Lesson 4: Ion exchange reactions
Overview
This lesson introduces general principles of ion exchange reactions and setup simulations for ion exchange reactions in well-mixed batch reactors using CrunchFlow.
Learning Outcomes
By the end of this lesson, you should be able to:
- Understand the importance of ion exchange reactions;
- Understand the important controls of ion exchange reactions;
- Simulate ion exchange reactions in well-mixed batch reactors using CrunchFlow.
Lesson Roadmap
| Recommended Readings |
|
|
|---|---|---|
| To Do |
|
|
Questions?
If you have any questions, please post them to our Questions? discussion forum (not e-mail), located in Canvas. The TA and I will check that discussion forum daily to respond. While you are there, feel free to post your own responses if you, too, are able to help out a classmate.
4.1 Ion Exchange Reactions
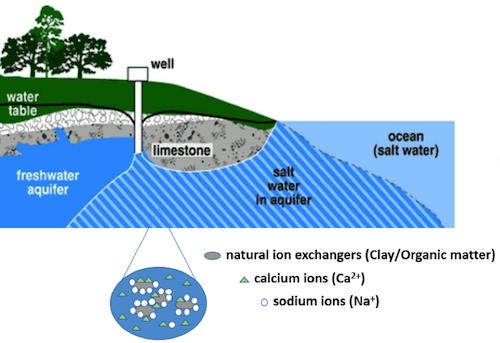
Ion exchange reactions occur when ions in water exchange with those electrostatically bound to the solid phase. They occur commonly in the presence of iron oxides, organic matters, and clay minerals with large surface area. Ion exchange reactions are important in determining natural composition in surface waters (e.g., rivers, lakes) and ground water. They can alter water composition and trigger other reactions including mineral dissolution and precipitation. For example, in the coastal freshwater aquifer, Na+ in seawater displaces presorbed Ca2+ from solid phase (Figure 1). In this reaction, Ca2+ is replaced by Na+ and the water changes from Na-rich to Ca-rich (Appelo and Willemsen, 1987; G., 2008; Slomp and Van Cappellen, 2004) while the solid surface change from Ca-rich to Na-rich. In northeastern United States, the use of road salt as deicer increases the salinity of fresh water and mobilize metals through ion exchange reactions (Kaushal et al., 2005).
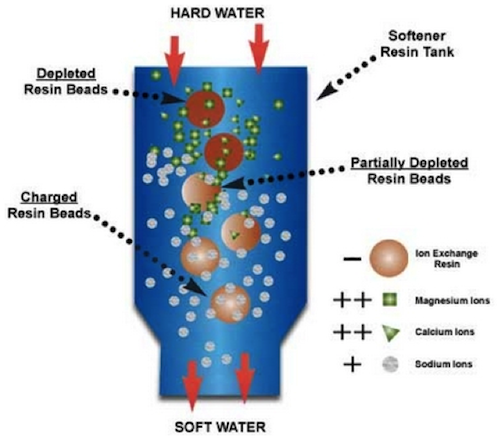
Applications of ion exchange reactions in industry include drinking water softening (Figure 2), desalination, ultra-pure water production (Rodrigues, 1986), and chromatography. In petroleum refining processes, ion exchange reactions are often used to purify, separate, and dry natural gases (Marinsky and Marcus, 1995). Natural gas extraction in the Marcellus shale has led to the production of large quantity of wastewater with high metal concentrations. Ion exchange is commonly used to remove metals such as Ba2+ and Sr2+ (Gregory et al., 2011).
4.2 Reaction Thermodynamics and Important Parameters
Cation exchange reactions
Ion exchange reactions occur when ions exchange their positions at the soid-surface – water interface. These reactions are typically assumed reversible and occur instantaneously at time scales ranging from microseconds to hours. Ion exchange is typically represented in the following form:
Here (s) and (aq) refer to solid and aqueous phases, respectively; X- denotes the negatively charged surface sites that bound cations Au+ and Bv+, with u and v being the charges of A and B, respectively. In this reaction, A and B are cations that compete for sorption sites (Appelo and Postma, 1993; Sposito et al., 1981; Vanselow, 1932). The equilibrium constant Keq of reaction (1) can be expressed as
Here the parentheses [ ] represent activities. Aqueous concentrations are easily related to activities through concentration and activity coefficients. Activities of ions on solid phases are typically expressed as a fraction of the total, either as molar fraction or as equivalent fractions. The total number can be based on the number of exchange sites or as the number of exchangeable cations.
The units meq is often used in ion exchange calculations. An meq is the number of ions that sums a specific quantity of electrical charges. For example, an meq of K+ is about 6.02 x 1020 positive charges. On the other hand, an meq of Ca2+ is also 6.02 x 1020 positive charges, however only 3.01 x 1020 ions because Ca2+ has two positive charges. For an ion Ii+ with a charge of i, the equivalent fraction bI is calculated as:
where I, J, K are exchangeable cation with charges i, j, k, respectively. A molar fraction $\beta_{I}^{M}$ is obtained by the following form:
Here TEC is the total exchangeable cations in mmol/kg sediment, not cation exchange capacity. The use of fractions should give the summation of fractions being 1, that is, .
