Module 7: Ocean Acidification, Red Tides, and Monster Jellyfish
Module 7: Ocean Acidification, Red Tides, and Monster Jellyfish
Introduction
There is one definite advantage of being over the age of forty. If you were lucky enough as a kid, you had the opportunity to snorkel or dive in a place like the Florida Keys or the Bahamas when coral reefs were healthy and diverse, colorful, and teeming with life. There are very few places on Earth where this is the case today. Tragically, the large majority of reefs are in a profound state of decline, with limited growth of new coral, and overgrowth by slimy green or brown films of algae. Piles of coral debris are commonplace. The water is often muddy, and the plethora of life has departed. Reefs are no longer magical places. There is absolutely no argument about it, the decline in coral reefs is human inflicted. To this point, the largest culprit is probably pollution, but that will change in coming decades. Enter Ocean Acidification, a process that has already begun, tied to our excessive CO2 emissions, and one that will accelerate soon. Corals cannot grow once the pH drops below a certain level, and if we don't act fast, that level will approach by mid-century. Reefs have survived the two largest mass extinctions the Earth has faced, but they may not survive the mass extinction humans are causing.
Ecosystems, such as reefs, are governed by a delicate balance of interactions between animals and plants. Yin and Yang. If that balance is upset even slightly and one part of the ecosystem is favored at the expense of others, havoc can break out. The recent surge in harmful algal blooms along many coasts and the outbreaks of massive jellyfish in the western Pacific Ocean are signals of Yin or Yang, but not both. Our oceans are getting out of whack.
In the latter part of Module 6, we learned about changes in circulation and health of the oceans that are predicted to occur with climate change. In this module and Modules 8-11, we continue to address the impacts of climate change on natural systems. These changes are very much central to the issue of sustainability of the planet and its populations. "Sustainability" has a variety of definitions, and, in particular, the meaning for environmentalists is substantially different from the meaning for businesses. Regardless of where you might be coming from, sustainability means the preservation of society and our way of life. Most directly, we are concerned with maintaining the needs of people today and in the future, and this very much hinges, as we will see in this module, on sustaining the life support systems of the planet.
Global warming and an array of environmental changes resulting from human activities are already causing profound impacts on organisms across the spectrum of the marine food chain. Warming of the ocean and subtle changes in its chemistry are combining with pollution and overfishing to alter the habitat of many marine creatures. In the near future, these habitats look to be further impacted, and potentially destroyed, with possibly devastating biological and economic consequences, including very negative impacts on people. The goal of this module is to learn about three very different but equally significant impacts of climate change and human activity on life in the ocean: ocean acidification, red tides, and blooms of jellyfish.
To set the stage, watch the Award Winning video Sea Change, produced by Craig Welch.
Video: Craig Welch on "Sea Change" in Indonesia (2:32)
Here in the Coral Triangle in Malaysia, Thailand, Indonesia, Papua New Guinea, this part of the world relies heavily on coral reefs and fish that will be affected by ocean acidification for its food. So, we decided to find a community that relies on ocean fish for its protein more than some others. We wound up in this village of former sea gypsies in the South Sulawesi region of Indonesia. It is a stark and amazing way to live, and an incredible place to work as a journalist. The first thing you notice, of course, is that people here live in houses built on stilts above the water. But the next thing you notice, right away, is that everything here is loud, between the wind and the waves and the coughing diesel engines. People here are just used to constant shouting. It’s hard to explain how far removed this place is from the Western world. Just getting here from Seattle took six flights and a three-hour boat ride. We stayed on nearby Hoga Island, at a mostly empty research station, where monitor lizards fought over kitchen scraps and the walking paths were frequented by banded sea crates, a deadly type of ocean snake. Our translator Immon connected us with a boatman, and every day we paid that boatman, Duda, to take us to the stilt village to visit with his neighbors. Duda, like most villagers, never let an opportunity on the water pass without attempting to catch some fish. Talking to some of the more senior villagers, such as Ambelia, required working with multiple translators. The poverty at times was overwhelming. We met a widow who made her living getting paid to collect dead coral and to stack it below people's homes for support. The everyday risks were sometimes hard to ignore. We saw adults who had lost limbs to dynamite fishing accidents. Adults and children alike daily traverse crumbling boardwalks. Some of the homes were connected to these boardwalks by a single flimsy, treacherous log. Our attempts to cross provided villagers no end of amusement. But the Sama people, often referred to as Bajau, were open to us from the start. They shared their homes and their lives without question. No matter what happens with climate change in ocean acidification, it's clear that the people who live here don't have many options. They will continue to go out on the water and fish every day, and the women will go to the marketplace and sell that fish wherever they can, because, for the moment at least, they don't appear to have any other options.
Goals and Learning Outcomes
Goals and Learning Outcomes
Goals
On completing this module, students are expected to be able to:
- describe the physical, chemical and biological controls on the livelihood of coral reefs, calcareous plankton, dinoflagellates, and jellyfish;
- explain how human activity is causing ocean acidification, coral bleaching, red tides and blooms of jellyfish
- interpret evidence for the decline in reef health and its relationship with changing sea surface temperature
Learning Outcomes
After completing this module, students should be able to understand the following topics:
- projected change in pH in 2100 under SRES A2
- significance of pH, omega
- dissolution and precipitation reactions
- diversity of reef environments
- biology of corals, polyps, hard and soft corals, reproduction, role of zooxanthellae
- changes in calcification in the Great Barrier Reef
- causes and effects of bleaching
- coralline algae and their significance
- impact of acidification on plankton, pteropods, foraminifera, and coccolithophores
- causes of HABs
- life cycle of dinoflagellates and diatoms
- general impacts of toxins on fish and humans
- Pfiesteria controversy
- origin of CyanoHABs
- origin of giant jellyfish blooms
- impact of giant jellyfish blooms on coastal ecosystems
Assignments Roadmap
Below is an overview of your assignments for this module. The list is intended to prepare you for the module and help you to plan your time.
| Assignment | Location | |
|---|---|---|
| To Do |
|
|
Ocean Acidification
Ocean Acidification
Background
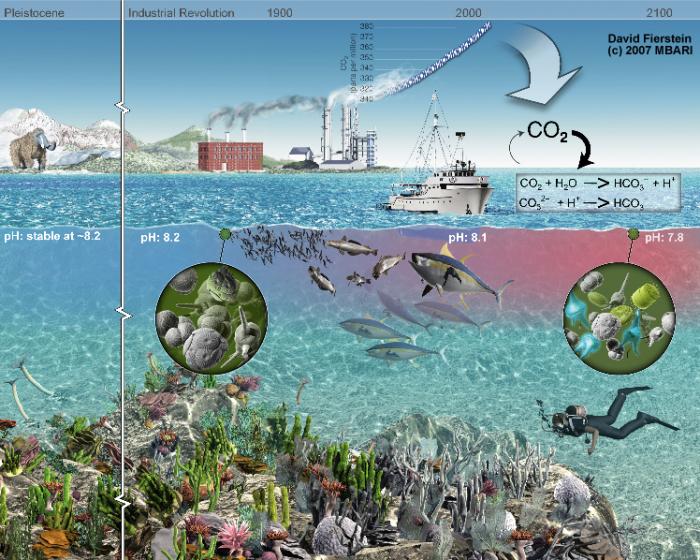
Increasing levels of CO2 in the atmosphere are slowly causing the surface of the ocean to become more acidic. This is because the ocean absorbs some of the CO2, forming a weak carbonic acid. At present, the ocean absorbs about a third of fossil fuel emissions, but this amount is likely to increase to 90% in the future. Over the last century, the average pH of the ocean has decreased, and there are hints that the current levels are beginning to impact organisms that make their shells out of the minerals aragonite and calcite (both composed of CaCO3). Aragonite is more susceptible to dissolution in more acidifc waters than calcite. Coral reefs that are made of the mineral aragonite and are particularly vulnerable to ocean acidification. A recent study has found, for example, that the area of coral covering the Great Barrier Reef in Australia has been cut in half since 1985. However, coccolithophores and foraminifera, organisms that serve a vital role at the base of the marine food chain that are composed of calcite, are becoming increasingly susceptible. Moreover, the future appears to be even more bleak; some CO2 projections suggest by the year 2100 there will be a 150% increase in the ocean’s acidity compared to preindustrial times. Here we review the chemical changes in seawater that result from increasing CO2, and then we discuss the impact on reefs and planktonic organisms in the ocean. Finally, we discuss the evidence for acidification in ancient oceans and its impact on life in the past.
The following video provides a thorough overview of the potential impact of acidification on the oceans.
Video: Acid Test: The Global Challenge of Ocean Acidification (21:34)
[MUSIC PLAYING]
SIGOURNEY WEAVER: The ocean keeps secrets. Hidden beneath its waves are worlds within worlds. Over billions of years, the ocean has created endless varieties of life, life that enchants us, that sustains us. And despite our science, life that mystifies us still.
STEVE PALUMBI: The oceans are an incredible place full of the most amazing kinds of life, life that you can never imagine really working. Things that if somebody just thought of them and showed them to you, you'd think, that's ridiculous. Nothing like that could ever live. But it does.
BRUCE STEELE: In 35 years of diving, it's quite a picture. I've spent my life on the bottom of the ocean with black sea bass. I've seen white sharks underwater. I've been in schools of bait that would be so big that they will dark out the sun. I can only hope that the ocean maintains that vitality. It's an incredible place of mystery, and it's something that's beautiful beyond description.
LISA SUATONI: People rely on the oceans in so many ways. Some ways are obvious, like food, recreation, transportation.
STEVE PALUMBI: They clean our shores. They protect our coastlines from storms.
LISA SUATONI: The oceans regulate climate and provide the world with most of its oxygen.
SIGOURNEY WEAVER: But we are now certain of one awesome fact. The ocean's power to create life is rivaled by our own power to destroy it.
[MUSIC PLAYING]
Scientists refer to ocean acidification as the other carbon problem. The first, of course, is global warming.
LISA SUATONI: People have heard about global warming for years, but it's only over the past five years that experts really understood that the carbon dioxide is causing a problem for the oceans as well. And what's worrisome is it hasn't even been on our radar.
SIGOURNEY WEAVER: Carbon dioxide pollution is transforming the chemistry of the ocean, rapidly making the water more acidic. In decades, rising ocean acidity may challenge life on a scale that has not occurred for tens of millions of years. So we confront an urgent choice, to move beyond fossil fuels or to risk turning the ocean into a sea of weeds.
KEN CALDEIRA: When we burn coal, oil, and gas, we introduce carbon dioxide into the atmosphere. But the atmosphere touches the ocean over 70% of Earth's surface. [THUNDER] So this carbon dioxide we're putting into the atmosphere we are also getting into the ocean.
SIGOURNEY WEAVER: Carbon dioxide, or CO2, exists naturally in our atmosphere. Plants need it to grow. Animals exhale it in every breath. But carbon dioxide is also a byproduct of burning fossil fuels. And in large amounts, it is a dangerous pollutant. Since the Industrial Revolution, the ocean has absorbed roughly one-quarter of the carbon dioxide produced by burning fuels. Scientists once thought this beneficial. After all, that carbon dioxide would otherwise accelerate global warming. But what happens when so much carbon dioxide, 22 million tons of it each day, mixes with ocean water? In terms of chemistry, the answer is simple. It becomes an acid.
LISA SUATONI: Since the Industrial Revolution, the ocean acidity has increased by 30%. With mathematical models, scientists have demonstrated that if we continue to pollute as we are now, the ocean acidity will double by the end of the century compared to pre-industrial times. That is a big problem.
SIGOURNEY WEAVER: Scientists only recently stopped to think about what this would mean for life in the ocean. Thousands of ocean species build protective shells to survive. Some are so prolific they can be seen from space. These organisms create their shells, which can be paper thin, by drawing certain molecules from the water around them. But rising acidity depletes those molecules.
