there was a way of making a material breathable able to allow the sweat to pass through without losing the waterproofing on the outside of the jacket. The answer is that designers have started to use gore-tex a design a polymer. Gore-tex uses layers of different polymers they include an outer layer typically made from nylon or polyester. This makes the outer layer strong inner layers are made from polyurethane and miss provides water resistance.
Other membranes are made of PTFE which has millions of holes. These holes are small enough to allow the water vapor or sweat to pass out but does not allow larger water droplets from the outside to pass into the soft lining. Designer polymers come up in many everyday situations. Contact lenses, the traditional polymer PMMA, did not allow oxygen to pass through and touch the eye. This is because no blood visits the cornea this would block your vision. Therefore, all of the oxygen needed for the cells comes from the air. It was rigid and uncomfortable and the cells were starved of oxygen. How do you think the design of this polymer was improved? Pause and continue when you're ready.
The answer is the polymer used now is a special hydrogel. It's more flexible, softer, and is breathable. This improves the health of the eye to fillings. If you have a traditional filling the chances are it is made of silver amalgam this looks false as it contrasts against the tubes natural color. Designer polymers use a composite polymer resin which is tough, contains no dangerous chemicals like the mercury metal found in your tradition of silver amalgam fillings. The designer polymer can be matched at the tooth's natural color it's a photopolymer and when treated with lights will harden and match the color of the tooth. In summary, a designer polymer is one that has been designed to respond to a change in environment or uses properties that are better than the traditional polymers.
Some examples of these minor polymers include breathable clothing made from gore-tex, hydrogels found in contact lenses, in babies nappies, and finally tooth fillings.
To Watch
The second video (4:28) is about research into how to make flexible and lightweight electronics.
Plastic electronics are electronic devices in which the active components are made out of carbon-based materials. So these are plastics, or polymers, or small molecules and the reason you want to make plastic electronics is because you want to make use of the attributes of these plastic materials. These include their mechanical flexibility, they're lightweight, they can be produced with tunable properties, and this is something you can't easily do with inorganic materials. My name is Yu Lin Lieu and I work in the field of plastic electronics.
In the field of plastic electronics, it all starts with chemistry. We need to make or synthesize new materials that are conductive or semiconductive, so they have the electrical properties that we would like so that when we incorporate them into electronic devices they're active. So in our group, some of the researchers make new materials, some of the researchers characterize the structures are these materials, and some other incorporate these materials to understand their potential in applications like transistors and solar cells.
Polyaniline is a conducting polymer that changes color. In here, this color change is triggered by applying a voltage to the sample. So the potential applications for polyaniline, in addition to being electrodes, we can use it as electrochromic displays, as well as sensors that change color when exposed to a specific chemical or reagent.
We use a process called spin coating to make thin layers of these compounds. The layers end up to be about a hundred nanometers thick that's about a thousand times thinner than my hair.
Here we examine the films we make under the microscope to see how the crystals grow during spin coating. We try to control the size of the crystals in the film. The bigger the crystals, the better the devices will turn out.
To make devices, we have to make electrical contact to the film by evaporating gold. Gold is evaporated through a mask. The pattern of the mask determines where gold is cooled. After the placement of a mask, we put the sample in the gold evaporator. Alternatively, we can evaporate gold electrodes on a clear silicone rubber-stamp, and laminate the rubber stamp onto the polymer film to make our devices. The structure of the devices depends on their function. In my opinion, their beauty derives from their functionality. Compared to inorganics like silicon, classics had unique attributes which include their lightweightness and their mechanical flexibility, their potential in their tunability in terms of their properties and soon to incorporate all these attributes into electronic devices would be really nice.
Well, the field's really exciting because it's a young field and it's growing and it's directly tied to applications that can have direct implications on the quality of our lives. Imagine electronic wallpaper that changes patterns from green stripes to pink polka dots at a click of a switch. Imagine tinted windows that can also generate power during the day. Imagine disposable sensors that would change color if the water sources contaminated or yet think of smart plastic patches that can monitor your health and deliver medication when you're sick. The possibilities are endless.
Now that you have finished these videos please proceed to the next page of our course which will introduce the video for this lesson, Plastics: The Secret Life of Materials. This video will tie together the history, concepts, and usages of polymers that we have been discussing in this lesson, as well as, highlight some possible future usages of plastic.
Video Assignment
Now that you have read the text and thought about the questions I posed, go to Lesson 8 in Canvas and watch "Plastic: The Secret Life of Materials" (51 minutes) about the manmade and artificial materials which have changed how we live. In "Plastic: The Secret Life of Materials," materials scientist Dr. Mark Miodownik explains how we have turned our backs on nature and began to create our own better and cheaper materials.
Video Assignment
Go to Lesson 8 in Canvas and watch the Plastic: The Secret Life of Material video. You will be quizzed on the content of this video.
Summary and Final Tasks
Summary
Polymers are composed of repeating units which are repeated in four possible chain structures: linear, branched, crosslinked, and network. In this lesson, we discussed how the chemical and structural characteristics affect the properties and behavior of polymers. The seven basic end uses for polymers (plastics, fibers, coatings, adhesives, films, foams, and advanced materials) were introduced. Most polymers are not biodegradable which coupled with their heavy use in today’s society leads to a major source of waste at the end of a polymer's usage.
In the next lesson, we will look at how composites are formed from two or more distinct materials to achieve the best of two worlds.
Reminder - Complete all of the Lesson 8 tasks!
You have reached the end of Lesson 8! Double-check the to-do list on the Overview page to make sure you have completed all of the activities listed there before you begin Lesson 9.