Lesson 9: Mineral Reactions in One-Dimension Systems
Overview
In this lesson we use a mineral reaction example to illustrate 1) how physical processes and geochemical processes are coupled in the short and long time scales, and 2) how to set up physical and geochemical processes in a 1D column in Crunchflow. The set up combines mineral dissolution (and precipitation) and physical flow processes in previous lessons. Please review these two lessons if needed.
Learning Outcomes
By the end of this lesson, you should be able to:
- Understand the importance of mineral dissolution and chemical weathering;
- Understand the fundamental principles and key parameters that govern chemical weathering and reactive transport process coupling in general;
- Set up simulations in CrunchFlow with flow, solute transport, and mineral dissolution and precipitation reaction.
Lesson Roadmap
| References (optional) | Review reading materials and examples on transport in 1D and mineral dissolution and precipitation |
|---|---|
| To Do |
|
9.1 Overview
Mineral dissolution and precipitation occur ubiquitously in natural systems. As minerals dissolve, chemicals in rocks are released into water, reducing solid mass and increasing aqueous concentrations. Mineral dissolution also opens up porosity for water storage. The opposite occurs when mineral precipitates and clog pore spaces. In the deep subsurface where we extract energy (e.g., oil, gas, geothermal energy) from, mineral dissolution and precipitation change the geochemical surface and water-conducting capacity, therefore regulating the energy extraction processes. Over geological times, the chemical and physical property alteration transforms rocks into soils and is called chemical weathering. Chemical weathering, together with physical weathering (erosion), shapes the Earth’s surface. Chemical weathering consumes carbon dioxide and locks them into carbonate minerals, which regulates the atmospheric CO2 level and sustain relatively clement earth conditions (Kump et al., 2000).
9.2 An example of mineral reactions in a 1D column
Example 9.1
A bedrock column is 10.0 meter deep with a porosity of 0.20. Mineral composition is simple with 80% v/v of quartz and 20% v/v of K-Feldspar. As the rainwater infiltrates through the rock, these primary minerals dissolve whereas a secondary mineral kaolinite precipitates, as listed in Table 1.
Conditions
The annual net runoff (precipitation – evapotranspiration) is 0.5 meter/year. Although we know that not all runoff infiltrates through the bedrock, for simplification here we assume the runoff flows through the bedrock and exits from the bottom of the bedrock. Assume the diffusion coefficients of all species are 1.0E-10 m2/s, and dispersivity is 0.1 m. The cementation factors are the default values of 1.0.
Let’s assume the initial pore water composition (all concentrations in molal): pH 7.0, Cl- (charge balance), K+ (4.0E-5), Na+ (4.0E-5), Al3+ (4.0E-6), HCO3- (1.0E-5), and SiO2(aq) (8.0E-5). One of the major drivers of chemical weathering is acidity. The acidity in soils comes primarily from the soil CO2 gas from root respiration and organic acids from root exudates. Here we set the total HCO3- concentration (equivalent to total inorganic carbon (TIC)) in the rainwater to 0.3 mM, which is equilibrated to soil gas CO2 of 9000 ppmv.
Question:
Assume chemical weathering occurs for 0.1 million years. Please set up the simulation in Crunchflow. Plot the profiles of pH, Saturation index $(\mathrm{SI}=[\log10(\mathrm{IAP}/\mathrm{K_{eq}})])$, $\phi$ ( volume of mineral/ volume of total porous media) of minerals and $\tau$ values of K and Si (relative to Zr in this soil column, we will explain later) at t = 0, 5,000, 10,000, 20,000, 40,000, and 100,000 years. How does the chemical composition and physical properties (e.g., porosity, permeability) evolve over time?
Note:
Setting up this lesson will involve what you learned from the lesson on mineral dissolution and precipitation, and the lesson on 1D transport. If needed please revisit these two lessons.