KEN CALDEIRA: So by removing the essential building block for shell formation, it's making the organisms work a lot harder to build their shells. And that means they have less energy to get food. They have less energy to reproduce. And eventually, the organism can no longer compete ecologically. The surprise is how sensitive some marine organisms are to this increased acidity from carbon dioxide.
SIGOURNEY WEAVER: And when acidity gets too high, shells dissolve.
STEVE PALUMBI: We're changing the basic rules of everything. And because of that, a lot of organisms may not be able to survive.
KEN CALDEIRA: Already we've seen water showing up off the coast of northern California that's acidic enough to start actually dissolving sea shells. It's thought that this kind of corrosive water showing up will become more and more common.
BRUCE STEELE: Most of the West Coast of North America's shellfish-- that's Dungeness crabs, lobsters, mussels, oysters, sea urchins, shrimp-- all those life forms are at risk.
KEN CALDEIRA: By mid-century, if we continue emitting carbon dioxide the way we have been, entire vast areas of both the Southern Ocean and the Arctic Ocean will be so corrosive that it will cause seashells to dissolve.
SIGOURNEY WEAVER: Scientific models show that in just a few decades we will profoundly alter the ocean's chemistry. Such conditions haven't existed since the extinction of the dinosaurs. Recreating those conditions so quickly could leave many ocean animals unable to adapt. What if shellfish could no longer build shells? Would they cease to exist? Perhaps. Shelled creatures such as corals and plankton play a key role in the ocean food web. Terrapods are a kind of plankton that live all around the world and in great abundance in polar waters. Terrapods are especially vulnerable.
SPEAKER 6: Should I focus in that?
VICTORIA FABRY: Yeah, maybe right in here.
SPEAKER 6: Right in there?
VICTORIA FABRY: Mm-hm. We're looking at terrapod shells, which are planktonic snails with a calcium carbonate shell, that we collected from Antarctica this past winter. And you can see it looks like there's this lip where it may have already started to dissolve and kind of curled over. Because that's what it looks like when it dissolves. It kind of melts, almost like a candle and wax melting. The shell thickness along that leading edge right here is less than one micrometer thick. These are the thinnest terrapod shells I've ever seen.
SIGOURNEY WEAVER: There's growing alarm that higher acidity will extinguish creatures like terrapods that are a basic food source for fish. In many parts of the world, fish are a basic food source for people.
STEVE PALUMBI: So you can't just worry about the big things in the ocean. You have to worry about what they eat and where their food comes from. If the smallest things in the ocean are affected by ocean acidification, then it ripples all the way up the food web, making the largest things in the ocean even more endangered.
[WHALE SONG]
SIGOURNEY WEAVER: As individual strands disappear, the entire food web becomes weaker, more vulnerable, less beneficial to humans.
OVE HOEGH-GULDBERG: And many of us are concerned about what that means for the Earth's marine ecosystems, but also for the many millions of people that depend on these systems for their food and income.
SIGOURNEY WEAVER: Ocean acidity will rise most quickly in cold water regions and areas where deep water wells up to the surface.
LISA SUATONI: That is disconcerting because it coincides with the regions of the most productive fisheries in the world.
BRUCE STEELE: I'm a fisherman. Every single day I have to make a prediction where I'm going to go fishing, whether I'm going to find fish where I go. And every single day the decisions I make make the difference between whether I stay a fisherman and make a profit. I can make predictions. I think these things are dire problems. Either we change what we're doing on land, or it will have profound effects on fisheries as we know them.
SIGOURNEY WEAVER: Marine life that might withstand warming temperatures or rising acidity may succumb when confronted by both. Coral reefs already struggle to survive in warming waters. Rising ocean acidity puts them in double jeopardy.
OVE HOEGH-GULDBERG: We know that coral reefs are particularly sensitive to ocean acidification. And the reason for that is that corals are unable to form their skeletons as quickly as they used to. And reefs are starting to crumble and disappear. We may lose those ecosystems within 20 or 30 years. And in those structures live an estimated million species. One in every four species in the ocean lives on a coral reef. We've got the last decade in which we can do something about this problem. But it's very, very clear that if we don't start to deal with it right now with very, very stern cuts to emissions, we are going to condemn oceans to an extremely uncertain future.
KEN CALDEIRA: We're really in the last decades of coral reefs on this planet for at least the next, say, a million-plus years unless we do something very soon to reduce CO2 emissions. We're moving from a world of rich biological diversity into essentially a world of weeds.
STEVE PALUMBI: Today we're in a really remarkable history of the ocean. 100 years ago it was inexhaustible. You couldn't touch it. You couldn't harm it. In 100 years, it might be dead.
KEN CALDEIRA: When people say there was high CO2 100 million years ago so we have nothing to worry about, that high CO2 was achieved over a slow process of millions of years. And if we achieve high CO2 over millions of years, the Earth will be able to handle it. If we achieve high CO2 over decades, the ocean is in big trouble.
SIGOURNEY WEAVER: Earth is the only planet we know of where life exists. To understand our own actions, we sometimes need to view them in a larger context. Planet Earth was formed 4 and 1/2 billion years ago. 3 and 1/2 billion years ago, life began. 250 million years ago, dinosaurs appeared. And 200,000 years ago, homo sapiens. Within that framework, human civilization is brand new, our industrial society but an instant. Yet in that instant, we have altered the course of nature. We have heated the Earth's surface, acidified its oceans, and consumed much of its natural habitat. Now something extraordinary looms, a mass extinction of animals and plants caused not by volcanic eruption or the collision of a meteor but by the actions of one species, ours.
KEN CALDEIRA: If we destroy these ecosystems, it will take millions of years for them to recover. It's as if somebody, just because they had the ability to do it, decided to run through the Metropolitan Museum with a knife slashing the great paintings of the world.
SIGOURNEY WEAVER: We have created this problem. We should be able to solve it. The ocean, after all, is resilient. Given the chance and enough time, it can heal itself. So how can we give the ocean that chance? Marine protected areas like national parks in the sea shelter ocean life from industry and development. Sustainable fishing practices allow fish stocks to regenerate. The ocean can better defend itself against rising acidity and temperature if its systems are healthy.
LISA SUATONI: To make the oceans more resilient to these changes, we need to do a better job of keeping the oceans healthy. That means restoring depleted fish populations, establishing marine protected areas all around the globe, and reducing pollution, particularly nutrient pollution in the coastal zones.
STEVE PALUMBI: Solving those local problems gives those ecosystems a chance to survive, a chance to make it through while we solve the global problem. We know how to solve the local problems of marine ecosystem health. We know how to solve the global problems. The question is, will we?
[JET ENGINE]
SIGOURNEY WEAVER: The only way to stop acidification is to emit less carbon dioxide. Our Industrial Revolution began more than two centuries ago. Technology has advanced rapidly since then. But we still make energy as we have for hundreds of thousands of years, by setting things on fire. Often, we squander the energy we make, using more than necessary to accomplish our goals. But now we know how to use energy more efficiently, how to do more with less.
RALPH CAVANAGH: There was a time when people thought about energy efficiency and conservation as sacrifice, doing without-- dark homes, shuttered economies. That is emphatically not what we're talking about. We're talking about getting dramatically more work out of less energy with better technology. Those energy efficiency solutions are particularly promising because the whole world will want to adopt them. If we take that initial step, we will also, in addition to reducing carbon pollution, have the very welcome dividend in the form of economic stimulus because we will be reducing energy bills.
SIGOURNEY WEAVER: We know how to capture energy cleanly from sunlight, wind, tides, and the heat of the Earth's core.
RALPH CAVANAGH: Imagine that you're living in a house that gets some of its electricity from its own solar panels, feeds some of that back into your own vehicle when it's plugged in at night, provides you with energy services-- and maybe this is the most important single piece of it-- at costs below those you're paying now. That double dividend was never more needed by the US and world economy than it is right now.
SIGOURNEY WEAVER: We are on the verge of a green industrial revolution, a revolution that will expand our economy, protect our resources, and give us real energy independence. There is much we don't know about how carbon pollution will affect our world. Still, we have to choose. We can go on as we have, forcing future generations to survive somehow without the vast ocean resources that have sustained us. Or we can move beyond fossil fuels, securing a future that works for all of us, for all living things. What will we choose?
[MUSIC PLAYING]
KEN CALDEIRA: You're not going to use this. I'm going to just say it, though. What makes a Greek tragedy a tragedy is that you can see it coming. Oedipus goes and marries his mother and eventually tears his eyes out. And you want to tell him, look, no, don't marry your mother. You can stop this process now.
STEVE PALUMBI: And you think, if I could only go back and change that one little tiny instant, then things would have been different. I think we're in that instant right now.
KEN CALDEIRA: We sit by feeling almost helpless because we see this unraveling, leading to its tragic end.
LISA SUATONI: Researchers are predicting significant substantial changes in the next two decades to our oceans. So it is not necessarily a problem we're passing off to future generations. It's a problem that we're generating for ourselves.
OVE HOEGH-GULDBERG: I think what gets me up in the morning is that I don't want to see coral reefs disappear on my watch. And I know that my fellow scientists feel this way as well. So we feel compelled to communicate the message that this is a serious issue and that changes that haven't happened for millions of years are starting to happen right before our eyes. I think it's important to point out that it's not all over yet.
BRUCE STEELE: I don't expect people to understand what terrapods or what various forms of plankton are likely to survive or not survive. But I do expect our policy-makers to take serious an issue that is so closely tied to life on this planet and the future of life on this planet. I have hope. You can't fish and not have hope.
Chemistry 101
The ocean contains a massive reservoir of dissolved CO2, hundreds of times more than in the atmosphere, and, actually, by contrast, the amount derived from fossil fuel burning is relatively modest. Since the beginning of the industrial revolution, about 340 to 420 petagrams carbon (a petagram or Pg is 1015 grams) in the form of CO2 has been emitted to the atmosphere, with about a third of that amount absorbed by the ocean, approximately 118 Pg. Seawater today may already contain more CO2 than at any time in many millions of years.
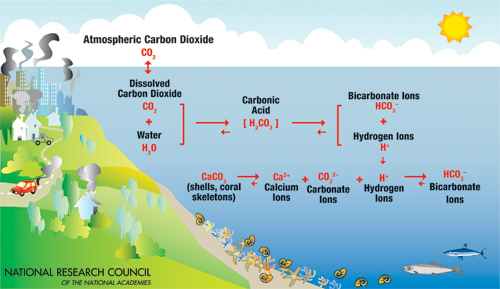
As we discussed in Module 5 on the Carbon Cycle, the absorption of CO2 in the ocean forms weak carbonic acid (H2CO3). Some of this acid dissociates in seawater releasing H+ ions, which make the water more acidic, as well as HCO3- (bicarbonate ions) and CO32- (carbonate ions). This reaction is as follows:
CO2(aq) + H2O = H2CO3 = H+ + HCO3- = 2 H+ + CO32-
Going back to your elementary chemistry course, you might remember that a pH of greater than 7 is regarded as alkaline whereas a pH of less than 7 is acidic. Surface ocean waters have a pH of between about 7.9 and 8.3, which means that they are, by definition, alkaline. Anthropogenic CO2 is thought to have decreased the mean pH of the ocean by 0.1 unit since 1800. This may not sound like that much, but more ominous is the projection that if CO2 levels continue to rise unabated (i.e., projections based on SRES A2 “business as usual”, pH levels will drop a further 0.3 by 2100. As we will see below, in parts of the ocean, these levels would be extremely damaging to organisms that build their skeletons out of CaCO3, which is very sensitive to CO2 addition.