Three common conventions used in writing ion exchange equilibrium constants are Gaines-Thomas convention, Gapon convention, and Vanselow convention. If the ion exchange is between cations of the same valence (homovalent exchange), the convention does not make a difference. If the exchange is between cations of different valences (heterovalent), the convention makes a difference. For example, the ion exchange reaction between Na and Ca can be written as follows:
With
Here if the equivalent fraction of the exchangeable cations is used for $β$ values, the Gaines-Thomas convention is followed (Graines and Thomas, 1953). If we use molar fractions for $β$ values, we follow the Vanselow convention (Vanselow, 1932). If the activities of cations on exchange sites are expressed as a fraction of the number of exchange sites (X-), we follow the Gapon convention, the reaction will be written as follows:
Where
Here the activities are expressed in terms of the mole fraction of the total number of exchangeable sites. To fully understand the difference between different conventions and calculation, please go over Example 6.3 in chapter 6 of Appelo and Postma (2005).
Selectivity coefficient
The capabilities of ions to compete for exchange sites are governed by their affinity to the surface of the exchangers. Selectivity coefficients have been reported in literature for common ions including Na+, K+, Ca2+, and Mg2+, however rarely for trace metals. The larger the selectivity coefficient, the higher affinity of the ion to the solid phase and the more competitive for exchange. In general, ions have high affinity to exchange sites when they have higher valence, are less solvated with water molecules, and react strongly with the surface sites. The following Table 1 lists selectivity coefficients reported in literatures for ion exchange on kaolinite (Appelo and Postma, 2005; Bundschuh and Zilberbrand, 2011).
| No. | Ion exchange reaction | log K |
|---|---|---|
| 1 | $2 \mathrm{Na}-X+\mathrm{Mg}^{2+} \Leftrightarrow \mathrm{Na}^{+}+\mathrm{Mg}-X_{2}$ | 0.60 (0.44 ~ 0.78) |
| 2 | $2 \mathrm{Na}-\mathrm{X}+\mathrm{Ca}^{2+} \Leftrightarrow \mathrm{Na}^{+}+\mathrm{Ca}-X_{2}$ | 0.80 (0.44 ~ 0.104) |
| 3 | $2 \mathrm{Na}-X+\mathrm{Ba}^{2+} \Leftrightarrow \mathrm{Na}^{+}+\mathrm{Ba}-X_{2}$ | 0.91 (0.44 ~ 0.104) |
| 4 | $2 \mathrm{Na}-X+\mathrm{Sr}^{2+} \Leftrightarrow \mathrm{Na}^{+}+\mathrm{Sr}-X_{2}$ | 0.91 (0.44 ~ 0.104) |
For typical ion exchangers, the sequence of affinity is as follows: Ba2+> Pb2+ > Sr2+ > Ca2+ > Ni2+ > Cd2+ > Cu2+ > Co2+> Zn2+ > Mg2+ > Ag+> Cs+ > K+ >NH4+ >Na+ >H+. When there are multiple cations co-exist in the solution, the surface exchange site composition is largely determined by the cation aqueous concentration and their affinity to the exchange sites. To calculate the exchange site composition, that is, the mole fraction of each cation on the exchange site, please follow the example 6.4 in chapter 6 of Appelo and Postma (2005).
Cation exchange capacity
The cation exchange capacity (CEC, meq/kg solid) is a measure of the solid phase capacity for ion exchange reactions (Meunier, 2005). The CEC of different porous media are very much associated with their clay content, organic carbon, and grain size. Different materials have different CEC values. CEC values of clay minerals such as muscovite, illite, kaolinite, and chlorite are high for their grain sizes smaller than 2 μm (Drever, 1982). In general, organic matter has the highest CEC values (1500 - 4000 meq/kg). Iron oxides play a vital role in natural processes and controls nutrient availability and heavy-metal mobility (Houben and Kaufhold, 2011). Iron oxides, such as goethite and hematite, have CEC values from 40 to 1000 meq/kg. Many solid materials have iron oxides or organic matters coated on their surface and therefore have large CEC values. For soils, CEC value is a function of solution pH depending on hydrolysis reactions of surface sites. In general, cation exchange occurs due to the broken bonds around the crystal edges, the substitutions within the lattice, and the hydrogen of exposed surface hydroxyls that may be exchanged. Higher pH values give rise to more negative charges on clay, resulting in higher CEC. CEC values increase as the grain size decreases due to the large surface area associated with smaller grains.
| Minerals | Grain size (μm) | Surface area (m2/g) | CEC (meq/kg) |
|---|---|---|---|
| Kaolinite | 0.1-5.0 | 5-20 | 30-150 |
| Illite | 0.1-2.0 | 15-40 | 150-400 |
| Montmorillonite | 0.01-1.0 | 600-800 | 800-1200 |
4.3 Setting up Ion exchange simulation in CrunchFlow
Example 4.1.
Marcellus Shale flow back and produced waters typically contains high concentrations of metals, including Na(I), Ca(II), Mg(II), Sr(II), and Ba(II) (Chapman et al., 2012). The release of Marcellus Shale waste water into natural soils and sediment can lead to ion exchange reactions and partition of surface sites between major cations. Here we set up a batch experiment in CrunchFlow to model the major cations exchange on clay surface from Marcellus shale produced water.
Assume that we have the following Marcellus water in a 300 ml batch reactor. The water is in equilibrium with partial pressure of CO2 at 3.15×10-4 atmosphere. Please note that the concentrations are in ppm and in mol/kgw. Both can be used directly in CrunchFlow.
| Component | Conc. (ppm) | Molar (mol/kgw) |
|---|---|---|
| pH | 7.02 | |
| Co2 (aq) | Co2(g), 3.15e-4 atm | |
| Na(I) | 7900.0 | 3.43e-1 |
| Ca(II) | 2774.0 | 6.94e-2 |
| K(I) | 82.5 | 2.11e-3 |
| Mg(II) | 239.0 | 9.83e-3 |
| Ba(II) | 183.0 | 1.34e-3 |
| Sr(II) | 6.5 | 7.38e-5 |
| C1(-I) | charge |
“Charge” means that the ion concentration is calculated from charge balance.