| Primary mineral dissolution | log10Keq |
log10k (mol/m2/s) |
Specific surface Area (m2/g) |
|---|---|---|---|
|
$\mathrm{SiO}_{2(\mathrm{s})}\text{(quartz)}\ \leftrightarrow\mathrm{\ SiO}_2\text{ (aq) }$ |
-4.00 | -13.39 | 0.01 |
| $\begin{array}{l} \mathrm{K} \mathrm{AlSi}_{3} \mathrm{O}_{8} \text { (K-Feldspar) }+4 \mathrm{H}^{+} \leftrightarrow \\ \mathrm{Al}^{3+}+\mathrm{K}^{+}+2 \mathrm{H}_{2} \mathrm{O}+3 \mathrm{SiO}_{2}(\mathrm{aq}) \end{array}$ |
-0.28 |
-13.00 -9.80aH+0.5 -10.15aOH-0.54 |
0.01 |
| Secondary mineral precipitation | |||
| $\begin{array}{l} \mathrm{Al}_{2} \mathrm{Si}_{2} \mathrm{O}_{5}(\mathrm{OH})_{4} \text { (Kaolinite) }+6 \mathrm{H}^{+} \leftrightarrow \\ 2 \mathrm{Al}^{3+}+2 \mathrm{SiO}_{2} \text { (aq) }+5 \mathrm{H}_{2} \mathrm{O} \end{array}$ |
6.81 | -13.00 | 0.01 |
*: Thermodynamic and kinetic parameters from EQ3/6 database [Wolery, 1992]. The three different rate constant values indicate the rate constants for neutral, acidic and basic kinetic pathways, respectively.
9.3 Governing Equations
A general governing equation for the mass conservation of a chemical species for a primary species i is the following:
where $\phi$ is porosity, Ci is the concentration (mol/m3), Dh is the hydrodynamic dispersion (m2/s), v is the flow velocity (m/s), ri,tot is the summation of rates of multiple reactions that the species i is involved (mol/ m3/s), and Np is the total number of primary species. Here we notice that this equation is very similar to the advection-dispersion equation that we discussed in previous lessons except for two major differences. One is that it has an additional reaction term ri,tot to take into account sources and sinks coming from mineral dissolution and precipitation. The other difference is that this is written for the species i, which is one of the Np primary species. Note that if the species is K+, the ri,tot in Equation (3) will be the summation of the K-feldspar reaction rate rK-feldspar and Kaolinite reaction rate rKaolinite. This is because K+ is involved in both of the kinetic reactions, the K-feldspar and Kaolinite reactions (either dissolution or precipitation). The mineral reaction rate laws follow the Transition State Theory (TST):
Where kH2O, kH, and kOH are the reaction rate constants with values of 10-13.0 mol/m2/s, 10-9.80 mol/m2/s under acidic conditions, and is the reaction rate constant of 10-10.15 mol/m2/s under alkaline conditions as shown, respectively (Table 1). The exponents of the H+ and OH- activities indicate the extent of rate dependence on H+ and OH-. A is the reactive mineral surface area (m2/m3 porous media). For kaolinite we have
Where kH2O is the reaction rate constant of 10-13.0 mol/m2/s as shown in Table 1.
In the chemical weathering context, K-feldspar dissolves and releases Al3+, K+, SiO2(aq), whereas Kaolinite precipitates and is the sink for Al3+ and SiO2(aq). The rate term in the parenthesis (1-IAP/Keq) is positive when minerals dissolve and is negative when minerals precipitate so we don’t need to change the sign of the rate term in the equation.
Species. The chemical species from the minerals are Al3+, K+, SiO2(aq). The species from the rainwater include H+, OH-, CO2(aq), HCO3-, CO32-, Na+, and Cl-. Here the major anions are the carbonate species so we can assume the aqueous complexes are KHCO3 (aq) and KCl (aq) and we have a total of 12 species. We can pick Al3+, K+, SiO2(aq), H+, HCO3-, Na+, and Cl- as primary species (7). This means we have secondary species including KHCO3(aq), KCl(aq), OH-, HCO3-, and CO32-. We need to solve for 12 (total number of all species) – 5 (total number of secondary species) = 7 primary species (governing equations).
9.4 At the short time scale: steady-state conditions
As the rainfall contacts the rock, cations and anions are released from the rock. The processes change properties of both solid and aqueous phases. However, the solid rock is dense in terms of amount of chemical mass compared to the aqueous phase so that the solid phase properties change at rates that are orders of magnitude slower than the aqueous phase. The system therefore often reaches steady state where the concentrations of species relatively constant over time.