CaCO3 is the dominant material used by invertebrate organisms to build their skeletons. There are two different minerals made of CaCO3, known as polymorphs: calcite and aragonite. These minerals have the same composition but different crystal lattice structure and thus their properties and behavior in seawater differ, including their ability to dissolve. To understand how CaCO3 dissolves and precipitates, we need to introduce a term Ω that represents the saturation state of the water. Where waters are highly saturated with respect to CaCO3 and Ω is high, calcite and aragonite are less likely to dissolve than where these waters are less saturated or even undersaturated and Ω is low. Likewise, calcite and aragonite are more likely to precipitate under higher Ω values. The dissolution and precipitation reactions are as follows:
Dissolution reaction: CaCO3 (solid) = Ca2+ + CO32-
Precipitation reaction: Ca2+ + CO32- = CaCO3 (solid)
An increase in CO2 from the atmosphere presents a double whammy for skeletons formed from CaCO3, both aragonite and calcite. The H+ ions and carbonate ions (CO32-) that derive from the dissociation of carbonic acid combine to form bicarbonate ions (HCO3-). This rapid reduction in available carbonate ions decreases Ω and limits calcification by organisms with aragonite- and calcite-based skeletons. However, here we need to dispel two myths. The first myth is that the precipitation of CaCO3 is directly controlled by pH. In fact, precipitation is affected principally by the decrease in CO32, which is coincident with the addition of H+ ions, and reduction in pH. The second myth is that precipitation of CaCO3 can occur in any water that is oversaturated with respect to the particular CaCO3 mineral. In fact, both corals and coccolithophores have been shown to have difficulty calcifying in environments when waters were actually oversaturated. Different organisms can calcify at very different Ω values, but for most the decrease in saturation that results from decreasing CO32- content is a direct threat to calcification. Fiinally, it is key to move that aragonite is more susceptible to dissolution than calcite. Thus, shells made of the CaCO3 polymorph aragonite, including the corals, will be the first to dissolve, followed by those made of the polymorph calcite.
The following video explains the threat of ocean acidification to the calcareous plankton.
Video: The Other Carbon Dioxide Problem (3:57)
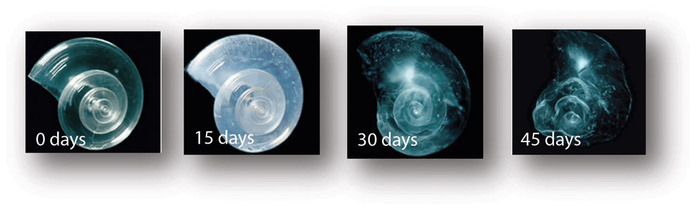
Marine pteropods, or sea butterflies, are found throughout the ocean and are a major food source for fish and other organisms. In laboratory experiments, this pteropod shell dissolved over the course of 45 days, in seawater adjusted to an ocean chemistry projected for the year 2100.
What changes in the ocean might be putting pteropods at risk? The burning of fossil fuels has caused levels of carbon dioxide in the atmosphere to rise. This carbon dioxide, or CO2, doesn't just stay in the atmosphere. Roughly 30% of it is absorbed by the ocean. When co2 reacts with water, it forms carbonic acid, which quickly releases a hydrogen ion, or H+. The more hydrogen ions, or the lower the pH, or more acidic, a solution is. As CO2 levels increase over time, seawater will progress towards the acidic end of the pH scale, a process called ocean acidification.
We can make projections of this change in the ocean pH over time. It is expected that later this century, if CO2 emissions continue at the current rate, the average pH of the ocean will drop from 8.2 to 7.8. This relatively small decrease in pH may not seem significant, but it could impact many species of marine life. For pteropods, corals, and other species that depend on shells and exoskeletons, ocean acidification will lead to a decreased availability of dissolved calcium and carbonate, the chemical building blocks they use to make their shells and skeletons. If ocean chemistry changes as expected, shells with calcium carbonate mineral structures may begin to dissolve, depending on where they live in the ocean.
How do we know that ocean acidification is happening? Scientists at NOAA, and across the world, have measured these chemical changes over the past 40 years during research cruises, taking hundreds of thousands of ocean water samples. Shown here are the results from cruises in 1991 and 2005, where the availability of the shell mineral was sampled. In both years the highest concentration of available shell minerals were found in the surface waters, as shown by the dark yellow colors. Deeper waters naturally have very low availability of these minerals. However, when we look at how these values have changed between 1991 and 2005, we see that the availability of the shell mineral decreased much more in the surface waters, as indicated by the blue colors. In other words, ocean acidification is impacting shallower areas much more than deep areas. Shallow waters are where the bulk of the ocean productivity occurs, including the most diverse and economically important species and habitats. And these changes aren't occurring in just certain parts of the ocean, they're occurring throughout the entire ocean.
Today, repeated research cruises and permanent sampling stations continue to monitor changes in pH and availability of calcium carbonate minerals. As the ocean approaches a critical transition between shell building and shell dissolving, food webs in the world's oceans could be impacted. Phytoplankton and zooplankton, like pteropods, form the basis of most oceanic food chains. Coral reefs form the foundations of the most diverse marine habitats. And shellfish, such as oysters and crabs, and finfish, such as salmon, support economically important fisheries in many of the world's coastal communities. People depend on the viability of these species to ensure a healthy future of our own.
The saturation of CaCO3 in the oceans is also a function of temperature and pressure. A delicate balance exists between the production of CaCO3 via the formation of skeletons in the shallow part of the ocean and the dissolution of this aragonite and calcite in the colder and deeper realms of the ocean where waters are less saturated. In most parts of the ocean, undersaturation occurs far below the surface. However, recent increase in dissolved CO2 is leading to a shoaling of the saturation horizon of CaCO3, and, in the future, this will impact especially the organisms that live at depth or in colder waters as well as those that make their shells of the mineral aragonite, which is more soluble in seawater than calcite.
Check Your Understanding
Coral Reef Introduction
Coral Reefs
Introduction
Coral reefs have existed for hundreds of millions of years and provided a habitat for some of the richest diversity on the Earth’s surface. They are the marine version of tropical rainforests. Reefs harbor a slice of the marine food chain, all the way from tiny autotrophic protistans (autotrophs fix carbon through photosynthesis) to large, predatory fish. Hundreds of millions of humans live near reefs and receive important resources from them. Reefs host productive fisheries; they also provide protection to low-lying coastal areas from storms and are vital for a number of key habitats, including mangrove forests.
Examples of Reef Corals
Examples of Deep Water Corals
The organisms that have constructed reefs, largely corals, have evolved over time, and with that change so have the locations of reefs and the dynamics of the reef community changed. Over their long history, reefs have had several intervals of crisis; in particular, they almost ceased to exist at the Permian-Triassic boundary, where over 90% of marine species became extinct, and during the Cretaceous about 100 million years ago when giant clams took over these structures for several tens of millions of years. Both of these ancient times were potentially characterized by ocean acidification. However, reefs have been remarkably resilient over geologic time and generally have been able to adapt to environmental change. For example, as we will see in Module 10, they are able to grow fast enough to keep up with very rapid rates of sea level rise.
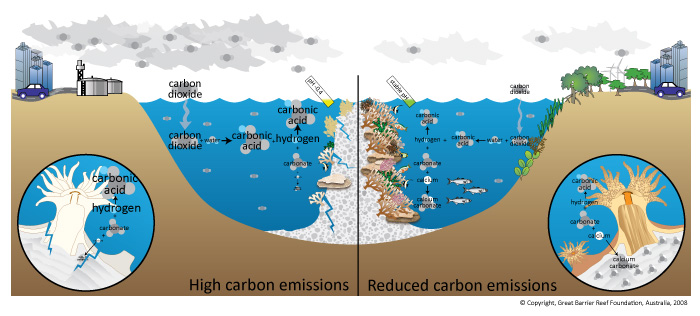
With this background, recent human activity has placed reefs in as precarious a position as at almost any time in their history. The last fifty years have witnessed an extremely dramatic decline in the health of many of the major reefs around the world, including reefs of the Caribbean, the Bahamas, and the Florida Keys as well as those in the Indian and Pacific Oceans, including the massive Great Barrier Reef of Australia. The outlook for these rich and complex ecosystems is about as bleak as any ecosystem on Earth. As it turns out, ocean acidification is one of several environmental threats to reefs, with warming, pollution, overfishing and physical destruction all exerting major threats to reefs in the future. As we will see, acidification is perhaps the greatest of all of these threats long term.
Coral Biology, Growth and Evolution of an Atoll
Coral Biology, Growth, and Evolution of an Atoll
Before we start, let's consider how reefs grow. The main organism that constructs modern reefs, the coral, includes a number of species belonging to the Cnidaria, a phylum of organisms that uses stinging cells to capture their prey. Modern corals are colonial structures of millions of individual polyps that grow primarily in shallow and clear tropical and subtropical waters, restricted to these areas by light levels and temperatures as well as by nutrients.
Both types of coral reproduce both sexually and asexually. Asexual reproduction involves simple cell division, or budding, and takes place within the colony, whereas sexual reproduction involves the release of gametes into seawater. This is an amazing process that for many species happens once a year, timed by the lunar cycle. The fertilized egg forms a larval planula that settles before forming a new colony.
Video: Coral Reefs 101 National Geographic (3:54)
Coral reefs, their bright, vivid colors, can be seen in tropical ocean waters around the globe. Beyond their brilliant appearance lies a hidden significance. Coral are animals. Though they may look like colorful plants, coral are, in fact, made up of tiny animals called polyps. These invertebrates can range from the size of a pinhead to a bit larger than a basketball. Each polyp consists of a soft, saclike body topped by a mouth covered in stinging tentacles. To protect their soft bodies and add support, the polyps secrete limestone skeletons, or calicles. Corals are mega builders. Polyp calicles connect to one another, creating a colony that acts as a single organism. As colonies grow over hundreds and thousands of years, they join with other colonies and become reefs that can grow to hundreds of miles long. The largest coral reef is Australia's Great Barrier Reef, which began growing about 20,000 years ago. Coral reefs are some of the most diverse ecosystems on Earth. Though they cover less than 1% of the ocean floor, coral reefs are home to 25% of all marine creatures. It's been estimated that up to two million species inhabit coral reefs, rivaling the biodiversity of the rain forest. The reefs provide rich habitat that helps protect young fish as they grow. Coral are translucent. Coral reefs get their rainbow of colors from algae, or zooxanthellae, that live in their tissue. Though corals use their tentacles to capture some food, most of their food comes from the algae they house. When coral become stressed by pollution or other factors, they evict their algae. Coral bleaching results, revealing corals' white skeletons. Coral provide a window to the past. As coral grow, their limestone skeletons form layers, similar to tree rings, that vary in composition and thickness based on ocean conditions at the time. With some coral reefs growing for thousands or even millions of years, scientists can study these layers to reveal what the Earth's climate may have been like in the ancient past. Unfortunately, climate change is putting coral's future in danger, along with the millions of species that inhabit the reefs and the half-billion people that rely on reef fish for food. Warming waters result in prolonged coral bleaching that kill coral reefs or leave them vulnerable to other threats. Without significant action on climate change, our oceans could lose many of their colorful reefs by the end of the century.
Large reef structures including fringing and barrier reefs, as well as atolls, represent the growth of these colonies over many thousands or millions of years.
Evolution of an Atoll (1:20)
Many reefs develop in shallow, tropical, volcanic islands. In this situation, the reef evolves in a manner first described by Charles Darwin. Initially, very few coral polyps will settle upon the volcanic island, but once the volcano becomes dormant, the reef will rapidly colonize the island in a manner called a fringing reef. In the fringing reef, the reef lies right next to the island itself. As the island becomes further eroded, the reef becomes a barrier reef and is separated from the island by a shallow lagoon. This is often what we think about when we think about a tropical island with a reef.
When the island itself becomes completely eroded, we have what is known as an atoll, in which the reef lies along a ring separated by a shallow lagoon in the middle. Many reefs that lie along continents are either fringing reefs, where the reef is right next to the land, or barrier reefs, like the Great Barrier Reef in Australia, where the continent and the reef are separated by some distance.
