Lesson 6: Types and Applications of Metal Alloys
Overview
In this lesson, we will discuss the wide range of commercial applications of ferrous alloys, which includes steel. However, ferrous alloys do have some limitations including having low electrical conductivity compared to other metals, being heavy, and corroding in typical application environments. In addition to the ferrous alloys in this lesson we will look at a range of other (non-ferrous) metal and alloy systems: copper, aluminum, magnesium, and titanium alloys; the refractory metals; the superalloys; the noble metals; and miscellaneous alloys, including those that have lead, tin, zirconium, and zinc as base metals. Many of these non-ferrous metals and alloys have advantages over the ferrous alloys for particular applications.
Learning Objectives
By the end of this lesson, you should be able to:
- Name four different types of steels and cite compositional differences, distinctive properties, and typical uses for each.
- State different types of nonferrous alloys.
- List current and historical applications of nonferrous alloys.
- Cite distinctive physical and mechanical characteristics of nonferrous alloys.
Lesson Roadmap
Lesson 6 will take us one week to complete. Please refer to Canvas for specific due dates.
| To Read | Read pp 136-179 (Ch. 7 & 8) in Introduction to Materials ebook |
|---|---|
| To Watch | The Secrets of the Viking Sword |
| To Do | Lesson 6 Quiz |
Questions?
If you have general questions about the course content or structure, please post them to the General Questions and Discussion forum in Canvas. If your question is of a more personal nature, feel free to send a message to all faculty and TAs through Canvas email. We will check daily to respond.
Types of Metal Alloys
In this lesson, we are going to take a closer look at metal alloys. First, we will define what an alloy is and how dislocations strengthen alloys. The e-textbook breaks metal alloys into two classes of metal alloys: ferrous and nonferrous alloys. Ferrous is simply the Latin name for iron, so ferrous alloys are simply iron alloys (which means that it is mostly iron mixed with lesser amounts of other metals or nonmetals) and nonferrous alloys are non-iron alloys. In the reading for this lesson, you will see the composition, properties, and applications of a wide variety of metal alloys. The video for this lesson highlights the properties and applications of some of the metal alloys and puts the materials development of the highlighted metal alloys in historical context.
What Are Metal Alloys?
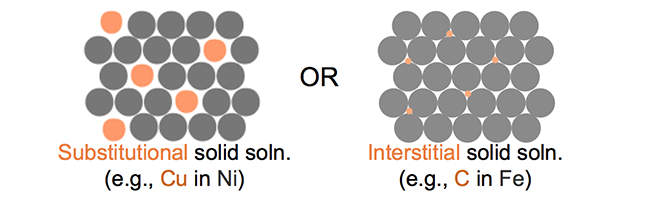
An alloy is a mixture of a metal with another element, either metal or nonmetal. If we start with a base metal and we add impurity atoms there are two possible outcomes if the two mix. The two different cases are highlighted in the figure below. In the substitutional solid case, the impurity atoms replace the host atoms in the lattice. In the interstitial situation, impurity atoms squeeze between the host atoms.
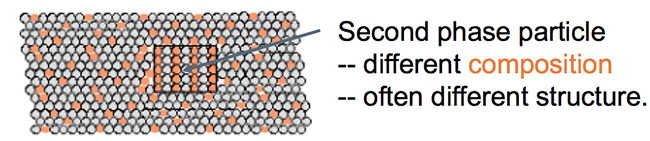
In addition to mixing, it is possible for regions of a new phase to form. An illustration of the formation of a second phase in a solid solution is shown below. The second phase can have a different composition and often a different structure.
To Watch
Now watch the following video (4:44) on alloys and how dislocations harden alloys:
In this video we see how different metals bond together to form alloys which still retain the metallic properties of the starting metals but are usually stronger. Metal atoms are typified by having only a few electrons in their outer shells. This means that even when they bond there's always room in this valence shell for more electrons. Each metal atom can bond with up to 12 others in the close-packed lattice. Look at the red atom. It is surrounded by six in its plane and three on top and three underneath.
A less compact crystal structures are possible too. For example, this arrangement where each atom is bonded to eight others. Because there are still not enough electrons to complete the outer shell any of the atoms the electrons can move easily from one atom to another making metals good conductors of both electricity and heat. And because the electrons are not localized in fixed bonds, the atoms can slide past each other making them ductile allowing the metal to change shape. It also means that when you try to react metals together the atoms normally just mix into the lattice forming metallic bonds with each other and with no fixed proportions and randomly distributed. These structures are called alloys. Contrast this with compounds between metals and nonmetals or between nonmetallic elements where the proportions of each element is fixed.