The water is in equilibrium with a sediment with kaolinite being the major clay with a specific surface area of 13.9 m2/g and a CEC value is 100.0 meq/kg (the units in CrunchFlow is eq/g). The kaolinite occupies 0.005 of the total volume.
The selectivity coefficient of the cations are as follows in the database (values from (Appelo and Postma, 1993; Li et al., 2010)):
| No. | Ion exchange reaction | log K |
|---|---|---|
| 1 | $\mathrm{Na}X\Leftrightarrow\mathrm{Na}^++X^-$ | 0.0 |
| 2 | $\mathrm{K} X \Leftrightarrow \mathrm{K}^{+}+X^{-}$ | -0.69 |
| 3 | $\mathrm{Ca} X_{2} \Leftrightarrow \mathrm{Ca}^{2+}+2 X^{-}$ | -0.39 |
| 4 | $\mathrm{Mg} X_{2} \Leftrightarrow \mathrm{Mg}^{2+}+2 X^{-}$ | -0.30 |
| 5 | $\mathrm{Ba} X_{2} \Leftrightarrow \mathrm{Ba}^{2+}+2 X^{-}$ | -0.45 |
| 6 | $\operatorname{Sr} X_{2} \Leftrightarrow \mathrm{Sr}^{2+}+2 X^{-}$ | -0.45 |
Questions
- Without having any aqueous complexation reactions for the cations, what are their concentrations in water and on the exchange site? i.e., mole fraction of each species on the exchange site. Compared to its own original total aqueous concentration, what is the mole fraction of each cation on its exchange sites? Please make a table and a figure comparing different species. Which one has the largest percentage of its own mass on exchange sites? Why?
- Rerun the simulation with the cations have some major aqueous speciation reactions that lead to the formation of the following aqueous complexes: Cl species (CaCl+, MgCl+, BaCl+, NaCl(aq), SrCl+), and OH- speices (CaOH+, MgOH+, BaOH+, NaOH(aq), SrOH+). Answer the same sets of questions as in question 1.
- Compare the answers in question 1 and 2, does aqueous speciation make a difference in ion exchange reactions? What is the major difference?
CrunchFlow setup summary:
To set up CrunchFlow simulation please read CrunchFlow manual page 62 – 63 for details. Please also refer to the Exercise 5: Cs exchange on Hanford sediments.
You will need to set up the ION_EXCHANGE key word block in the input file to define the name of the exchange site and give parameters related to ion exchange in the CONDITION block. In addition, you will also need to define exchange reactions and equilibrium constants in the “Begin Exchange” block. For example, if you call your exchange site “XKao”, then you have the following block:
ION_EXCHANGE
exchange XKao- on Kaolinite
convention Vanselow
END
The first line specifies name of the exchange site and the mineral that the exchange is on. The second line specify the calculation convention that is used.
Li Li: All right. Let's start the lesson on ion exchange. I discussed in the text that ion exchange is somewhat similar to surface complexation. But then you have multiple cations. And these cations have different affinity to the solid surface. So essentially, they are competing with each other. As one with high affinity and high concentration in the water phase, it ends up to have more on the solid phase.
So if you look at the Keq values of the different-- you can get more or less a sense of their relative affinity. But also, if one particular species has high concentration in the water, it also ends up to be more on the solid phase because it has high concentration if you write a Keq values. In order to get the Keq, you need higher corresponding solid phase concentration.
So let's look at the example. Actually, this really should be 4.1. And that's 3.1 So let's go through this example. And we'll talk about how to set it up. This example, I'm kind of thinking about Marcellus Shale water, produced water or flowback water. And you have some concentration. We know there are a lot of different cations in this type of water.
So I'm giving you a typical composition of Marcellus Shale water, with high sodium, chloride, calcium. But you also have barium, strontium. Actually, the barium, strontium concentration is relatively low in what you have here. So we are here.
And I'm giving you both PPM and mole per kilogram water. Actually both of them, you can put in directly. So it's your choice. Now, I'm saying you have 3,000 milliliter of batch reactives. And water is equivalent with partial pressure of CO2, which is this is atmospheric concentration of CO2, try to simulate condition in soil.
And then, I'm giving you this table that essentially can document the K value for each reaction process. And the question I'm asking you, the first is, without any aqueous complexation reaction. So you are not going to put-- you're still going to put OH minus, carbonate, and all that. But you are not going to put, for example, calcium chloride, calcium OH plus, and all these secondary species.
I'm asking you to see, how much is absorbed on the exchange site. And what is the percentage of the absorption on the exchange sites compared to its original total concentration. Essentially, I'm asking you to make a table for each major cation and list aqueous concentration column and list another exchange site column, and compare different species.
But also calculate the percentage of the ones that are on the exchange site. Because they all start with aqueous concentration. So comparing the absolute value sometimes does not tell a lot of things. So you need to normalize them to the original total concentration.
But then I also want you to look at, on the exchange site, how much is occupied by each of these cations? How much is sodium? How much calcium? How much is barium, strontium, et cetera. So that's the first question.
And then second question is, running the simulation with speciation or complexation reactions. List the formation of chloride species and OH minus species, all these listed here. And then I'm asking you to answer the same question. So comparing one to, essentially, you getting some information about how important are these aqueous complexation. Does aqueous complexation help or not help these cations to go on the service site?