Examples of modern corals living in symbiosis with dinoflagellate algae called Zooxanthellae
The algae live within the coral polyp and receive CO2 from the polyp that it requires for photosynthesis. Through photosynthesis, the Zooxanthellae convert the CO2 to O2 and provide this vital gas to the coral as well as crucial nutrients. This is important because by removing CO2, the Zooxanthellae drives up saturation which facilitates calcification in the coral skeleton. Reefs are made up of much more than corals and their algal symbionts. Other organisms, including coralline algae (algae that secrete high magnesium calcite and conduct photosynthesis on their own), are active framework builders in modern reefs. Today, sponges, sea anemones, sea urchins, a diverse array of fish, and many other organisms live within reefs, some playing a vital role in building reefs and keeping them healthy and others taking advantage of their decline.
Examples of Sea Urchins, Sponges, Sea Anemone and Coral Reef
Check Your Understanding
Coral Bleaching and Calcification
Coral Bleaching and Calcification
Although, as we will see, ocean acidification is only just beginning to affect reef growth, there is already another very serious affliction in many reefs around the world---coral bleaching. One of the key changes in corals over the last few decades has been an increase in the frequency and severity of bleaching events. As we have discussed, shallow-water coral species live in symbiosis with algae called a dinoflagellate or Zooxanthellae whose colorful pigments give living corals such beautiful colors, purple, brown, green, yellow, and more. As discussed the symbiosis is key to the coral, the Zooxanthellae removed CO2 from the water, boosts the saturation, and facilitates calcification of the coral. When temperatures become too hot, however, the Zooxanthellae can’t tolerate the heat and it temporarily or permanently leaves the coral leading to a bleached white color.

Bleaching today is common when temperatures increase very slightly (by about 1°C) in summer months and thus is often an annual summer event. Bleaching is accompanied by slower growth and increased coral mortality. The response to bleaching differs considerably between different species. Some species are able to recover normal growth rates quite rapidly, whereas others are more vulnerable and unable to recover. Bleaching also changes the ecology of a reef, promoting the growth of algae that blanket corals, making the recovery of corals more difficult.
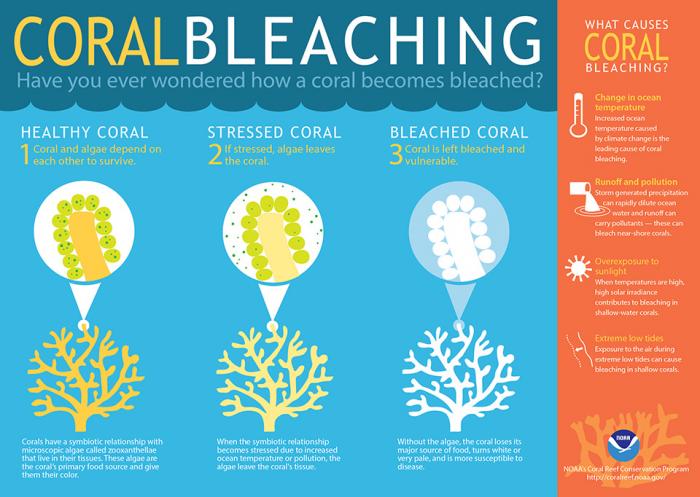
Click here to see a text description.
Coral Bleaching - Have you ever wondered how a coral becomes bleached?
- Healthy coral - coral and algae depend on each other to survive: Corals have a symbiotic relationship with microscopic algae called zooxanthellae that live in their tissues. These algae are the coral's primary food source and give them their color.
- Stressed coral - if stressed, algae leaves the coral: When the symbiotic relationship becomes stressed due to increased ocean temperature or pollution the algae leave the coral's tissues
- Bleached coral - coral is left bleached and vulnerable: without the algae, the coral loses its major source of food, turns white or very pale, and is more susceptible to disease
What causes coral bleaching?
- Change in ocean temperature: increased ocean temperature caused by climate change is the leading cause of coral bleaching
- Runoff and pollution: storm-generated precipitation can rapidly dilute ocean water and runoff can carry pollutants–these can bleach near-shore corals
- Overexposure to sunlight: when temperatures are high, solar irradiance contributes to bleaching in shallow-water corals
- Extreme low tides: exposure to the air during extreme low tides can cause bleaching in shallow corals
A major coral bleaching event took place in the Florida Keys in summer 2023 when water temperatures rose to 101 deg F and there was widespread bleaching. It is too soon to determine the long-term impacts of this bleaching on the reefs in the Keys but the images are dramatic and the future does not look promising.

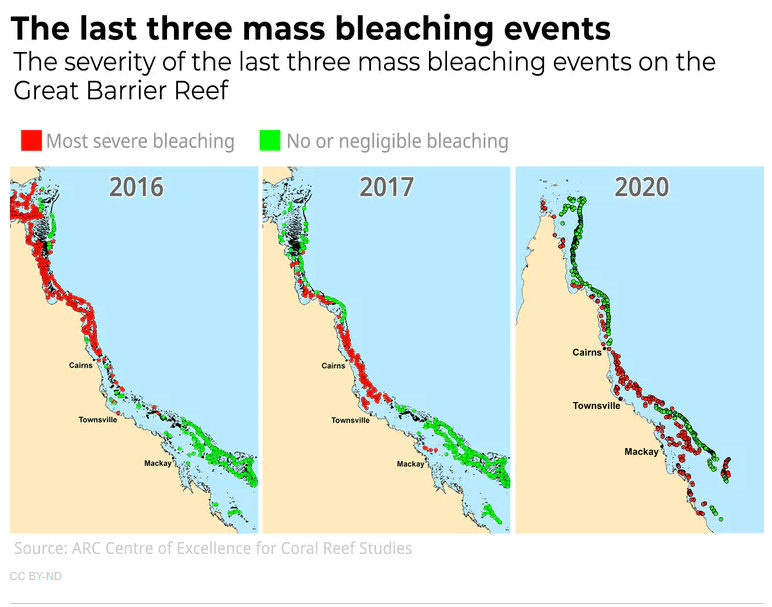
One of the most significant bleaching events took place in 2016 and 2017 in the Great Barrier Reef. This catastrophic event caused by warmer than average water caused up to 50% coral mortality in some areas. Bleaching was also extreme in 2020 and you can see from the maps below that it extended southward which in the southern hemisphere means it afflicted cooler waters. The extent of bleaching shocked ecologists and is clearly a window into the future. Now it seems every year, bleaching is a threat to the Great Barrier Reef, and with the warm El Nino conditions, the Austral summer of 2023-2024 may be the most severe bleaching event ever. One scientist has remarked that the reef looked like it had been "carpet-bombed".
Deep Water Corals
Not all corals live in shallow water. A very diverse group of corals are able to thrive in the deep realms of the oceans. These corals lack Zooxanthellae and are able to calcify at cooler temperatures and lower levels of saturation than exist in the shallower waters. The number of known deep-water species has increased dramatically with new methods of exploring the deep oceans, including ROVs (remotely operated vehicles) and AUVs (autonomous underwater vehicles).
Examples of Deep Water Corals
These organisms exist in isolated patches at depths down to 2000 meters. In these conditions, the corals grow considerably slower than shallow-water reefs. They feed on zooplankton and in some cases use chemicals coming out of the sea floor for a source of nutrition. Like their shallow-water counterparts, deep coral reefs provide a habitat for a diverse array of creatures. However, at the same time, it is clear that deep corals are in a particularly precarious situation, as we will see later on.
Corals and Calcification
The growth rate of corals, known as the rate of calcification, corresponds more closely to aragonite Ω rather than any other variable. Cores of corals from the Great Barrier Reef indicate a 14% decrease in calcification rates from 1990 to 2005. Corals generally maintain a high Ω at the site of calcification, but with decreasing CO32-, precipitation of aragonite requires more energy, hence the decrease in calcification rate. Overall, the net calcification rates of reefs decrease to zero when the Ω of aragonite is <1, and thus we can predict that worldwide reefs will switch from actively growing via calcification to shrinking via dissolution when CO2 doubles to 560 ppm (the year 2050 in emission scenario A1B). The same logic leads to predictions of decreases in calcification rates of 10-50% by 2050. Experiments show that modern coral species have very different abilities to grow in water with lower saturation, with some species able to continue growing while others can't.
Experiments are one of the best ways to forecast the future, but they have significant limitations. In particular, it is impossible to replicate natural growing conditions in the lab, and further, experiments are conducted at intervals that are significantly shorter than the changes that are occurring in nature.
Field studies and experiments often show very different results. Strangely, field results indicate a much higher susceptibility of growth rates to decreasing levels of saturation than do laboratory experiments. These studies suggest that there is much left to learn about calcification and how it will change in the future. At the same time, it is clear that there is significant variability between species; some species having a much greater likelihood of survival in the future. Moreover, even within individual species, it is apparent that some reefs likely have the ability to survive better than others. Factors such as community composition, growth rate, nutrient levels, and local variations in seawater chemistry and sediment composition will also play a vital role in determining which reefs will survive.
Since the coralline algae are more tolerant of colder waters, they have a very widespread distribution in the oceans, extending from the tropics to polar regions. The algae are also able to use low light levels for photosynthesis and therefore live considerably deeper than most corals. Their distribution suggests that they have great potential to adapt to variable environments. As it turns out, however, they may be in a more precarious position than the corals.
The recent decrease in CO32- has also begun to lower calcification rates of the coralline algae. These species are composed of high Mg calcite, which is the most soluble form of CaCO3 (more so than low Mg calcite and aragonite), so they are particularly prone to ocean acidification. Experimental work confirms that calcification of the coralline algae is particularly sensitive to CO2 levels with growth rates slowing significantly, and actually, dissolution beginning at moderately high CO2 contents.
Perhaps the coralline algae will be the “canary in the coal mine” for the dissolution of all framework structures under rising CO2. For the corals, it is very apparent that the threat varies considerably from reef to reef. Deep-water corals are in particular jeopardy because calcification rates are lower in colder waters, because saturation levels at these depths are lower, and because, in the future, saturation stands to decrease more rapidly in deep than in shallow waters. However, the fate of reefs in the tropics is also likely to vary significantly.
Future Predictions
Making predictions about the exact impact of ocean acidification in the future of coral reefs of all types is inherently difficult. Acidification will occur in parallel with other deleterious effects such as bleaching and sea level rise. Species that are most susceptible to the effects of bleaching may turn out to also be more susceptible to extinction. For example, by impacting zooxanthellae, bleaching impacts calcification and thus may exacerbate the impact of acidification. However, these same corals also have the ability to recover from bleaching events more quickly, possess shorter generation times, and thus may have the ability to evolve more rapidly to tolerate bleaching in the future. At the same time, nutrient levels in reef environments have and will continue to decrease as stratification of the upper ocean increases, and this will put considerable strain on future reefs. Temperatures, nutrient levels, local rates of sea level rise, and human activities are factors that will likely decide which reefs are the most vulnerable, possibly with acidification pushing the most threatened systems over the edge into extinction.
Probably the largest uncertainty with respect to the future of reefs is whether corals will be able to adapt to lower saturation levels, increased temperatures, and lower nutrient levels, and if so, how rapidly. As we have seen earlier, corals have shown great resilience in surviving numerous environmental threats in the past. So, why do reefs recovered after times of environmental stress in the past, at times when temperatures were even warmer than they are today, and CO2 levels higher, appear to be in such dangerous territory today? The answer to this question is that in the past, temperature and CO2 perturbations occurred slowly enough that ultimate increases in weathering (remember feedbacks we discussed in Module 3) essentially decreased the rate of CO2 addition and buffered the ocean with CO32- before extinction occurred. What concerns scientists is that warming today and CO2 addition are rapid enough that the weathering feedback will lag the decrease in saturation by several thousand years.