The oldest example of an alloy perhaps is the way bronze took over from copper in the early human communities of Europe around 6,000 years ago. During the late Stone Age, axes began to be made of pure copper but they were fairly soft. When small amounts of tin were added to make bronze you got an ax which was twice as hard and worked well. The Bronze Age had arrived. The atoms in a metal lattice are held by non-directional bonds a sort of sea of loose electrons as we said allowing the atoms to slide past each other still touching making metals relatively easy to melt and bend but hard to vaporize. When metals change shape atoms actually slip over each other like this. However, this process does not happen all at once but bit by bit rather like trying to move a carpet by putting a rock in it.
Here is the way it happens in the metal. You see the slip moving easily one atom at a time where there's a dislocation in the lattice. It is this easy movement of atoms in the crystal lattice that makes most pure metal soft. Now put a smaller or bigger atom into the lattice and this easy movement of the dislocation is blocked. See the way the bigger atom stabilizes the dislocation which gets no further unless you put greater force meaning that it's harder to bend the alloy.
To finish let's look at some well-known alloys. Bronze, three quaters copper, quarter tin, for sculptures, boat hardware, screws, and grille work. Brass 70 percent copper, 30 percent zinc. Musical instruments, coins, door knockers. Carbon steel 99 percent iron and up to one percent carbon. The building construction, tools, car bodies, machinery rails, etc. stainless steel iron with about 18 percent chromium and eight percent nickel. Used for tableware, cookware, surgical tools, and so on. Aluminium alloys for planes contain a few percent of copper or other metals. Amalgam is mercury with silver and other metals. Once used for dental fillings. Solder lead and tin for joining electrical wires and components. Melts very easily. Gold is usually an alloy containing another metal such as silver for increasing hardness. The number of carats, k, defines how many mass parts of pure gold are found in 24 parts of the alloy.
After watching the video, please proceed to the next section on the development of iron smelting.
Why Did it Take So Long?
Why did it take so long (~2,000 years) for humankind to apply the concepts of smelting copper and bronze to the development of iron? And then another 3000 years to develop steel?
The major issue with the smelting of iron is that with the technology used for smelting of copper and bronze the temperature that is obtainable results in solid iron. So rather than having molten iron, the smelting of iron results in a sponge-like solid mass of impure iron.
As we will see later in the video, impurities could be pounded out of iron by hitting it. So, until the Industrial Revolution, iron could only be produced as a wrought alloy. A wrought alloy is amenable to being mechanically deformed, i.e., pounding it into a desirable shape. Since iron could not be melted it could not be cast in the molds. There were also limits to controlling impurities.
In England in 1709, Abraham Darby started to use coke instead of charcoal as his fuel source to smelt iron ore. Coke, a form of coal, allowed him to build larger and more efficient furnaces than charcoal could support. These furnaces allowed Darby to reach higher temperatures. The temperatures reached were still not high enough to melt pure iron. However, iron that has around 4.3 weight percentage of carbon has a much lower melting temperature than pure iron. Although not pure iron, the iron that he could cast (since it was molten) allowed him to manufacture cast iron pots that could compete successfully with brass.
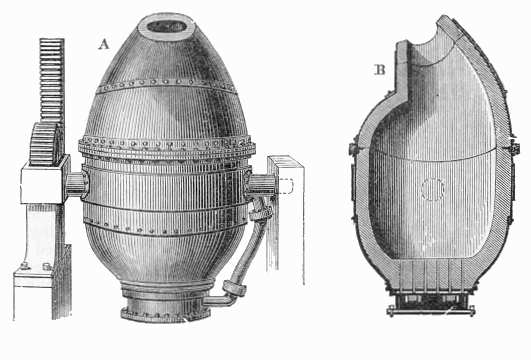
In the 1850s, Henry Bessemer proposed an incredibly bold idea. Bessemer began using very large blast furnaces (shown below), which could produce 3 to 4 tons of molten iron in a single run. Oxygen was blasted through the furnace, which resulted in higher temperatures and the oxygen combining with carbon to form CO2 gas, which bubbled out of the iron. Initially, Bessemer’s process was not reliable. There were issues with phosphorus and sulfur contamination as well as difficulty producing iron with desired target carbon content. This latter issue was resolved by removing all carbon during the process and adding in desired amounts of carbon after purification of the iron.