The relative magnitude of advection, diffusion/dispersion and mineral dissolution are often compared using dimensionless numbers. The dimensionless numbers are quantitative measures of the relative magnitude of the characteristic time scales of different processes. In this case we have three characteristic time scales: the residence time related to advection $\tau\ \text{advection}=\frac{l}{v}$, the time for diffusion/dispersion $\tau\ \text {diffusion/dispersion}=\frac{l^{2}}{D_{h}}$, and the reaction time to reach equilibrium $\tau\ \text {reaction }=\frac{C_{e q}}{k A}$. Here we introduce two dimensionless numbers comparing the different time scales: DaI (Damköhler number for advection), DaII (Damköhler number for dispersion):
where l is the characteristics length of a domain (m), k is the rate constant (moles m−2 s−1), A is the reactive surface area (m2m−3), Dh is the diffusion coefficient in porous media (m2 s−1), and Ceq is the mineral solubility (moles m−3). When Damköhler number >>1, reaction times are much faster than transport times so that the system is transport controlled. The relative importance of advective versus dispersive transport is compared by the Péclet number that we have introduced before.
when Pe>>1, advective transport dominates and dispersive and diffusive transport are negligible.
9.5 At the long time scale: chemical weathering
If you have paid attention to a roadside cut, you will notice that the color, texture or structure along the direction of flow varies. This is because rocks at different depth have been subject to different extent of chemical weathering, resulting in gradients in mineral composition, reaction tendency, and porosity. Soils are generated over thousands to millions of years of chemical and physical alteration of their parent bedrock, which typically have much lower porosity and permeability than soils. Why and how are soils at different depth weathered differently? How do different parameters control chemical weathering?
Chemical and Physical Property Evolution
Mineral volume fraction
Using mass conservation principle, the code calculates the change in mass and volume in each mineral phase, which updates individual mineral volume fractions using the following equation:
Where $\phi_{j m, t}$ is the volume fraction of each mineral phase jm, Vjm is the molar volume of mineral jm, $r_{j m, t+\Delta t}$ is the the reaction rate of the mineral jm at time $t+\Delta t$. This can be done for each grid block in the domain.
Porosity
The porosity at any time $t$ can be updated as follows:
Where $\phi_t$ is the porosity at time $t$ and mtot is the number of solid phases.
Reactive surface area
The reactive surface area for mineral jm at time $t$, $A_{j m, t}$ is calculated based on the change in porosity and mineral volume fraction compared to their values at initial time 0 (Steefel et al., 2015):
Permeability
The local permeability in individual grid blocks is calculated using local porosity values from equation and the Kozeny-Carman equation (Steefel et al,. 2015):
Where $K_{t+\Delta t}$ is the permeability at time $t+D t$updated from time $. With updated permeability, flow velocities can be updated using Darcy's law. In this example, however, we use constant flow velocity for simplicity.
Quantification of chemical weathering: mass transfer coefficient $\tau$
Within a soil profile, $\tau$ is used to assess the effect of the chemical and physical alteration. Note that this is different from the time scale $\tau$ that we used previously on dimensionless numbers. We have to stick to this notation because the chemical weathering community uses $\tau$ for mass transfer coefficient, which is calculated by:
where $\tau_{i, j}$ is the mass transfer coefficient of element j relative to reference element i. cj,w and cj,p are the concentration of element j in weathered soil and in parent rock, respectively. ci,w and ci,p are the concentration of element i in weathered soil and in parent rock, respectively. The element i is considered as an immobile reference element to exclude the effects of the physical forces such as expansion/compaction. Titanium and zirconium are usually considered as immobile elements in calculating mass transfer coefficient. A negative $\tau_{i, j}$ value indicates mass loss while a positive $\tau_{i, j}$ value indicates mass enrichment for element j relative to element i. When $\tau_{i,j}=-1$, element j has been completely depleted as a result of the chemical weathering process. In calculating $\tau$ of K and Si relative to Zr, it is typically assumed that the soil column always has a constant concentration of immobile reference element Zr, which means $\frac{c_{Z r, p}}{c_{Z r, w}}=1$ all the time.
9.6 Example 9.1
Question
Assume the chemical weathering occurred for 0.1 million years. Please set up the simulation in Crunchflow. Plot the profiles of pH, Saturation index (SI, equal to [log10(IAP/Keq)]), $\phi$ ( volume of mineral/ volume of total porous media) of minerals and $\tau$ of K and Si relative to Zr in this soil column at the initial time, 5 thousand years, 10 thousand years, 20 thousand years, 40 thousand years and 100 thousand years. In calculating $\tau$ of K and Si relative to Zr, please assume the soil column always has a constant concentration of immobile reference element Zr, which means $\mathrm{C}_{\mathrm{Zr}, \mathrm{p}} / \mathrm{c}_{\mathrm{Zr}, \mathrm{w}}=1$ all the time. Discuss what you observe.