So, we are left with a lot of questions: will the rates of saturation decrease and temperature rise be too rapid for modern species to adapt? Will algae take over the niche of shallow-water corals and dominate the low pH oceans of the future? Or will a few species of coral and possibly coralline algae develop the ability to calcify rapidly enough to survive the current threats and take over the niche of species that do not? Will Zooxanthellae themselves be able to adapt and assist corals in calcifying? Finally, when will feedbacks, largely through weathering, come into play and make conditions more favorable for calcifying organisms? For some of these questions, it’s a matter of wait and see. However, ongoing research should shed light on others. For example, the genetics of coral populations are currently being explored to understand the ability of corals to adapt to environmental change.
At this stage, however, any outcome is possible for corals, ranging from complete extinction by late in the 21st century to the adaptation to a new set of environmental parameters. Ultimately, like many elements of the ecosystem, the fate of reefs may rest on how well we manage CO2 emissions in the future.
Check Your Understanding
Lab 7: Reef Ecology
Lab 7: Reef Ecology
The goal of this lab is to:
- observe and compare the health of reef by exploring different parts of the ocean.
Introduction
First, you will be watching videos from the Catlin Seaview Survey as well as photos to learn how to determine the health of reefs. Then you will be looking at before and after photos of reef bleaching events. Finally, you will be looking at the risk to reefs in the future.
Please watch the videos below. It is easiest to watch the videos in your browser (click button top right of Google Earth) in full screen mode. To move around, insert your cursor in the videos to manipulate the camera and stop on particular items of interest or to change direction. The best way to move the camera is with your keyboard arrows (side to side) and cursor for up and down.
The goal of this part of the lab is for you to show you can identify different types of coral as well as overall health of the reef at different locations. Make sure you have read the material on reefs in the module before attempting to complete the lab. Below are the different types of coral for you to identify. In addition, we show pictures of algae that colonize reefs as well as reef damage from storms. Healthy corals show a variety of colors from the different algal symbionts. Unhealthy corals show fewer colors, more algal colonization, more breakage and often are bleached white. Remember, algae are some of the key markers of an unhealthy reef.
Photographs of different types of coral
Photographs of algal colonization
The following images show some of the range of morphologies and colors of colonizing algae
Photographs of reef damage from storms or people
Files to Download
ReefsAllFin.kmz [17]
Prep and Instructions
Load the ReefsAllFin.kmz file [18]. The locations of reefs of interest are shown with flags. The videos are shown with diver markers, the still photographs are shown with wave markers. The videos run best if you open them a browser such as Firefox (see Google Earth window, top right). Before submitting your lab, let’s begin with the practice. Make sure you do this part of the lab to get comfortable with the tasks you will be asked to do and to receive feedback about your answers. Watch the videos at the following locations and answer the questions below. Make sure you maneuver up and (especially) down, as well as side-to-side.
Practice Questions
Part A
In this section, you will be looking at the health of reefs using their color, the presence and abundance of algal overgrowth and the and the presence of bleaching. Go to the Belize reef (click on the flag in Google Earth, then open the address in your browser). Look around the reef and answer the following questions.
- Is there living coral? (look for different colored coral) (Yes/No)
- Is there brain coral? (Yes/No)
- Which types of coral do you see?
A. Table bottom
B. Table bottom and staghorn
C. Staghorn and brain
D. Table bottom, staghorn, and brain - Is there any bleached coral? (Yes/No)
- Is there any algal overgrowth? (Yes/No)
- How would you describe the health of the reef?
A. Very healthy
B. Moderately healthy
C. Not healthy
Part B
In this part of the lab, you will compare photographs from before and after major events that have impacted reefs. Answer the questions about the changes in the abundance of different types of coral, algal overgrowth, or percent bleaching.
Go to Lizard Island. Please look at the two pictures and answer the following questions.
- Which picture has the healthiest reef? (upper/lower)
- What is most diagnostic of the health of the upper photograph?
A. The healthy reef has very tall coral
B. The healthy reef has a lot of different colors
C. The healthy reef has bleached corals - What is most diagnostic of the health of the lower photograph?
A. The unhealthy reef is bleached
B. The unhealthy reef is growing quickly
C. The unhealthy reef is covered by algae
Acidification: Effect on Plankton
Acidification: Effect on Plankton
Introduction
Corals and coralline algae are not the only organisms highly susceptible to ocean acidification. Coccolithophores, foraminifera, pteropods, three very different groups of plankton (a term that refers to organisms that float passively in the upper ocean) are also threatened by increasing atmospheric CO2 levels. Pteropods, often called sea butterflies, are tiny snails made of aragonite that thrive in shallow waters and play a particularly important role in polar ecosystems.
Pteropods

Because of the susceptibility of aragonite to decreasing saturation levels combined with the effect of temperature (calcification requires more energy in cold water) on saturation, pteropods may cease to exist in polar latitudes by the middle of the 21st century. In some polar areas, these organisms account for over 60% of the zooplankton biomass, thus their extinction or migration to warmer regions could have major repercussions to organisms up the food chain.
Foraminifera and Coccolithophores
The coccolithophores and foraminifera are constructed of low-Mg calcite and therefore are more stable than pteropods under conditions of increasing CO2.
As we have learned earlier, foraminifera are groups of zooplankton with habitats both near the surface of the ocean (planktonic foraminifera) and on the ocean bottom (benthic foraminifera).
Foraminifera
The planktonics are sensitive to changes in CO2, have shown subtle changes in shell mass during the Pleistocene in response to CO2 fluctuations, and have already begun to get lighter in response to recent CO2 increase. This recent thinning of shells is likely a result of the effect of decreasing CO32- on calcification by foraminifera. Interestingly, many planktonic species also harbor dinoflagellate symbionts that play a role in calcification. Changes in calcification appear to be a threat to the planktonic foraminifera, but we do not yet know how serious this threat is. However, we do know that the foraminifera are also threatened by the potential loss of their dominant source of food, the coccolithophores, as a direct result of CO2 addition.
Coccolithophores
Coccolithophores are ubiquitous in the oceans, essentially serving as the dominant species of phytoplankton in vast regions of the open ocean that are characterized by lower nutrient levels. For this reason, considerable attention has been devoted to the potential effects of increasing CO2 on the coccolithophores.
Coccolithophores are haptophyte or golden brown algae (similar to diatoms) that produce tiny calcite scales known as coccoliths during certain phases in their life cycle.
The plates are several microns in diameter and can remain attached to the cell covering the soft organelles with a protective shield or break off after they are fully grown. Coccolithophores reproduce asexually, and, given the right conditions have the potential to multiply rapidly. When conditions are suitable, coccolithophores can form blooms of millions of cells per liter of seawater. These organisms are consumed by foraminifera and copepods, and transported to the bottom of the ocean as marine snow.
Video: Marine Snow (00:33)
Much of the calcite and aragonite transported to the seafloor is delivered by particles known as marine snow. Marine snow is aggregates, compounds of calcite and aragonite, that are held together by mucus secreted by phytoplankton and bacteria. In addition to marine snow, calcite and aragonite particles are also transported by fecal pellets that are excreted by zooplankton.
Because of their prolific production and global distribution, coccolithophores are a vital part of the carbon and carbonate cycle of the oceans. Modern coccolithophorids are dominated by the species Emiliania huxleyi, a species with very small (1-2 micron) and delicate coccoliths. Due to their size and ecology, coccolithophores are inherently more difficult to study in the natural environment than are corals. However, samples can be collected using filters and cores of sediments can be studied to determine the effects of past changes of CO2 on coccolithophore morphology. Coccolithophores can also be cultured in the laboratory where CO2, CO32- and pH levels can be altered to observe the effect of these variables on calcification. Although the results of both field and lab studies are by no means simple, it appears that in the case of E. huxleyi, increasing CO2 and decreasing CO32- has the effect of causing thinner coccoliths with smaller masses. In addition, laboratory studies show that such conditions also lead to malformations in E. huxleyi coccoliths, which are a potential sign of difficulty in calcification. Such trends of decreasing mass are by no means universal, however. Some lab experiments and field collections in particular environments show that species other than E. huxleyi actually grow thicker in low CO32- conditions and even E. huxleyi recently has increased in mass in parts of the ocean. More study is required to determine the precise outcome of the coccolithophores in a high CO2 world. However, the current signs generally point to significant reduction in the rate of calcification, which could lead to significant changes both in marine ecosystems and in the carbon cycle.
Coccolithophores have a spectacular 220 million year fossil record. This record allows paleontologists to observe the effects of past climate change, including increasing CO2, on the livelihood of this group of plankton. Ancient global warming events, including those at 120 million years before present and 55 million years before present, have been gleaned for evidence of ocean acidification in the morphology of coccolithophores. The event at 120 million years shows evidence for a decrease in coccolith size, but this change is not apparent in the 55 million year event. As the species existing during these ancient events was entirely different from those at the present time, it could be that ancient species responded in a different fashion from those living today. Alternatively, the changes in ancient plankton assemblages may be in response to environmental variables other than CO32-, for example, temperature and nutrients. The coccolithophores, like the corals, have been able to survive intervals of great ecological upheaval in the past. However, given that the rates of modern environmental change are so rapid compared to ancient events, we cannot assume that these same groups will have the ability to adapt to the changes that are to come. Moreover, while the coccolithophores appear to be less endangered by increasing CO2 than the corals, largely as a result of their mineralogy, the impact of the decline of these vital algae would likely be even more devastating to the oceans.
Red Tides
Red Tides
Red tides are common events in warm and polluted coastal oceans. They form when dinoflagellate algae explode to huge population levels. Because the dinoflagellates have red plastids, the waters literally turn red. Dinoflagellates take advantage of harsh environmental conditions that kill off other organisms. As you will find out in the pages to follow, these tides can be major public health hazards.
I remember that the grouper was delicious. Blackened, spicy, with an ear of corn and some slaw. On a white paper plate. I polished it off with a Heineken. It was a warm early January day, a seafood festival in Florida City. We went canoeing after lunch. I remember waking up the next morning with an intense thirst and extreme nausea. When I tried to get out of bed, I sprawled on the floor, my left side was completely paralyzed! I rolled to the sink, crawled up, and poured myself a glass of water, I gulped it down, and to my horror, it felt boiling hot! The neurological symptoms soon went away, but nausea and fatigue lasted weeks, I lost at least ten pounds. The doctors took that long to diagnose the problem---I had Ciguatera poisoning. That delicious grouper had ingested a lot of toxic dinoflagellates and transmitted the toxins to me. My first experience with harmful algae.
Red tides represent one of the most serious threats to coastal ecosystems today. Most red tides result from the input of an excessive amount of nutrients from fertilizers, sewage, and soils of nearby land areas to bays, estuaries, and shallow seas. These nutrients cause explosive growth of microscopic species of algae, a number of which carry toxins that are harmful or even lethal to other organisms. Red tides can be caused by major storms such as hurricanes, which cause excess runoff from the land and resuspension of the seed stages of the algae.
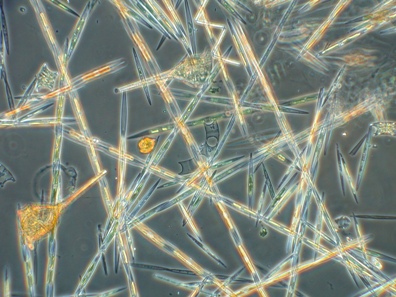
Red tides occur when dinoflagellates, and rarely diatoms, grow in massive quantities in surface waters. The photosynthetic organelles of these organisms, known as plastids or chloroplasts, are red, or golden brown in the case of diatoms, and the profusion of cells in surface waters imparts a red or brown color. Some of the culprit dinoflagellate and diatom species produce by-products that are highly toxic to many other organisms living in the coastal zone, all the way from fish to turtles to large mammals such as dolphins, manatees, and whales, as well as to humans. In some cases, shellfish or small fish such as sardines that consume the plankton are not harmed by the toxin but concentrate it for organisms that feed on them, a process known as bioaccumulation. Ingestion of the toxins can result in developmental, immunological, neurological, and reproductive damage of the host organism. For this reason, red tides are also known as harmful algal blooms (HABs). In fact, we will use the term HAB here because these events are not associated with ocean tides, because many HABs are not associated with a red color, and because blooms of dinoflagellates are often not harmful.