Now, proceed to the reading and video assignment for this lesson. We will then explore in more detail aluminum alloy and, one of my favorite alloys, metallic glass.
Reading Assignment
Things to consider...
When you read this chapter, use the following questions to guide your reading. Remember to keep the learning objectives listed on the overview page in mind as you learn from this text.
- Often a materials problem is really one of selecting the material that has the right combination of characteristics for a specific application. Do all the ferrous alloys have the same materials properties? What are some of the differences?
- What are the different types of nonferrous alloys?
- How have nonferrous alloys been used in the past?
- How are the nonferrous alloys being used currently?
- What are the distinctive physical and mechanical characteristics of the different nonferrous alloys?
- What are the five types of cast iron?
- How do the microstructure and mechanical characteristics of the five types of cast iron compare?
Reading Assignment
Read pp 136-179 (Ch. 7 & 8) in Introduction to Materials ebook
Video Assignment: Secrets of the Viking Sword
Now that you have read the text and thought about the questions I posed, take some time to watch this 53-minute NOVA video about using cutting edge science, old-fashion detective work, and modern craftsmanship to reconstruct a legendary Ulfberht Viking sword. As you watch this video see if you can apply what you know about carbon content in ferrous alloys to the properties of the sword being manufactured in this video.
Video Assignment
Go to Lesson 6 in Canvas and watch the Secrets of the Viking Sword video. You will be quizzed on the content of this video.
Aluminum Alloy
Aluminum and its alloys were introduced in your e-textbook. The history of the development and applications of aluminum and its alloys were covered in the video for this lesson. Now I am going to expand on this material and highlight the role of aluminum in airplane development.
Aluminum is the third most abundant element in the Earth's crust, after oxygen and silicon, and is the Earth's most abundant metal. It is about 8% of the crust by mass, but it is rarely found as a native metal as it is very chemically active. Its oxide forms more readily than the oxide of iron and, unlike the oxide of iron, once formed it blocks oxygen and water from penetrating the aluminum oxide. This results in aluminum being very corrosion-resistant. Iron, on the other hand, forms rust which does not block oxygen and water, so iron pieces will rust to completion if left long enough in a wet atmospheric environment. Aluminum has a low density, which makes it a candidate for lightweight applications.
Aluminum Electrolysis
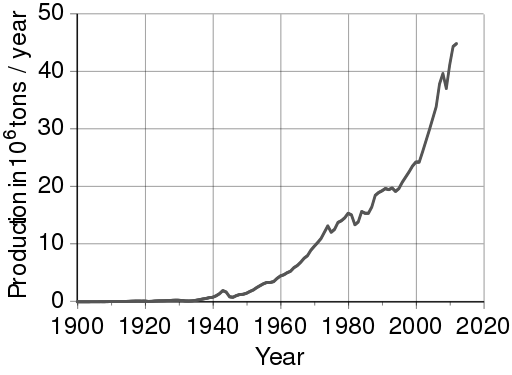
Although aluminum is abundant in nature, it occurs chemically bound to other elements, and there is no known way to smelt aluminum using traditional smelting methods. Because of this limitation, before the 19th century, pure aluminum was rarer than gold. In the 19th century, people learned how to use electrolysis to extract aluminum from aluminum oxide, AlO2. As you can see from the figure below aluminum production has continued to increase ever since.
Typically, aluminum oxide is extracted from the mineral bauxite, and then aluminum is further processed from the aluminum oxide. Visit this website to access the list of where aluminum oxide is produced [5]. Although aluminum oxide is used as an abrasive material most of the aluminum oxide is used for the production of aluminum. For more on the electrolysis process for the extraction of aluminum from aluminum oxide, please watch the fuseschool.org video linked below.
Watch Now
Please watch the following short video (3:13), How to Extract Aluminum Using Electrolysis, on the extraction of aluminum using electrolysis before proceeding to the next section on building lighter aircraft.