So let's start doing this. We had a discussion today. And then you are saying, maybe you can start with a blank one. So today I'm going to start with a blank input file. Essentially, there's not much information there. Except that you will be doing like all the runtime parameters up there.
And I also put a bit of output keyword specification there. I put two spacial profile time points. So it will be running into 100 minutes. And then, initial condition, this is already specified. We will specify initial condition later.
So let's go through each agree grid block-- keyword block. First one is discretization. This one here, you essentially would specify the size. And we know it's one grid block. So you would be doing-- first of all, you need to put in the units for length.
And we talked about this is 300 milliliters. So if you want to put centimeters or meters, that's fine. Last time we put centimeters. So this time, maybe, we'll just put in meters to have some variation. So the distance unit will be meters. And then before, we always specify x zones. You essentially only have one species.
You will specify one zone. And then, because it's 300 milliliters, so that's 300 centimeters cubed. And converting to meters would be 3 times 10 to the minus 4 meters cubed. I mentioned before, this is essentially, when we do this, we specify that this is essentially the length of x zone.
But by default, you actually have y zone and z zone. And the code is going to assume in these other two directions that you don't specify, it's going to 1 meter and 1 meter. So here you have specified 0.003 meters. So essentially your volume is 0.0003 cubic meters, which is 300 milliliters.
So that's here. Now you specify, essentially, the size of your system. Now we probably can try this now, just to be sure that you are not making any mistakes taking this block. I always think it's a good practice to input one by one. And after each keyword block, you kind of check if everything is correct, you didn't make any wrong mistakes or whatever in the input file.
So the top one is read and write. But because we didn't specify any condition, it doesn't know what to do. That's OK. At least it didn't give me anything about like it's not specified correct or something like that. So let's do that again. Let's continue.
So let's specify primary species. And for that, we need to look at our table. So always put H plus first. I mentioned that before, probably multiple times. But it's worth to mention again. Always put H plus. And we know we have CO2 aq. So essentially, it follows that table. The system is with CO2 gas. So you need to put to CO2 aq. No, actually you don't need because it's just primary species.
And then you will be putting all the different species like sodium-- just calcium, potassium, magnesium, barium, strontium, and chloride. That's your primary species on the table.
But remember is that we also have kaolinite. And I mentioned that for each mineral you put-- so later on we'll put kaolinite in the mineral block. Remember that in order for it to work for the mineral you will need to look up what this mineral is made up of. So let's look through.
Let's open with Notepad. So let's search for kaolinite. Let's see what it can post up. So kaolin coming up. You have H plus, you already have. You have aluminum. You have silica, SiO2 aq H2O. So essentially it's aluminum silica. Because aluminum silicon is not there on the list, so let's put these two in so we don't get messed up.
So you would have aluminum. And then you have SiO2 aq. So these are the species we can think about it. So that's the primary species list.
Now I'm saying that we don't put any secondary species like the chloride OH species. But we still need to put OH- in. Because you can't do anything without OH-, because that's water dissociation-association. And then you also have CO2 species. So you have to have bicarbonate and carbonate species. So these are the main species that you have to complete.
So that's a secondary species. And gases, do we have gases? We do because we have the CO2 aq that is in equilibrium with gas. So you do need the CO2 gas phase. And then, you have ion exchange. So this is a new keyword block. We learned this in this unit.
So the key here is exchange, the keyword exchange. I shouldn't do it in Word. And then I'm going to give it a name, which is x representing surface species. But this one is on kaolinite. So let's do this. Let's call that. And this is on the mineral kaolinite. So essentially, you put as an exchange, you have the keyword block and name of the surface site, exchange site, and then the name of the mineral.
But also, the other thing you need to specify is what convention are you using for the calculation. We talked about three different-- like Vanselow, Gaines-Thomas for example. And let's here, let's do Vanselow. Vanselow is the one the most commonly used conventions. So that's ion exchange.
So this keyword block is somewhat new. And you need to think about, OK, doing that, your flux will be here. But again, here, you have the kaolinite. And you need to put in the mineral kaolinite there, the name there. If it's a mineral dissolution precipitation, you would need to put it in like kinetic information, for example, dissolution rates and everything. But here we are not really looking at it the dissolution precipitation kaolin. So we can just specify kaolinite.
So these are all the kind of building blocks of your system, chemistry building blocks of your system. And now we're going to specify what condition we have. And again, we're looking at the table we have. It's Table 3. So let's just copy the species from aqueous, the species we had before. And we'll copy it. And then we will add whatever concentrations they need to be.
We probably should have units first. Actually, in condition, we all need to specify units. And this will, let's say, do PPM. And we can do temperature. We must always do temperature, which is 25.0. You can specify H plus. But this table, I'm giving you 7.02. So it's easier to do just pH, which would be 7.02.
And you have CO2 aq, we discussed last time in last lesson, you can specify the CO2 concentration with gas phase in this way, when you have a specific at partial pressure of that gas. E minus 4, so that's for CO2. And then we have sodium, which is 7,900.0. Calcium, 2,774.0. And the you have potassium, 82.5.
I always think it's good to line up and make it neat and clean. Because if you're not clean and lined up and all that, it's very easy to make mistakes. It's almost like when you write code. You want to keep it very clean and organized so that minimizes the chance of making mistakes. And when you check, it's easier to see. So it's worth the effort. 6.5. And chloride, we'll be doing that with charge. So you just put the charge there.