Note:
Setting up this lesson will involve what you learned from the lesson on mineral dissolution and precipitation, as well as the lesson on 1D transport. If needed please revisit these two lessons. In this lesson we do not provide videos and template anymore.
Please watch the following video: Mineral Dissolution and Precipitation 1D Columns (20:09)
Click here for CrunchFlow files [1]
Click for a transcript of the mineral dissolution and precipitation 1D Columns video
Mineral Dissolution and Precipitation 1D Columns
Li Li: OK, so this lesson we're going to talk about mineral dissolution, precipitation in one dimension, like, 1D columns. So this is different from the previous lessons that we had before on mineral dissolution and precipitation, which was in batch reactor, well-mixed systems. So it is really no dimension. Or you can call it a zero dimension because, in every spatial point, they are the same. So here, we are talking about 1D. What that means is that, essentially, you have a column. So we're talking about processes like, for example, rainfall comes in, hitting parent rock, and things dissolving out. And then, over a long period of time, these chemical species release out. And rock becomes soil.
So I'm going to use this as one example to talk about mineral dissolution and precipitation in 1D columns and how we set up these equations. But it's not really only about chemical weathering. It's really about these mineral dissolution precipitation reactions in general. In the short-terms, these reactions actually change, for example, water chemistry. If you think about a ground-water system, it would actually tend to-- we can see the change if there is mineral dissolution precipitation -- happen pretty fast. But in order to see change in the solid phase, we do need to wait a bit longer for enough change to happen. So today, I'm going to use this system. So I'll talk about this. If we're talking about rock transformation to become soil, we are setting up a system that has rainfall come in at a rate, maybe, for example, some way that is about annual precipitation minus e-t, something like that, at this flow velocity. And first thing, usually, we would have an unsaturated system in these topsoils, but for simplicity, we would just think about everything at the fully saturated system.
So it's easy at the beginning. So we have these systems that have essentially two minerals as parent rock. One is quartz -- and dissolves pretty slowly. Everyone knows about that. The other is K-feldspar, which is a very common silicate in rocks. K-feldspar has a formula of KAlSi and then O3-8. So essentially, you can think about this system-- if you think about rainfall, it comes in, dissolving out, and then releases these chemical species, that you can think about several processes as happening at the same time. One is advection. So this is the same advection we talked about in one unit set with a chaser, essentially, as the flow brings out the chemicals, but also there's a dispersion, diffusion. This again, is the same as what we talked about last time in the AD equations. But then the last one that is different from the previous, one other reaction, which is mineral dissolution precipitation reaction. So essentially this is like combining the AD equation unit with the mineral dissolution precipitation reaction unit. So if you need to go back, you're welcome to review this material. But essentially, because these three different processes are happening at the same time-- but then we also have multiple minerals -- we know the qualities are pretty slowly. And we can more or less think about this as almost non-reactive. Because compared to K-feldspar, it's a relatively very slow reaction. And when we think about the reaction that's happening, the K-feldspar will be dissolving out. So you would have this K-feldspar dissolving out to become released calcium, which is very important nutrients, metals, cations. And then it would be also released aluminum plus SiO2, so that's aqueous. These are species that are going to be coming out.
But at the same time, you also think about, some of these chemical species, when they reach a certain level, actually they can precipitate out as solid phase again. For example, aluminum plus SOi2 actually will become another mirror, which we call kaolinite. So essentially it will have the formula of Ai-Al2-Si2-O5-OH4. This is kaolinite. This is one type of very common clay in our system. Or if you go digging some soil, you will very easily see this type of clay. So essentially, you can think about the whole process as, like, you have parent bedrock. Rainfall comes in, releases some of the chemical species. But then some of these species also re-precipitate out. So it's almost like a redistribution of minerals. But at the same time, because some of species dissolving out over a long period of time, it would change the properties of the system, for example, porosity, permeability. Make this solid phase more permeable and have more pore space for water to flow through. So there's a change in physical properties as well.