Examples of Dinoflagellates and Diatoms
Harmful algal blooms are a global phenomenon and have increased in frequency in the last thirty years. Part of the increase may result from awareness of the phenomenon, but increasing pollution is also considered responsible. There are a number of reasons why climate change may further increase the occurrence of HABs. For examples, the increase in the frequency of large storms such as hurricanes will lead to greater runoff and input of nutrients from land. In addition, changes in temperature, wind patterns, upwelling, and stratification will alter the distribution of species. Because the degradation of a large amount of cellular material produced in HABs consumes oxygen, HABs can result in hypoxic or anoxic conditions.
Check Your Understanding
Harmful Algal Bloom 101
Harmful Algal Bloom 101
Dinoflagellates are a group of microscopic single-celled organisms or protists that are dominantly autotrophic (i.e., primary producers). Interestingly, many species are also mixotrophic, having the ability to ingest their prey as a source of energy. Some species are entirely heterotrophic, lacking chloroplasts or plastids, and have been termed carnivorous. In fact, there have been reports in the scientific literature that some species have the ability to consume fish after having paralyzed them with neurotoxins. These claims are controversial but have given dinoflagellates a near-mythical reputation among the oceanic plankton.
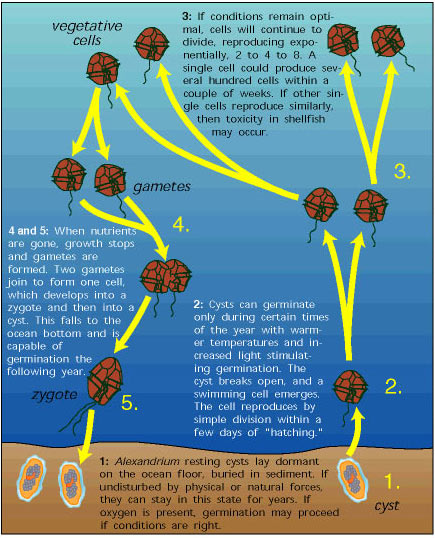
Life Cycle of a Single Algal Cell
- For certain red tide species, a resting cyst lays dormant on the ocean floor, buried in sediment. If undisturbed by physical or natural forces, it could stay in this state for weeks, months, even years.
- Warm temperatures and increased light cause the cyst to germinate. It breaks open and a swimming cell emerges. The cell reproduces by simple division within a few days of "hatching."
- If condition remains optimal, cells will continue to divide, reproducing exponentially 2 to 4 to 8 to 16. A single cell could produce 6000 to 8000 cells within one week.
- (and 5.) When nutrients are gone, growth stops and gametes are formed. Two gametes join to form one cell, which develops into a zygote and then into a cyst. This falls to the ocean bottom, ready to germinate.
Diatoms and dinoflagellates are most common in the coastal oceans but also have the ability to live in freshwater environments and in intermediate salinity environments where fresh and marine waters mix in estuaries. They are the most prolific group of primary producers in the ocean. Dinoflagellates have a highly complex life cycle that consists of an alternation between a motile stage and a resting or cyst stage. In short, dinoflagellates enter the resting stage via sexual reproduction when conditions in the surface ocean are not suitable for them to thrive. They can remain dormant for weeks, months or years before they “excyst,” when surface conditions improve, reproduce vegetatively, and populate the surface ocean. Excystment and repopulation are triggered by changes in temperature, light, or oxygen levels or even resuspension of cysts by storms. Dinoflagellates generally thrive when nutrient levels are elevated, and, under conditions of extremely high nutrient levels, cell division can be so rapid that extremely high cell counts (millions of cells per milliliter of seawater) are reached, resulting in red tides. The cyst stage acts as a very effective mechanism for seeding blooms.
Dinoflagellates
The following video summarizes the life cycle of the dinoflagellates.
Video: Life Cycle of the Dinoflagellates (1:27)
The basis of red tides are dinoflagellates. Dinoflagellates are incredible organisms that have the ability to reproduce extremely rapidly, producing large amounts of toxin that compose red tides. The original stage of the dinoflagellate is the cyst. The cyst is the stage that resides in the sediment, and it can stay in this location for many years. When there's a trigger in the overlying water column, often increase in nutrients or increase in temperature, the dinoflagellate will excyst into the vegetative stage. The vegetative stage is a stage that reproduces asexually and this reproduction can occur extremely rapidly, producing many millions of cells in the overlying water column in a short period of time. When conditions change, often decreasing amounts of nutrients or lower light levels, cooling temperatures, the dinoflagellate will reproduce sexually producing gametes. And these gametes will form the zygote stage that will then, in turn, form the cyst stage which will fall to the bottom of the ocean and reside in the sediment once again, for years, until conditions change again.
Dinoflagellates have a broad range of different ecologies. As we saw earlier in the module, they can be endosymbionts of corals, facilitating calcification in the host colony. They have this same role in foraminifera and radiolarian, a group of siliceous zooplankton.
Diatoms
Diatoms are autotrophic protists that produce a delicate, microscopic test of opaline silica.
They are non-motile, and, for most of their life cycle, they reproduce asexually. Many nearshore diatom species also have a resting stage akin to the dinoflagellates, allowing them to exit the surface zone when conditions are unfavorable for their growth. This may occur in winter when surface waters are cold or at times when nutrients are depleted. The resting spore stage actually may resemble the vegetative stage of dinoflagellates. Like the dinoflagellates, diatoms are able to reproduce extremely rapidly by simple cell division, and this allows them to rapidly dominate the surface ocean when nutrients are readily available.
Video: Diatom Life Cycle (1:38)
Normal diatom reproduction is dominated by asexual or vegetative cell division. In this type of reproduction, an individual diatom will divide and the new diatom cells will reside in two new diatom shells, one which has one valve from the parent, and one which has the other valve from the parent. Because this is the way that the cell divides and the shells form, over time the shells become smaller and smaller and smaller. And as a result, if this can process continued unabated, diatoms would become minuscule. When the cell reaches a certain size it becomes fertile and it reproduces sexually, releasing eggs and sperm and fusing into a new diatom cell, that forms a stage called an auxospore. The auxospore stage is naked, in that it does not contain a silica shell, and this auxospore stage expands and forms an initial cell, which is much larger than the fertile cell size. In this way, the vegetative cycle begins again with the size of the diatom restored to its original level.
Species that are harmful belong to the pennate diatoms that are long and thread-like and have the ability to attach to a host, although the relationship is not symbiotic. Both dinoflagellates and diatoms cysts can move around the oceans by currents, storms, dredging of the ocean bottom, and when cysts act as ballast on ships or even higher-level organisms. Toxins are not known in the cyst stage of either group.
Other HAB Species
Other HAB Species
Of some 60 or so species that cause red tides, only a handful is known to be toxic. Dominant dinoflagellate HAB genera include Alexandrium, Karenia, and Pfiesteria. The diatom genus most commonly associated with HABs is Pseudo-nitzschia. Each of these genera produces a different toxin and thus has a different role on organisms further up the food chain. Next, we discuss some of the HAB species in detail.
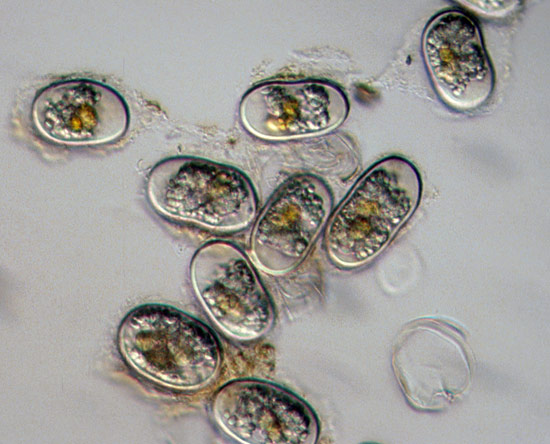
Alexandrium spp. is the dominant taxon in coastal regions of New England and eastern Canada but it is also found from California to Alaska.
It is a heterotrophic dinoflagellate that produces a saxitoxin, one of the most powerful known types of neurotoxins. These toxins destroy the function of nerve cells and can thereby cause paralysis. Saxitoxins are most effectively concentrated by shellfish such as clams, quahogs, mussels, scallops and oysters that filter large volumes of seawater to acquire their nutrition. Although the saxitoxin does not harm these shellfish, even in small quantities, the toxin can be extremely dangerous for humans, resulting in a serious illness known as paralytic shellfish poisoning (PSP). The saxitoxin attacks the human nervous system within 30 minutes of ingestion with symptoms that may include numbness, tingling, weakness, partial paralysis, incoherent speech, and nausea. In severe cases, the toxin can lead to respiratory failure and death within a few hours. Alexandrium spp. toxins have also been harmful to whales, sea otters and birds.
The following videos describe the causes and impacts of red tides as well as possible antidotes for shellfish poisoning.
Video: Florida's toxic red tide is a perfect storm for the Gulf Coast (7:47)
PBS Reporter: The growth of an algae, known as red tide, has exploded in Florida this year, growing bigger and lasting longer than years before. It has killed huge numbers of marine life and dealt a hard blow to the gulf coast economy. As William Brangham reports from Sanibel Island, there are many causes driving this red tide, including warmer waters tied to climate change. And there are other questions about what role humans are playing. It's part of our weekly series on the leading edge of science, technology, and medicine.
William Brangham: This is now a typical morning on Florida's gulf coast. Not a tourist in sight, just work crews cleaning up the daily toll of dead fish. They were killed by red tide, an almost annual bloom of algae in the Gulf of Mexico. At high levels, the algae release a neurotoxin that's deadly to marine life. It poisons them or makes it so they can't breathe. But this year's bloom, which actually began last fall, has been particularly bad.
Female reporter: It now spreads across 130 miles of coast.
Male reporter: Thousands of dead fish floating along Lido Beach.
Male reporter: This red tide bloom is being called the worst in more than a decade.
Brangham: In the last few months, red tide has killed dolphins, sea turtles, manatees, even a 26-foot whale shark.
Veterinarian: ...some deformities here.
Brangham: Veterinarian Dr. Heather Barron runs the Clinic for the Rehabilitation of Wildlife on Sanibel Island. These baby loggerhead turtles just came in. Barron suspects that they or their mom was poisoned by the red tide.
Dr. Barron: There's probably not a whole lot we're going to be able to do for them.
Brangham: She says they've seen four times the number of sea turtles poisoned by red tide this year.
Dr. Barron: It is overwhelming and catastrophic the number of patients that are coming in. We've had as many 100 patients come in in two days, all affected with red tide, way out of the ordinary. And so, when that happens, one of the things that you sometimes have to do is to be able to triage those animals and decide who are you likely to be able to save, who needs help the most, and who are you probably not going to save regardless.
Brangham: This pelican came in near-death several weeks ago. But after intensive treatment, he seems to be turning the corner. So too are these sea turtles.
Dr. Barron: I think, even if you're not a bunny hugger like I am, you know, even if you don't care about the wildlife, you should care about what that means for your health and your children's health, and your pet's health, and your food supply's health, it's not going to be just wildlife that' going to be affected by this.
Brangham: While the red tide is obviously devastating to wildlife, it's also difficult for humans as well. The toxins emit a terrible smell, they burn your eyes, they burn your throat, and as you can see, nobody is on these beaches. This year's red tide has been brutal to tourism, blanketing more than 100 miles of southwest Florida. Here on Sanibel Island, the Chamber of Commerce estimates eleven million dollars in lost income in just the first half of August.
Trasi Sharp, Over Easy Cafe: We're probably off about another 45-50% of what we usually do this time of year. Thank you guys for coming in.