How do I know that this is going to be positive charge without this? You can try database sweep first, if you want. But I kind of know because I know there's a lot of cations there. You see there's a lot of cations there. But there's almost the only species for now will be chloride in OH minus. And pH you know is 7. So OH would be 10 to the minus 7 or something. So it must be positive charged.
And then we don't really have much of the aluminum. But we can put, for example, let's say we did before with this small concentration, just 3%. It's there. Acidic. So these are the primary species, our total concentration of primary species.
Now I need to, so, in a beaker, that's the water phase composition. But then you also need to specify how much solids is there and how much surface area of a solid, how much CC and all of that. So let's say we have volume fraction of the kaolinite is 0.005. I think I didn't specify that in the file. Let's do that with sediment with kaolinite being the major-- with specific surface area. And then I'll add the kaolinite occupies 0.005 of the total volume.
So you can actually calculate that because you have 0.005. And then the total concentrated volume is 300 milliliters. So that means you have about 1.5 milliliter of the kaolinite. And we should specify the same thing in the input file. So leave a space line here just to have the division between the solid phase and aqueous phase.
So kaolinite, first thing you need to specify is 0.005 volume fraction. Actually, of the solid phase, all these-- So essentially, you have 0.005. So that's actually smaller. We started talking about it. We didn't really put the porosity. We actually should put a porosity there.
Let's finish this mineral for a specific surface area. We said it's 13.9 meters squared per-- so it's volume fraction first on solid phase, and then specific surface area using SSA in meters squared per gram.
So that's a mineral. But you also need to specify the surface site and exchange. So you have xkao, which is your name that's specified in the exchange. This needs to be consistent with the exchange grid block. And then you have this kaolinite CC, which specifies how much kaolinite exchange capacity you have.
Let's do a bit of calculation here. Let me just make some notes here. You have 100.0 meq per kilogram, which is essentially 100.0 eq per gram. And this is equivalent to 1.0 E minus 2 eq per 100 grams, and also equivalent to 1.0 E minus 4-- oh, I'm sorry. This is actually not right.
You have kilogram to gram. And then that's a solvent, And meq to eq is 10 to minus 3. So this is actually 1.0 E minus 4. This is essentially the same as-- let me check the units of CrunchFlow. It's eq per gram. So essentially, eq per kilogram.
So this will be actually-- we should convert to eq per kilogram. Already kilogram, so eq 100 times 10 to minus 3, which is 10 to minus 2. I'm sorry. This should be 10 to minus 1, essentially 0.1 eq per kilogram. We actually don't really need that. We can do our conversion.
Now, so that means you need to put-- because this is CrunchFlow units. So this will be essentially 0.1. So you specify the mineral volume fraction keq. But actually, OK, here I said we actually need a little bit porosity. So let's say you have-- we probably should say 99.5% is water. So you actually porosity of 9--
So actually I should put kaolinite being 100%, 1.0, meaning all the solid phase of the kaolinite, because this is saying the total volume. So the rest of it is all water. And then so your porosity is 0.995. And you have 100%. They're all on the solid phase. This is volume fraction on solid. You only have kaolinite. That's what I mean. So that's a condition.
So I guess, do we need anything else? We have the dimension. Let's just try that. Dimension, Vanselow, secondary species, primary species, and all that. So we should be OK. We don't need to do any timestamping. Let's try if it works.
So it'd be lesson 4.1.in. Specify porosity smaller than zero. Let's see. Must be somewhere, something wrong in the input file. Maybe it's error. I don't know. 0.0. Let's just try again. 4.1.in. Hm. Interesting.
A lot of times when it reports one error, a lot of times it's because something else is wrong. I wonder if this is it because I have it opened in the Word format. So let me just copy everything and open in Notepad and see if it's because of that.
And we're going to call it Lesson 4.1 again. And we specify lesson 4.1.in. OK, because I'm opening that. Let's see. Let's try it again.
I do have this just specified. OK, let's see. Hm. Because of this, I think it's still the original one. Now it's in the-- let's do this. Hm. That's very interesting.
Oh, the volume -- Let me just think about it. I might have forgotten in. It does need to be the volume fraction total. So then you can-- I just realized that.
So the original file was right. It is a total volume. It's not the solid phase. That kind of mistake was different type of information. So let's try it. I think that's a reason. Because I specified one here. And then, it was seeing porosity-- it specified that it was 1 kaolinite So it will say porosity is smaller than 1 or equal to zero essentially.
Let's try it again. I believe that is the error. Now it runs. OK, great. So you now see the process of doing this. I also make mistakes. And it takes time to debug and to realize what's wrong. And in this case, actually, what is wrong here, it is porosity. So it does point out to the right location. So the lesson here is volume fraction on total volume system.
So later on, if for example, if you have multiple minerals here, let's say you specify kaolinite, you specify calcite, another mineral. So volume fraction of the mineral and the volume function of the porosity needs to add up to be 1.0. That's what you need to remember. Or the a batch reactor.
So now we can look at the output file, essentially. Let's look at, well, two files here for each because I specified two different times. But you can imagine that they should be the same. I can tell because the ion exchange reaction occurs really fast.
It doesn't have time dimension. So it doesn't really matter if it's 100 minutes or it's 0.1 minutes. Because it reached equivalence instantaneously. So if you compare this exchange one versus exchange two, they should have the same concentration, essentially.
So you have one, two. It's the exact same. So this is the exchange output, essentially, specifying in mole per gram solid, how much sodium on the surface, how much potassium, how much calcium, magnesium, strontium.