So one question we often ask is how does this water and rock change over time and also over space? We want to know how fast things change and how much they change and what they have become. And a lot of time, we want to be able to predict that. So in order to do that, we have to come set up the equations, reactions, and all of that, putting everything together, and think about how we solve them.
So again, with the system, every time we need to think about the chemical system and how they evolve over time and space, we think-- the first thing we need to think about-- the chemical species first. So you have several primary species again. If you review back some of the material we covered before, we had primary species, then existent system-- because we have aluminum, silica-- that they have to be there. This has to be part of the building block of the system. You have potassium too. And when the rainfall comes in, usually it brings some acidity. So H-plus should be there. You would also have CO2-aq. In addition, a lot of times, these rainfalls have a little bit of salt in them. So to be representative, we are also putting a bit of sodium chloride in that example-- sodium chloride as part of the rainfall. That means, also, in the second species, we need to decide, OK, what complexities would be formed, how much can complex reaction happen, and things like this. So to keep it simple, I'm going to just have potassium and then HCO3-aq and maybe KCl-aq as two complexities. And then, of course, we also need other species, for example, H-minus. We need the other form of bicarbonate and carbonate. So it's quickly become a long list of species.
So if we are talking these chemical species, we'll be essentially solving for 1, 2, 3, 4, 5, 6, 7. We'll have seven primary species. So that essentially means we would have seven independent equations to solve for. So what I'm writing here is a general type of governing equation, for one species, I. And again, this first term-- we call, mass accumulation term, that we discussed before. Is a summation of overall change. And then you have the advection term, diffusion term, dispersion term. These doesn't change, because of the system. But for example, the primary would change, but the terms wouldn't change. You could have different porosity phi or velocity or diffusion-dispersion coefficient for a different chemical species. But the term itself wouldn't change. And then the last term is new. It's a reaction term. So it would take into account, essentially, different types or the rates of different types of reactions that this species, I, is involved in. And you think about how fast these different reactions would change in the continuation of species I. And of course, this I needs to be written for 1 to Np, which is the number of primary species. So you need to write, for this system, seven different equations to solve. But then, on top of that, again, you have this number of secondary species you need to solve for in the form of algebraic relationships.
So how does this r-i-j look? These are terms different from the previous AD equations, that without this term. Now here, this r is a [INAUDIBLE] term. So you have that. So for this, r's, it needs to be-- everything about this particular r-- let's make this i is potassium. So for potassium, means this system-- you can think about two reactions involved in. One is that the K-feldspar dissolving out to release out calcium. And then it comes out. And it goes. So actually, one reaction-- so it would be just K-feldspar dissolving out. So if it's K, then you would just have this rate. And then you follow TST rate all right. So you have, probably, this complicated K-H-plus, dependence on H-plus, whatever term, empirical numbers, K-H2O and then a water plus KOH-minus and then Activity OH-minus-- some empirical exponent here as well. These are the regular part, early part, that depends on pH. But then, again, you have surface area. And the last term would be 1 minus IAP over K-aq. Again, it's how fast this is close to equilibrium or far away from equilibrium. So that's the general form of reaction rate law. So when we solve equals to this term, we'll be putting there-- so this is going to be a complicated term. But also, if you think about, for example, species like aluminum or silica, then they're actually involving two different reactions. One is K-feldspar dissolving out to release these. But a kaolinite also precipitated out. So there's one source reaction term and r-i term. And the other would be a sink term for kaolinite precipitating out. So it's a sink for aluminum or silica.
So we solve all of these equations. And what we come up with is all these Ci as a function of time and space. So you have X versus-- essentially, once we know all the parameters, initial conditions, bounded conditions-- what water comes in, what water comes out the beginning, what's the pore water composition in the rock and all that, this will give us this distribution of concentration from time and space. And over geological time, when this changes line-- and when I say geological time, I mean at least thousands of years, thousands or 10 thousands or close to 1 million years, things like that. So over this time scale, you would have these things keep on changing. And then the rock becomes soil, property of the porous media change. You have porosity increases typically. And you have, also, permeability change, because the water-- the resistance of this porous media to water becomes lower and lower. So you have permeability increase as well. So this would be the chemical weathering over long-term. Over short-term, is all of these reaction changes or water chemistry.