Brangham: Traci Sharp owns and runs the Over Easy Cafe. I's a small breakfast and lunch spot in Sanibel, just a few blocks off the beach. But, it's scary.
Trasi Sharp: Well, somehow we'll get through this. We're hoping it doesn't last at this pace for too long.
Ben Bury: You couldn't drive 100 years without passing a thousand or ten thousand dead fish.
Brangham: Ben Bury is a charter boat captain on Sanibel. He makes his living taking tourists out to fish, and picnic, and swim - things that many people just don't want to do right now.
Ben: I know as a captain, my business for the month of August, was down somewhere around 80%
Brangham: 80%?
Ben: Yeah. I don't think there' anyone that's in the tourism business in our area right now, or in any business that's on the water, that isn't really suffering right now.
Man holding equipment: This is an optical oxygen sensor.
Bringham: In fact, things have been so slow for Bury, that he's volunteered his boat to scientists who are studying the red tide, like his friend Dr. Rick Bartleson. Bartleson's an ecologist with the Sanibel Captiva Conservation Foundation. He's checking the water at various spots, measuring the concentration of red tide cells. He says with a usual red tide, one that will kill some fish, you'd see a hundred to two hundred thousand red tide cells per liter of water. But this year, the red tide has been ten times worse.
Dr. Bartleson: Instead of 200 thousand, we're seeing 2 million or 20 million cells per liter.
Brangham: I mean, that's a huge jump up.
Dr. Bartleson: Right, the other day we saw 40 million cells per liter about a kilometer off the coast.
Brangham: While the red tide has been disastrous for the gulf, this summer it's been compounded by a bloom of blue-green algae in Florida's freshwaters. It's created a perfect storm for fish, wildlife, and humans and it's leading some to wonder if one is exacerbating the other. The Everglades Foundation's Steve Davis took us along the Caloosahatchee River about 70 miles east, to show us one source of the problem. For thousands of years in Florida, water flowed down into Lake Okeechobee where it flooded out and south through Florida's everglade marshes. But as farming expanded around the lake, Lake Okeechobee was damned up, forcing that water to the east and to the west.
Steve Davis: The massive volumes of freshwater that are released from the lake to the Caloosahatchee, they result in harm.
Brangham: Davis says this causes two problems. One, that freshwater kills crucial habitat that needs saltier water, and two, that water is so polluted with runoff from farms and towns that it could be delivering a huge nutrient-boost to the red tide.
Davis: When you think about red tide, it's really another bloom of algae, but offshore. And these blooms of algae, they require large loads of nutrients in order to sustain themselves. It's kind of like thinking of a wildfire that requires fuel in order for it to continue burning.
Brangham: Davis and others argue water needs to again flow south, like it once did, to address both the green-blue algae and any potential impact on red tide. But the powerful agricultural industry in Florida, has successfully stymied these efforts before.
Ad on the TV: It's killing sealife, battering our economy, and making people sick, and it's fair to blame Rick Scott.
Brangham: And now, ahead of the mid-term elections, water quality is a central issue.
Tras Sharp: We need the state and federal help. There's industry and farming that dumps into the water, s of Okachobee but also it's on everyone - fertilizers in people's yards, the dumping of waste of various companies along the river. We just need to do something or this is going to last generations.
Ben: That's all I care about this election, is I want people that are going to get out there and do something and make things better, that are gonna make it possible for me to continue to do what I love for a living.
Brangham: A strong storm or cold front could break up this current red tide, and Tropical Storm Gordon, which just passed over Florida, may have done just that. But it's just a few months until the next potential bloom reemerges from the Gulf. For the CBS News Hour, I'm William Brangham, Sanibel, Florida.
Female Reporter: Such important reporting. Thank you, William.
Video: Prized Science Episode 4: Taming the Red Tides (4:28)
[MUSIC PLAYING] PRESENTER: "And all the waters that were in the river were turned to blood, and the fish that was in the river died and the river stank. And the Egyptians could not drink of the water of the river."
The Book of Exodus in the Holy Bible may give us the first account of a red tide. Red tides are a phenomenon in which certain pigmented algae, toxic algae, undergo population explosions. They bloom in enormous numbers, staining the water reddish brown. Toxins or poisons released by the algae periodically kill millions of fish and cause billions of dollars in losses to the global commercial fishing industry.
If health officials detect a red tide, they ban fishing for oysters, shrimp, and other shellfish. If they didn't, unsuspecting consumers would get hit with a virtual tidal wave of discomfort in the form of neurotoxic shellfish poisoning. This terrible form of food poisoning causes noxious nausea and vomiting, but tingling of the mouth, arms, and legs, as well as poor coordination and other very unpleasant symptoms. With no specific treatment, victims may suffer for days on end.
That may change thanks to the research of Michael Crimmins, a scientist at the University of North Carolina in Chapel Hill. His work focuses on brevetoxin A, the compound that causes neurotoxic shellfish poisoning. It could even lead to the world's first antidote for this painful condition. For that research, the American Chemical Society awarded Crimmins its 2010 Ernest Guenther award in the chemistry of natural products.
Natural products are chemical compounds produced by plants, animals, and other living things. Natural products or substances derived from them have been the source of almost one out of every three of our prescription drugs. For more, here's Dr. Crimmins.
MICHAEL CRIMMINS: So brevetoxin A is a very complex structure that was isolated from red algae. And this red algae, the scientific name is karenia brevis. It creates these massive algae blooms in the Gulf of Mexico and the Pacific in various places. And these massive growths of algae create what is called red tides. And these red tides are these red algae that have grown sort of out-of-control over miles and miles and miles of ocean. They can be 50, 100 miles wide or long. And these algae produce a series of neurotoxic compounds, of which brevetoxin A is one of those compounds.
PRESENTER: When shellfish encounter algae in these red tides, they take up the toxins. People who eat contaminated shellfish then become ill. The algae can also produce a toxic mist which is swept along by the wind, like fog. People along the beach or in ships can become sick simply by inhaling this contaminated sea mist. In their quest to develop medicines to treat neurotoxic shellfish poisoning, Crimmins and his colleagues have been gathering knowledge about brevetoxin A and related toxins.
MICHAEL CRIMMINS: What we've been trying to do is to develop a synthesis of these compounds in the laboratory. While they're incredibly toxic, they're not really available in very large quantities from the natural sources. So we've been trying to develop a chemical synthesis of the compounds in an effort to perhaps develop analogs that would be effective antidotes to these compounds.
PRESENTER: Dr. Crimmins is talking about a potential cure for neurotoxic shellfish poisoning. For those who might encounter the toxin, that could reduce an ancient health threat of biblical proportions to a mere ripple.
Karenia is the dominant taxon causing red tides in Florida and Texas, but rarely species of Karenia have also been found up the east coast in North Carolina.
Examples of Karenia
It produces a toxin known as a brevetoxin (named after a species of Karenia, K. brevis). Like saxitoxins, brevetoxins damage nerve cells, leading to disruption of normal neurological processes and causing neurotoxic shellfish poisoning (NSP). In humans, gastrointestinal symptoms and a variety of neurological ailments result, but there are no known fatalities. However, in fish, the brevetoxins attack the central nervous system and cause respiratory failure. Karenia dinoflagellates are responsible for massive fish and bird kills in the Gulf of Mexico. The brevetoxins are colorless, odorless, and heat and acid stable, thus they survive food preparation.

The genus Gambierdiscus lives in tropical waters, usually in reefs, and produces a toxin known as Ciguatoxin that causes gastrointestinal problems followed by mild neurological symptoms. This syndrome is known as Ciguatera fish poisoning. Because the toxin is fat soluble, it gets concentrated up the food chain by bioaccumulation from seaweed to smaller fish than to larger fish. The larger fish, which are the most dangerous to eat because they have the highest toxin concentrations, include commercially available seafood such as grouper, snapper, and barracuda. Ciguatera is responsible for more human illnesses—estimated between 10,000 to 50,000 cases annually—than any other HAB toxin.

Pseudo-nitzschia and the species Nitzschia navis-varingica are common diatom genera in Californian red tides. These taxa produce domoic acid, which is concentrated by filter-feeding shellfish. This neurotoxin can also bioaccumulate in fish such as anchovies that feed directly on the diatoms. Domoic acid causes a variety of gastrointestinal ailments, memory loss and brain damage in humans and is hence referred to as amnesic shellfish poisoning. Rarely, the neurotoxin can be fatal. It can also affect marine mammals, causing seizures.
The species Pfiesteria piscicida and P. shumwayae have been the most common dinoflagellate species in red tides in estuaries and bays along the east coast of the US from Delaware to Florida. The species occur in environments where freshwater and saltwater mix and have not been reported from freshwater environments or the open ocean. Pfiesteria blooms are restricted to summer months. Species have been associated with massive kills of menhaden and other estuarine fish in the Chesapeake Bay and the Tar-Pamlico and Neuse River Estuaries in North Carolina. The fish in contact with Pfiesteria rapidly develop bleeding lesions and have skin actively flake off them, and it has been proposed that the presence of live fish stimulates the production of toxin in the dinoflagellate. Ultimately the open lesions may destroy gill function and lead to death. Nevertheless, the connection between fish mortality and Pfiesteria is still doubted by some scientists. In fact, the effects of Pfiesteria on fish and human health has been one of the largest and nastiest controversies in marine science over the last 25 years and it does not appear that the conflict is anywhere near over.
The following video explains some of the research on Pfiesteria.
Video: Pfiesteria Update: An Enduring Debate (4:57)
PRESENTER: In the spring of 1997, sick fish began showing up in the pound nets of watermen working along the Pocomoke River.
And soon after, some of those same watermen began showing up in doctors' offices. In these pound nets, floating among the sick fish, was a microbe called Pfiesteria piscicida, first identified in the wild by a North Carolina scientist named JoAnn Burkholder. She claimed this one-celled organism could sometimes release a toxin in the presence of fish, and that toxin could make people sick. Pfiesteria could also change shape, shifting through 24 life cycle stages from tiny zoospores all the way to large amoeba forms.
The search for more answers about this new organism has led to conflicting, even contradictory findings, kicking off complicated debates among marine scientists. When Wayne Litaker investigated Pfiesteria, he found a very simple life cycle with no amoeba forms at all. He used sophisticated molecular RNA probes as well as computer-controlled photography.
WAYNE LITAKER: So, you actually have the computer turn the microscope on every so often, turn the lights on, and take a picture, turn it off, and then you put it all together in a movie. And you can actually follow what's happening through time. We never saw any transformations. We saw a normal seven stage dinoflagellate life cycle. So, we didn't see any of those transformations whatsoever.
PRESENTER: Other researchers contradicted Burkholder's claim that Pfiesteria used a toxin to kill fish. At the Virginia Institute of Marine Science, Wolfgang Vogelbein announced evidence that Pfiesteria kills fish by directly feeding on them, not by releasing a toxin.
WOLFGANG VOGELBEIN: OK, what you're seeing here is the tail fin of an anesthetized fish.
These fish and these dinoflagellates are directly attacking the fish and feeding on the skin. It's that simple.
PRESENTER: Vogelbein put a filter between his hungry Pfiesteria and his tiny test fish, a filter that would keep any Pfiesteria out, but let any toxin pass through. A simple experiment with repeatable results. None of his fish died.
WOLFGANG VOGELBEIN: We can actually see what's happening, and we're finding out that there isn't an exotoxin here, at least not in our cultures.
PRESENTER: Fish were not dying from Pfiesteria toxin, but from Pfiesteria feeding.
WOLFGANG VOGELBEIN: This happens. You've seen it today, we've shown it to you. Firsthand. You've seen it. It happens. This is how fish die in our cultures.
Others say toxins kill their fish. Fine, let's do a comparative study, let's get to the bottom of it.
JOANN BURKHOLDER: Toxic Pfiesteria is not on trial here. Toxic Pfiesteria exists. There is no question. It's been cross confirmed in multiple laboratories.