And you can see from the list, the dominant species is sodium, and then followed by calcium. Both of these species are very high concentration in the solution. Let's look at the input file again. Let's look at the table again.
So sodium and calcium are the highest two concentrations. Potassium was relatively low. Magnesium, barium is in hundreds. And strontium has lowest concentration. So let's look at this again.
So more or less, in here, because of the huge difference in concentration, you end up the sodium has the higher concentration. Calcium has the second highest. And then you have magnesium, barium, and strontium. So magnesium is a bit higher than barium, too. So the last one is barium. So that's for the exchange site.
And then if you look at the aqueous site, let's look at total concentration. That'll give you how much is in the-- total concentration is not in log units. So you have pH. You have CO2. Sodium, sodium goes from-- actually it's-- now you think about it.
The output is all in mole per kilogram water. So it's actually probably better if you're using the input file mole per kilogram water. So you can look at the differences between the output, which is after the reaction, to before the reaction. So now you have sodium at 0.34. Potassium, this much.
And you actually can calculate, for example. So these are the concentrations. This is per kilogram water. So you know there's 300 milliliters water. So this concentration times the volume of the water, that gives you the mass in the water phase. And for the exchange site, it gives you in units of mole per kilogram per gram solid.
And you can assume that we know it's 0.005 of the total volume of kaolin. So we actually can calculate from the 300 milliliter times 0.005, which is 1.5 milliliter of kaolinite. And so it's 1.5 centimeters cubed of kaolinite. You can convert that to gram of kaolinite using the density of the kaolinite, give you the total gram of the solid phase. And that, you can convert to how much mass on the exchange site.
So with that calculation, you essentially will be able to calculate how much for each species is in water and how much is actually in the solid phase for each species, the percentage. So you know which one more, actually. So that's for aqueous phase and solid phase and all that.
So the second question for that is when you do include these aqueous complexations. Let's do that. I'm closing this, closing this. I'm going to add the secondary species, more species. So this, it would be having all the OH species and chloride species. So let's see. Let's go through the list.
You have sodium chloride, which is aq because it doesn't have charge. And then you could have NaOH aq. Let's just go through each species. And then there's sodium. You could have calcium chloride positive, calcium OH positive, then you have potassium chloride aq. And then CKOH aq, Mg, MgOH plus, BaCl plus, BaOH plus. What else? Strontium. OK. Strontium, Cl plus. Strontium OH plus.
When I input these, I always try to be very careful. And I probably mentioned that you cannot use tab in the input file. Also you cannot use comments on the middle of the line. When you need to comment, you need to start new line. Otherwise, it could get confused.
So I to be more careful when putting this input file. Because if you're not careful, you end up spending more time debugging and all of the frustration and all that. So keep things neat. And be careful when you input these. That will minimize the chance of making a mistake and that. I mentioned that before.
So then you can save that original output file to a different folder. Otherwise, because I run, it will override to the original file. Here, just for demonstration, I don't do that. So if I run it again, it will surely overwrite everything. That's fine.
Secondary species input file not find in database. Let's see what happens. Hm. We just run it and it works. So it must mean I either made some mistake or let's see. Secondary species input file not found in database. I'm pretty sure that's not true. I'm sure there is a database for CO3. I'm going to search for CO3.
It should be there. Because, otherwise the previous run wouldn't finish. So it's there. So there is nothing wrong with the database. So it must be the input file. It's almost always our fault, as you can tell. Secondary species, just make sure everything is-- hm.
Looks like there might be something wrong there because I couldn't go up. Oh, OK. So there's a lot of space there. And when it started lines, that space might be too long. So it couldn't-- OK. Let's just do it again. This is probably it, I think.
So when you do this, OK, so you have this. Try to not have too much space in between. Otherwise, the code cannot handle it if it's too long. There are certain character limits in each line that the code can process. Let's do it again. I think that's the problem.
4.1.-- Now it runs. So it runs and you can see from the date modified, it's just the right time. So that means it's all the output file updated. And if you look at the exchange again and compare to the previous one if you saved it-- so you will. When you do homework, you will need to save it and compare and see how much difference does it make.
We went through the output file. So I don't think I need to talk more. What you will need to do is in each CrunchFlow, the one without aqueous speciation, make a table for the mass fraction of each of aqueous phase and mass fraction of the solid phase on the exchange site. And then do that for each major cation.
And then, do the second question when you do have these aqueous complexation, how much difference does it make? And you can also check the specific concentration of all the aqueous species to see which one dominant and all that. And for this, if you want is all the dominant species, you don't need to open each exchange file and everything. You actually can just look at the output file, less than 4.1 output file. All the concentrations should be there.
I used the output file just for you to see what is in this output file. But in here, this part should have everything there. And this part might not give you the unit so you can actually dig into the exchange or exchange.out in this file to look if you forgot about units.
For solid, it's mole per gram. For aqueous, it's mole per kilogram water. So it'd have all the different species. It'd also have all the exchange concentration. It'd also have all the aqueous concentration there. So you have everything you need.
I'm going to stop here. And you saw me make mistakes and then we debugged it. Actually, both times the code pointed the right place that I made a mistake. The first one was porosity because I didn't put porosity right. I put high kaolinite 1.0, so there's no room for porosity.
The second one is CO3 minus minus, and there's too many blank space in front of that. So the code doesn't realize it. Anyway, so that's the two mistakes I made. Try to make it kind of no space, just do enter for each species. You'll be fine.