But the last part of it, I want to mention a little bit about the dimensionless number, from the mass point-of-view, how these would change, how we analyze these systems. So last time, when we talked about this equation of AD with other reaction term, we talked about the Pe or Peclet number, which is the tau of the advection. It's a tau diffusion-dispersion over tau advection. Tau is a time scale, right? So the time scale of diffusion and dispersion, which is L squared over D-- so it's length of this column-- over diffusion-- because that's the time scale for diffusion-dispersion-- divided by-- or times, if I do it this way-- times 1 over tau-a, which is the opposite of lance over advection. So you would have v-L. So that cancels out. It becomes L-v over D. And so that's a Peclet number, that we talked of before. So this term essentially compares the relative magnitude of advection versus diffusion-dispersion. Which point is dominant under phosphoro or slow conditions? But then, because we have the reaction term, we have two more different dimensionless numbers, we call Damkohler numbers. So the first step-- because think about, we have two transport processes. One is diffusion-dispersion. Other is the advection. So the first one is comparing the time scale of reaction-- the relative time scale of advection to the relative time scale of reaction. And in that case, again, tau-a would be the same as L over v. But then, you'd be-- this is over tau reaction. For reaction, we are thinking about how much things can dissolve and, eventually, reach equilibrium. So you would have total bulk volume times porosity, which is how much pore space you have that can hold water. So that's a lot of volume times the maximum concentrations and you can dissolving out, divided by the rate constant as a maximum, K times A. So this would be, essentially, the reaction time, the characteristic time for reaction. And in notes, I have simplified this. And you can have that. You can look at these. For the second Damkohler number, you would be comparing the time scale of diffusion-dispersion over tau-r. And again, this would be going back to L squared over D, divided by the same thing. Because the reserve reaction term would be the same.
So if you think about-- we typically talk about, under fast reaction system, fast compared to transfer versus when you have very slow reaction systems. So when you have very fast reaction systems, the systems tend to reach equilibrium quickly. And we tend to be transporter-controlled. It would be determined by the bottleneck of all processes, which mean it's going to be transport-controlled. If the reaction is actually slower-- so you will see, by this analysis, you will be able to tell how the system would behave, whether it's transport-controlled or surface reaction-controlled. And how these will be changing over distance and time will give us some kind of grouping analysis that we can do.
So I'm going to end here. I'm going to go back and look at notes. And this, I went through pretty fast. So look at notes we have. Do some homework. And you will realize, these analyses will really help you to understand how things are different under different conditions.
Click for Solution
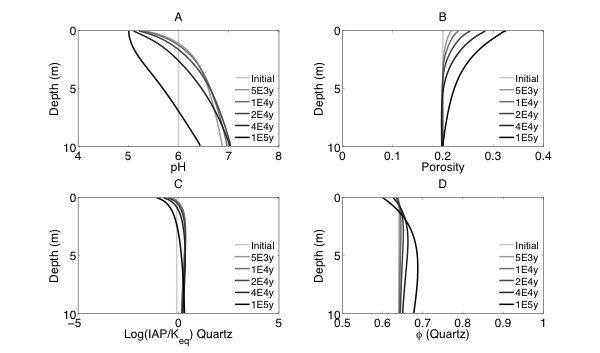
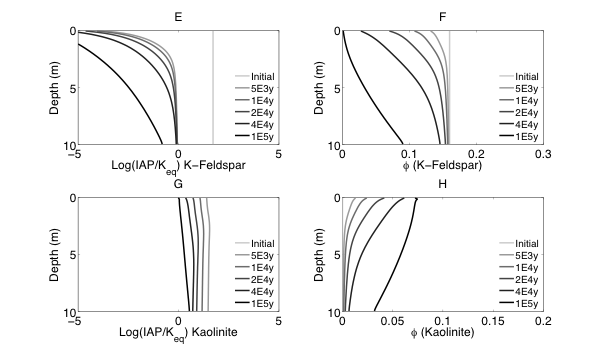
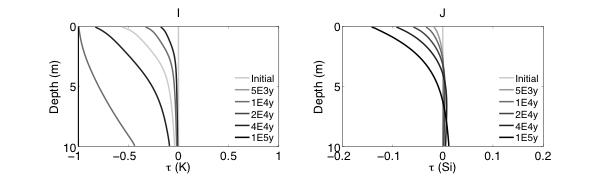
Figure 2 Evolutions of (A) pH (B) porosity (C) Saturation Index $\left[\log 10\left(I A P / K_{e q}\right)\right]$ of K-Quartz, (D) $\phi$(Quartz) (volume of Quartz / volume of total porous media) (E) Saturation Index $\left[\log 10\left(I A P / K_{e q}\right)\right]$ of K-Feldspar (F) $\phi$ (K-Feldspar) (volume of K-Feldspar / volume of total porous media) (G) Saturation Index $\left[\log 10\left(I A P / K_{e q}\right)\right]$ of Kaolinite, (H) $\phi$(Kaolinite) (volume of Kaolinite / volume of total porous media), $\text { (I) } \tau(K)$ and $\text { (J) } \tau(S i)$ as a function of soil depth at different times.