PRESENTER: Andrew Gordon runs one of the laboratories where a Pfiesteria connected toxin is killing fish. His technique runs water from a toxic culture through a filter removing all the Pfiesteria cells, then tests this filter in Pfiesteria-free water on small fish.
ANDREW GORDON: I have no doubt that in these systems in the presence of Pfiesteria, that there is a soluble toxin produced. And we've seen it. I've had undergraduates do the experiment; they're seeing it; they're all over the country now. And they're believers because they've done the experiment themselves. It's a very simple experiment to do.
PRESENTER: A simple experiment with repeatable results. In most of his experiments, all of his fish died.
ANDREW GORDON: The toxin that's produced is Pfiesteria-associated. You never see it in the absence of Pfiesteria, and you see it in the presence of Pfiesteria. So, that's pretty clear.
PRESENTER: What is not so clear so far from all the studies in the laboratory is how much harm these toxins can cause in our rivers, either to fish or to people.
JOANN BURKHOLDER: We have a long ways to go, and we just don't understand what turns on and off toxin production in these creatures. I wouldn't be surprised if the so-called nontoxic or non-inducible strains of Pfiesteria make toxin. But just can't somehow turn it on or activate it. And we'll be finding that out, I think, in the next couple of years.
PRESENTER: By the time Pfiesteria blooms again in the Chesapeake, scientists may have more answers to some of these contentious questions.
At least part of the debate has been fueled by the popular press, who have focused attention on the organism after studies suggested that it was carnivorous. These studies indicated that Pfiesteria species ingested the skin of fish after it flaked off. In fact, lab studies have shown that when fish were left in tanks with Pfiesteria, the fish died within hours. For this to occur, however, the dinoflagellate and fish must be in direct contact. Without contact, the same studies show that fish suffered no ill effects. Some scientists alternatively point to water molds or fungi including the species Aphanomyces invadans as the pathogen cause of the ulcerative lesions, skin loss, and damage to gills. A. invadans and other fungi are universally present in fish with ulcers and skin loss. Also weakening the case for Pfiesteria, this genus is still known to exist in North Carolina estuaries, but fish kills have become less frequent recently. Moreover, where lesions on the fish menhaden were observed, nearby fish including catfish, perch, and carp were unaffected. These disparities have cast some doubt on whether Pfiesteria is harmful to fish at all. In fact, great differences exist among public health professionals, and warnings from different state agencies are in conflict.
Examples of Pfiesteria
The health impact of Pfiesteria on humans is also uncertain, as is the method of transmission of the potential toxin. Scientists working with Pfiesteria in the laboratory have suffered from long-term neurological symptoms, such as memory loss, fatigue, and dermatological problems, and fisherman in contact with Pfiesteria-related fish kills have also suffered from similar ailments. However, other groups of fishermen who have come in close contact with lesion-covered fish have not reported adverse effects. There are reports that the hypothetical Pfiesteria toxin is transmitted via aerosols.
Until recently, the missing link in the Pfiesteria conundrum is that the toxin produced by this organism has been elusive. However, in 2007 scientists in a government lab claimed the first positive identification of a toxin associated with Pfiesteria. Even with this identification, questions remain; for one, the toxin is unstable in the natural environment, and second, it is not been proven to have adverse health effects. Thus, the controversy about Pfiesteria is far from over. In all reality, a number of factors may result in the fish kills; in particular, the fish in estuaries may have already been under great stress from other biological agents (bacteria, viruses, fungi, parasites), exposure to chemicals (pollutants, toxins), suboptimal water quality, and rapid water temperature change, that have the potential to cause lesions to form.
Far less controversial than the relationship of Pfiesteria with fish kills are studies that directly relate fish kills to low oxygen levels caused by algal blooms. In a number of estuaries along the eastern US, warmer waters in summer combined with increased production by algae, as a result of increased runoff and eutrophication, lead to severely decreased oxygen levels and major fish kills without the involvement of a toxin.
Check Your Understanding
Cyanobacteria and CyanoHABs
Cyanobacteria and CyanoHABs
Finally, HABs are not always produced by dinoflagellates and diatoms. Cyanobacteria, or blue-green algae, another group of single-celled organisms, but one that is prokaryotic rather than eukaryotic, is also known to produce extremely potent toxins that can cause illness in fish, birds, and mammals including humans. Because of their potential to be harmful, this group is known as CyanoHABs. Cyanobacteria are some of the oldest species on Earth and are known to tolerate very tough conditions including hot, cold, salty waters and darkness. They live under ice sheets, near hydrothermal vents, and were some of the first organisms to colonize the ocedans after the massive asteroid that killed the dinosaurs.
Samples of CyanoHABs
Although the full scale of health effects of CyanoHABs on humans is not yet determined, the toxins may have gastrointestinal, respiratory, allergic, and neurological responses, and potentially lead to liver damage. In addition, prolific growth of cyanobacteria can block sunlight, which can harm other organisms, and use up oxygen which can lead to hypoxia and anoxia. As in the case of the HAB dinoflagellates, the growth of CyanoHABs may be stimulated by nitrogen loading from agricultural industrial runoff, as well as sewage disposal.
HAB Ecological Forecasting
HAB Ecological Forecasting
As we have seen, there is a great variety in the biology and ecology of HAB species. However, they share one major thing in common: all of them have the ability to wreak havoc on coastal fisheries. Since the growth of most if not all of the species directly responds to nitrogen loading, limiting the harm on fisheries will require significant changes in agricultural practices combined with modifications of drainage in coastal regions. Such changes will be difficult, if not impossible, to accomplish in the near term. Thus, the best strategies to deal with HABs focus on the integration of highly detailed algal sampling programs, ecological forecasts, and resource management. For example, if HABs can be predicted, then warnings can be issued and areas placed off-limits to fisheries. Such strategies are being employed in areas where HABs are common, including the Gulf of Maine, the Gulf of Mexico, and the Pacific Northwest.
HAB forecasts integrate ecological models, based on the physics, chemistry, and biology of nearshore and offshore regions, with satellite data and in situ measurements of cell counts and toxin levels. For example, models can be used to predict the development and movement of a HAB in the region of interest. Models can be used to identify HAB triggers (i.e., nutrients or temperature), likely areas of cyst seedbeds, likely bloom toxicity based on cell density, and progression of the toxin through the food chain, as well as the ultimate decline of the HAB.
Giant Jellyfish
Giant Jellyfish
A new threat to fisheries around the world has developed over the last decade---a surge in the number of jellyfish in coastal waters. The most dramatic of these outbreaks is in Japanese waters, where the giant Nomura’s jellyfish has increased significantly, wreaking havoc with fisheries in the Sea of Japan.
Jellyfish populations are normally held in check by fish, mostly because these two groups compete for the same food sources. However, overfishing in many parts of the ocean has led to increasing jellyfish populations. Jellyfish may also be aided by warming ocean temperatures, which favors their development, and by the destruction of habitats of other natural predators such as turtles.
The massive Nomura’s jellyfish is a great threat to Chinese, Japanese and Korean fisheries. These creatures can grow to two meters diameter (the size of large refrigerators) with a weight of 200 kg.
Examples of Giant Jellyfish
Because of their size, they consume massive amounts of zooplankton, depleting this vital part of the food chain for other organisms. The key threat of the Nomura’s derives from the fact that this jellyfish reproduces extremely rapidly. A mature jellyfish has the ability to produce billions of eggs at a time, and they can do this when they are attacked. Once fertilized, these eggs develop into a resting polyp stage that also has the ability to multiply rapidly, effectively carpeting areas of the seafloor. When conditions are suitable, the polyp reproduces asexually, developing into the medusa stage, which grows into the mature jellyfish.
Video: Jellyfish Life Cycle (1:41)
The lifecycle of a jellyfish is similar in many ways to that of coral or a dinoflagellate. The mature jellyfish is called the Medusa, and that's what we recognize as a typical jellyfish that we encounter at the beach. The Medusa floats around in the surface of the ocean and often reproduces extremely readily, releasing millions of eggs on a daily basis, often triggered by light levels. Once fertilized, these eggs form the planula stage. And the planula continues to float in the ocean, until it finds a suitable substrate to colonize, often concrete or rock.
The polyp is the next stage and this is the stage that colonizes the hard rock, or the substrate, and this stage will continue to reproduce, this time asexually, forming new polyps. These polyps then continue to grow and often will bud to form colonies. Once conditions are right, often triggered by water temperature or nutrient levels, the polyp stage will then reproduce again asexually to form the new Medusa stage, which will then grow into the mature jellyfish. Because jellyfish are so productive, both in the Medusa stage and in the polyp stage, they are able to colonize massive areas with individuals. And this is the major threat of jellyfish, especially when overfishing in areas reduces their primary predator.
Once ideal conditions develop, either by increasing nutrients or warming of the surface ocean, Nomura’s jellyfish populations literally explode and render fishing virtually impossible because nets become filled with jellyfish. These jellyfish can also continue to reduce the number of fish in the oceans by feeding on their eggs. Moreover, there is evidence that jellyfish can tolerate conditions, like hypoxia, that fish cannot.
The following video summarizes the impact of giant jellyfish on Japanese fisheries.
Video: Monster Jellyfish (2:35)
[MUSIC PLAYING]
[NON-ENGLISH SPEECH]
[MUSIC PLAYING]
[PRESENTER] This year, we have more jellyfish than ever before. Tens of thousands are just caught in the net per day.
[MUSIC PLAYING]
[NON-ENGLISH SPEECH]
[MUSIC PLAYING]
[PRESENTER] They carry nearly a billion eggs. Reproductive potential of this jellyfish is just enormous.
[MUSIC PLAYING]
[PRESENTER] This is the baby jellyfish.
[MUSIC PLAYING]
Blooms of other jellyfish species are being reported in many other parts of the ocean. In the Gulf of Mexico, for example, the last thirty years populations of two species of jellyfish, the sea nettle, and the moon jellyfish, have exploded especially in dead zones as these are one of the few organisms that can tolerate hypoxia. Jellyfish in the Gulf now swarm over hundreds and perhaps even thousands of square miles each summer.

Here also, invasive species of jellyfish, including the Australian jellyfish, have been reported. Several other factors besides hypoxia have caused the increase in Gulf of Mexico jellyfish. As in the Sea of Japan, overfishing has reduced one of the main jellyfish competitors. In addition, drilling platforms have provided habitats in which jellyfish polyps can multiply. As in the Sea of Japan, jellyfish in the Gulf of Mexico are impacting the fishing industry.
Jellyfish swarms, too, have plagued other regions; they include northern Australia where the highly venomous box jellyfish has expanded its range, the Black Sea, and the Bering Sea off Alaska. Worldwide, jellyfish are one of the few organisms that can thrive in dead zones. With the spread of such dead zones in the oceans as a consequence of marine pollution and climate change, we could be entering the age of the jellyfish.
Module Summary and Final Tasks
Module Summary and Final Tasks
End of Module Recap:
In this module, you should have learned the following concepts:
- factors that cause the pH of the oceans to change and how it impacts saturation of the minerals calcite and aragonite;
- the biology and ecology of reef environments and how they are likely to change in the future with acidification and rising temperature;
- the potential impact of acidification on calcareous plankton groups and implications for the marine food chain;
- the biology of harmful algae including dinoflagellates and diatoms;
- factors that cause blooms of harmful algae, why they are likely to increase in the future, how they can be forecasted, and how they impact human health;
- about Cyanobacteria and the factors that cause them to bloom;
- about large jellyfish and factors that have led to major increases in their populations.
Assignments
You should have read the contents of this module carefully, completed and submitted any labs, the Yellowdig Entry and Reply, and taken the Module Quiz. If you have not done so already, please do so before moving on to the next module. Incomplete assignments will negatively impact your final grade.
Labs
- Lab 7: Reef Ecology