All right. So I'm going to stop here. And you'll be working on the homework and everything. Have fun.
Solution to Example 4.1
1) Without aqueous speciation reactions, the results are summarized in Table 5. Here we show a detailed calculation for Ca as an example.
CaXKao on exchange sites: The total cation concentration on the exchange sites (summation of all cations on exchange site) = $8.03 \times 10^{-5} \mathrm{mole} / \mathrm{g}$
The Ca fraction fraction $(\%)=1.54 \times 10^{-5} \mathrm{mol} / \mathrm{g} / 8.0 \underline{3} \times 10^{-5} \mathrm{mole} / \mathrm{g} \times 100 \%=19.3 \%$
Molar fraction (%) of CaXKao of original total Ca mass:
Batch reactor size: $0.3 \mathrm{L}=300 \mathrm{cm}^{3}$
Kaolinite volume: $300\mathrm{cm}^3\times0.5\%=1.5\mathrm{cm}^3$
Kaolinite molecular volume: $99.5 \mathrm{cm}^{3} / \mathrm{mole} $
Kaolinite molecular weight: 258 g/mole
So for the given batch reactor of 0.3 L, it has kaolinite volume of 1.5 cm3. Converting the kaolinite volume to the gram unit, $\left(1.5\mathrm{~cm}^3/99.5\mathrm{~cm}^3/\mathrm{mole}\right)\times258\mathrm{\ g}/\text{ mole }=3.89\mathrm{\ g}$
Number of mole of Ca on the exchange site: $1.54\times10^{-5}\mathrm{\ mol}/\mathrm{g}\times3.89\mathrm{\ g}=6.0\times10^{-5}\mathrm{\ mole}$
Number of mole of total Ca: $\text{ Ca: }6.92\times10^{-2}\mathrm{\ mol}/\mathrm{L}\times0.3\mathrm{\ L}=2.08\times10^{-2}\mathrm{\ mole}$
Ca fraction on the exchange site to total Ca: $6.0\times10^{-5}\mathrm{\ mole}/2.08\times10^{-2}\mathrm{\ mole}=0.289\%$
In Table 5, Na is the dominant cation on the exchange site (77.9%) because of its high concentration in water. Mg has the largest fraction (0.298%) of its own mass on the exchange site. The partition between mass in water and in solid phase depends on the intrinsic selectivity coefficient and original total aqueous concentration.
| Cation | *In water (mol/L) | On exchange site (mol/g solid) | Mole fraction (%) of total | Mole fraction (%) of its total mass) |
|---|---|---|---|---|
| Na+ | 3.44E-01 | 6.23E-05 | 77.9 | 0.235 |
| Mg2+ | 9.83E-03 | 2.26E-06 | 2.83 | 0.298 |
| Ca2+ | 6.92E-02 | 1.54E-05 | 19.3 | 0.289 |
| Sr2+ | 7.42E-05 | 1.62E-08 | 0.0202 | 0.282 |
| Ba2+ | 1.33E-03 | 2.90E-07 | 0.363 | 0.282 |
* Note that all the aqueous species are for total concentration, which means that we added up the concentrations of all relevant species. For example, for Ca2+, we added free Ca2+ and all other Ca-containing aqueous species including CaCl+ and CaOH+. 2) With aqueous speciation reactions, the results are summarized in Table 6. Ca has the largest percentage (0.296%) of its own mass on the exchange site, because Mg forms stronger aqueous complexes than Ca.
| Cation | In water (mol/L) | On exchange site (mol/g solid) | Mole fraction (%) of total | Mole fraction (%) of its total mass) |
|---|---|---|---|---|
| Na+ | 3.44E-01 | 6.18E-05 | 77.2 | 0.233 |
| Mg2+ | 9.83E-03 | 2.13E-06 | 2.67 | 0.281 |
| Ca2+ | 6.92E-02 | 1.58E-05 | 19.7 | 0.296 |
| Sr2+ | 7.42E-05 | 1.61E-08 | 0.0201 | 0.281 |
| Ba2+ | 1.33E-03 | 2.95E-07 | 0.369 | 0.287 |
3) Comparing Table 5 and 6, the speciation reactions do change cation concentrations on the exchange site and the molar fractions slightly. This is because the aqueous speciation affects the partition of cations between different species, thus determining the concentration of free available cation ions that are involved in ion exchange reactions. Aqueous speciation in question (2) leads to a different pattern of relative aqueous cation concentrations, which determines the cation concentrations on the exchange site. The speciation reactions in question (2) does not significantly change molar fractions. This is because in the current simulation set-up, the exchange sites are relatively limited compared to the aqueous cations. Thus, the molar fractions of cations on exchange sites to its original total concentration is small and relatively insensitive to the cation concentration changes on the exchange site. The situation would be different if the surface area, or CEC is much larger.
4.4 Homework Assignment
Question 1 (an extension of example 4.1):
As an extension of the example 4.1, we can look at different parameters and understand how they change the cation concentrations on the exchange site. Please include all Cl- and OH- aqueous complexes as in example 4.1. Please calculate the major cation concentrations on the exchange site (K, Na, Ca, Mg, Ba, Sr) by changing only the parameter discussed in each sub-question, with all other parameters being the same as those in example 4.1. For each sub questions, please plot major cation concentrations on the exchange site as the changing parameter.