Initially, the pH in the rock is 6 and porosity is 0.2. After the simulation starts, rainwater flows into the soil column from the top with a lower pH so the pH in the upper column (close to the surface) decreased. In the lower part of the column, incoming hydrogen ion is consumed during mineral dissolution so pH increases. As mineral dissolves, porosity is enlarged. Porosity increases more where mineral dissolution is faster. Because rainwater has lower pH and lower solute concentrations, Quartz is under saturated and dissolves from the top. However, because the lower part of column has a higher pH, Quartz is re-precipitating in the lower part of column. K-Feldspar is under saturated throughout the column so it always dissolves. Kaolinite precipitates as a secondary mineral as K-Feldspar and quartz dissolve and elevate the aqueous concentrations.
From the discussion above it is apparent that soil at different depth weathers differently primarily due to the availability of reactants for the dissolution reactions at different depth. The delivery through flow and transport and the consumption of reactants compete with each other in the soil profile, which determines the location and shape of the weathering front. In fact, the extent and speed of chemical weathering can often be interpreted inversely based on understandings of the vertical profile through numerical simulations [Brantley and White, 2009].
9.7 Homework Assignment
Please pick one of the following for homework assignments.
1) Chemical Weathering (Example 9.1 extension, total 60 points, 15 points each):
assess the role of dissolution rates, dissolution kinetics, rainwater chemistry, and rainwater abundance.
Continuing along example 1, please analyze the role of different parameters/conditions in determining chemical weathering rates and profile. In each of the questions below, please compare the base case in the example to two more cases with different parameter values. In each question, please keep all other parameters and conditions the same so we can fully focus on the effects of the changing parameter. In each question, draw the depth profiles of porosity, volume fractions of Quartz, K-Feldspar, and Kaolinite, and $\tau$ figures at 100,000 years:
- 1.1) Effects of K-Feldspar dissolution rates: K-Feldspar rate 10 and 100 times slower;
- 1.2) Effects of K-Feldspar equilibrium constants: 1 order of magnitude larger or 1 order of magnitude smaller;
- 1.3) Effects of rainwater composition: change the total inorganic carbon (HCO3-) concentration by 2 times larger or 2 times lower, to represent changing CO2 content in rainwater;
- 1.4) Effects of annual rainfall (flow velocity): increase and decrease by 2 times.
Click here for CrunchFlow Solutions. [2]
2) Calcite dissolution in a 1D homogeneous column (short time scale, total 60 points).
In a homogeneous column at the length of 10.0 cm and diameter of 2.5 cm, calcite and sand grains are uniformly distributed in the column. Detailed physical and geochemical properties are in Table 2.
| Parameters | Values |
|---|---|
| Calcite (gram) | 14.62 |
| Quartz (gram) | 76.10 |
| Grain Size Calcite ($\mu m$) | 225-350 |
| Grain Size Quartz ($\mu m$) | 225-350 |
| BET SSA of Calcite (m2/g) | 0.115 |
| BET SSA of Quartz (m2/g) | 0.41 |
| AT Calcite (m2) | 1.68 |
| $\phi \text { ave }$ | 0.40 |
| $k_{e f f}\left(x 10^{-13} m^{2}\right)$ | 8.20 |
| Local longitudinal dispersivity $a_{L}(\mathrm{~cm})$ | 0.20 |
The initial and inlet solution conditions are listed in Table 3. Flushing through experiments were carried out at flow velocities of 0.1, 0.3, 3.6, 7.2 and 18.5 m/d at pH 4.0. Prior to dissolution experiments, each of the columns was flushed with 10-3 mol/L NaCl solution at pH 8.8 at 18.0 m/d to flush out pre-dissolved Ca(II) for a relatively similar starting point.