- Specific surface area: 1.39, 13.9, and 139.0 m2/g;
- CEC: 10.0, 100, and 1,000 meq/kg;
- pH of the Marcellus water: 7.02, 8.00, 12.0. (Note that at pH=12.0, you need to use the Cl- concentration you got from pH = 8.0 and put Na+ as "charge" species)
For each question, calculate the mole fraction of each species on the exchange site, and the mole fraction of each species compared to its own original total mass. Please make a table and a figure comparing different species. Which one has the largest percentage of its own mass on exchange sites? Why?
Which parameter(s) have the largest impact on ion exchange reactions? In lesson 3 we learned that pH is important for surface complexation reactions. Does it make a difference for ion exchange reactions?
Question 2 (optional): This is part of problem 6.7 in chapter 6, A&P.
Read the paper (Valocchi et al., 1981). Native groundwater in the injection test of this paper had the composition of $Na^+=\ 86.5,\mathrm{\ Mg}^{2+}=18.2,\text{ and }\mathrm{Ca}^{2+}=11.1\mathrm{\ mmol}/\mathrm{L}$. Injected water has 14.66 meq Cl-/L. Selectivity coefficient were (Gaines and Thomas convention, activity = molal concentration) $\mathrm{K}_{\text{Na\Mg}}=0.54\text{ and }\mathrm{K}_{\text{Na\Ca}}=0.41$. Sediment $\mathrm{CEC}=750\mathrm{\ meq}/\mathrm{L}$ pore water. Calculate
- the composition of the exchange complex of native groundwater.
- the composition of water after the salinity jump, in equilibrium with the original exchange complex (compare with Figure 6.16).
- the composition of the exchange complex in equilibrium with injected water (cf. example 6.11
4.5 Summary
Ion exchange is an important chemical process in natural and engineered systems. Understanding ion exchange reaction is important for us to understand and predict the reactive transport and fate of chemicals in nature. Here we introduce the reaction thermodynamics of ion exchange, and the set up of ion exchange simulation in a well-mixed batch reactor in CrunchFlow.
References
(2008) St. Johns River Water Management District,. WaterWays: More about Water Below the Ground.
Appelo, C. and Willemsen, A. (1987) Geochemical calculations and observations on salt water intrusions, I. A combined geochemical/minxing cell model. Journal of Hydrology 94, 313-330.
Appelo, C.A.J. and Postma, D. (1993) Geochemistry, Groundwater and Pollution. A. A. BALKEMA PUBLISHERS
Appelo, C.A.J. and Postma, D. (2005) Geochemistry, groundwater and pollution. CRC Press.
Bundschuh, J. and Zilberbrand, M. (2011) Geochemical Modeling of Groundwater, Vadose and Geothermal Systems. CRC Press.
Chapman, E.C., Capo, R.C., Stewart, B.W., Kirby, C.S., Hammack, R.W., Schroeder, K.T. and Edenborn, H.M. (2012) Geochemical and Strontium Isotope Characterization of Produced Waters from Marcellus Shale Natural Gas Extraction. Environmental Science & Technology 46, 3545-3553.
Drever, J.I. (1982) The geochemistry of natural waters. Prentice-Hall, Englewood Cliffs, N.J.
G., C.J. (2008) Overexploitation and Contamination of Shared Groundwater Resources. Springer Netherlands.
Gregory, K.B., Vidic, R.D. and Dzombak, D.A. (2011) Water management challenges associated with the production of shale gas by hydraulic fracturing. Elements 7, 181-186.
Houben, G. and Kaufhold, S. (2011) Multi-method characterization of the ferrihydrite to goethite transformation. Clay Minerals 46, 387-395.
Kaushal, S.S., Groffman, P.M., Likens, G.E., Belt, K.T., Stack, W.P., Kelly, V.R., Band, L.E. and Fisher, G.T. (2005) Increased salinization of fresh water in the northeastern United States. P Natl Acad Sci USA 102, 13517-13520.
Li, L., Steefel, C.I., Kowalsky, M.B., Englert, A. and Hubbard, S.S. (2010) Effects of physical and geochemical heterogeneities on mineral transformation and biomass accumulation during biostimulation experiments at Rifle, Colorado. Journal of Contaminant Hydrology 112, 45-63.
Ma, C. and Eggleton, R.A. (1999) Cation exchange capacity of kaolinite. Clays and Clay Minerals 47, 174-180.
Marinsky, J.A. and Marcus, Y. (1995) Ion Exchange and Solvent Extraction: A Series of Advances. CRC Press.
Meunier, A. (2005) Clays. Springer, New York, Berlin.
Rodrigues, A.E. (1986) Ion exchange: science and technology. Springer.
Selim, H.M. (2012) Competitive Sorption and Transport of Heavy Metals in Soils and Geological Media. CRC Press.
Slomp, C.P. and Van Cappellen, P. (2004) Nutrient inputs to the coastal ocean through submarine groundwater discharge: controls and potential impact. Journal of Hydrology 295, 64-86.
Sposito, G., Holtzclaw, K.M., Johnston, C.T. and Levesquemadore, C.S. (1981) Thermodynamics of Sodium-Copper Exchange on Wyoming Bentonite at 298-Degrees-K. Soil Science Society of America Journal 45, 1079-1084.
Valocchi, A.J., Street, R.L. and Roberts, P.V. (1981) TRANSPORT OF ION-EXCHANGING SOLUTES IN GROUNDWATER - CHROMATOGRAPHIC THEORY AND FIELD SIMULATION. Water Resources Research 17, 1517-1527.
Vanselow, A.P. (1932) The utilization of the base exchange reaction for the determination of activity coefficients in mixed electrolytes. Journal of the American Chemical Society 54, 5.