| Species | Initial Concentrations (mol/L, except pH) | Inlet Concentrations (mol/L, except pH) |
|---|---|---|
| pH | 8.8 | 4.0 for all columns and 6.7 only for Kratio,Ca/Qtz at 0.5 (in dissolution experiment) 8.8 (in tracer experiment) |
| Total Inorganic Carbon (TIC) | 1.0x10-3 (Approximate, close to equilibrium with calcite) | 1.0x10-10 to 1.0x10-5 (depending on experimental conditions, some contain CO2 bubbles) |
| Ca(II) | Varies between 1.0x10-5 to 1.5x10-4 depending on experimental conditions | 1.0x10-20 |
| Na(I) | 1.0x10-3 | 1.1x10-3 |
| Cl(-I) | 1.0x10-3 | 1.0x10-3 |
| Br(-I) | 1.0x10-20 | 1.2x10-4 |
| Number | Kinetic reactions | Log Keq | kCa (mol/m2/s) |
|---|---|---|---|
| (1) | $\mathrm{CaCO}_{3}(s)+\mathrm{H}^{+} \Leftrightarrow \mathrm{Ca}^{2+}+\mathrm{HCO}_{3}^{-}$ | 1.85 | 1.0x10-2 |
| (2) | $\mathrm{CaCO}_{3}(s)+\mathrm{H}_{2} \mathrm{CO}_{3}^{0} \Leftrightarrow \mathrm{Ca}^{2+}+2 \mathrm{HCO}_{3}^{-}$ | -- | 5.60x10-6 |
| (3) | $\mathrm{CaCO}_{3}(s) \Leftrightarrow \mathrm{Ca}^{2+}+\mathrm{CO}_{3}^{2-}$ | -- | 7.24x10-9 |
Reaction (1) dominates when pH<6.0; reaction (3) dominates at higher pH conditions: reaction (2) are important under CO2- rich conditions.
Please do the following:
2.1) Calculate the pore volume (total volume of pore space), residence time $\tau_{a}$, and Peclet number (Pe) at each flow velocity;
2.2) Set up simulation for each flow velocity, and plot the breakthrough curves (BTCs, concentrations as a function of $\tau_{a}$) under each flow condition for Ca(II), IAP/Keq, and pH; plot one figure for each (Br, Ca(II), IAP/Keq, and pH) so you have all BTCs under different flow conditions in the same figure; comment on the role of flow in determining calcite dissolution and why; Here breakthrough curve is defined as concentrations vs time at the last grid block of the column.
2.3) Calculate the steady-state column-scale rates under each flow velocity by R = Q(Ceffluent – Cinfluent). Note that Q (volume/time) = u * Ac, where u is the Darcy flow velocity, Ac is the cross-sectional area of the column; Also note that "steady-state conditions" means the conditions under which concentrations in each grid do not change with time any more.
2.4) Calculate the DaI and DaII (again, under steady state) for each flow velocity;
2.5) Make a table of v, R, $\tau$, Pe, and Da numbers at each flow velocity. Plot R as a function of Pe and DaI and DaII.
9.8 Summary
Summary
In this lesson, we introduced the basic principles of transport and mineral reaction coupling in the context of chemical weathering process and how soils are formed.
References
Brantley, S. L., and A. F. White (2009), Approaches to Modeling Weathered Regolith, Thermodynamics and Kinetics of Water-Rock Interaction, 70, 435-484.
Kump, L. R., Brantley, S.L. and Arthur, M. A. (2000), Chemical weathering, atmospheric CO2, and climate, Annual Review of Earth and Planet Science, 28, 611 - 667.
Steefel, C.I., Appelo, C.A.J., Arora, B., Jacques, D., Kalbacher, T., Kolditz, O., Lagneau, V., Lichtner, P.C., Mayer, K.U., Meeussen, J.C.L., Molins, S., Moulton, D., Shao, H., Šimůnek, J., Spycher, N., Yabusaki, S.B. and Yeh, G.T. (2015) Reactive transport codes for subsurface environmental simulation. Computational Geosciences 19, 445-478.
Wolery, T. J. (1992), EQ3/6: A software package for geochemical modeling of aqueous systems: Package overview and installation guide (version 7.0), Lawrence Livermore National Laboratory Livermore, CA.