Aluminum is the most abundant metal on Earth, however, it is expensive because a lot of electricity is used to extract it. Aluminum conducts heat and electricity well, has a low density, and does not corrode. This makes it very useful for airplanes, drinks cans, electricity cables, and cooking pans. The aluminum ore is called bauxite. Bauxite is purified to yield aluminum oxide which is a white powder. Aluminum is then extracted from the aluminum oxide. The aluminum is extracted by electrolysis. In this video we are going to look at how aluminum is extracted using electrolysis. You should already know how electrolysis works. If you have forgotten, watch our video Electrolysis - How Does it Work, to refresh your memory.
In electrolysis, ions need to pass through the electrolyte and so the aluminum oxide must be made molten so that this can happen. Aluminum oxide has a very high melting point over 2000 degrees Celsius, so instead of trying to melt it the aluminum oxide is dissolved in molten cryolite. Cryolite is an aluminum compound with a much lower melting point than aluminum oxide, and so using this reduces some of the costs in extracting aluminum. The steel case is coated with graphite providing a negative cathode. The positive anodes are immersed in the molten cryolite and are also made of graphite. Remember that graphite is a form of carbon. When the battery is turned on and electricity flows the aluminum from the aluminum oxide in the cryolite forms at the negative cathode and sinks to the bottom of the tank. Here it can then be tapped off as a pure liquid metal. The aluminum sinks because it is more dense than the aluminum prior light solution. The oxygen from the aluminum oxide in the cryolite forms at the positive anodes. The oxygen reacts with the carbon of the graphite forming carbon dioxide. The positive anode therefore burns away and needs replacing regularly. This is another reason for the extraction of the aluminum being so expensive. The overall reaction is aluminum oxide to plus oxygen.
Let's have a quick look at the reactions at the electrodes. At the negative cathode where the aluminum forms the aluminum ions from the molten aluminum oxide cryolite solution are reduced. This means they gain electrons. At the positive anode, where the oxygen reacts with carbon to make carbon dioxide, the oxygen ions are oxidized. This means they lose electrons.
So, from this video you should know that extract aluminum electrolysis is used. Aluminum oxide needs to be molten for the ions to move through it, and so is dissolved in cryolite to lower the melting point. The anode is gradually ward away because the oxygen from the solution reacts with the carbon of the graphite anode producing carbon dioxide, and so the anode wears away and needs to be replaced regularly. Aluminum extraction is very expensive because a lot of electricity is needed.
Building a Lighter Aircraft
The lighter that we can build safe aircraft the better. Reducing the operating empty weight of commercial aircraft can allow for an increase in the passengers, baggage, and cargo that the plane can safely transport. Early aircraft were made of wood and fabric. An example of an early aircraft is shown in the figure below. This provided a good combination of lightness and strength but required reinforcing struts, which added weight and drag and resulted in multiple wing designs.
Improved airplane engine designs resulted in more powerful engines and higher airspeeds. As speed increases, drag increases nonlinearly. Single wing (less drag) airplane designs were required to take advantage of the improvements in speed.
All Metal Monoplanes
The first all metal monoplanes were developed during World War I. These were faster, but it was quickly realized that they did not climb well. Although more powerful than the initial airplane engines, the engines of World War I did not have enough power to lift the all-metal monoplanes quickly enough.
Wood, iron, and aluminum are possible materials for making aircraft wings. How do the densities of these materials compare? The density of water is 1 g/cm3 by definition at standard temperature and pressure. Wood floats in water so its density must be less than 1 g/cm3. Its density ranges from 0.45 to 0.85 g/cm3. Iron and aluminum do not float, so their densities must be greater than 1 g/cm3. Iron's density is equal to 7.9 g/cm3 and aluminum's density is equal to 2.7 g/cm3. So, in theory, it should be possible to reduce the weight of an airplane by utilizing aluminum instead of iron. Aluminum is about 1/3 the density of iron. But there is a problem: aluminum is not strong and alloying does not strengthen the material the way it does in bronze and iron. Aluminum needed to be strengthened, but how?
Hardened Aluminum Alloys
In 1901, German metallurgist Alfred Wilm was working to harden aluminum-copper alloys. The work was not going well so in frustration he went on holiday (vacation). Upon his return, he found a harder material and after many years of work developed a commercially viable age-hardened aluminum alloy. Age-hardened aluminum, which is about three times lighter than iron, replaced iron in aircraft manufacturing. A photo of an early aluminum-bodied aircraft is shown below.
Age hardened aluminum is not as strong as iron so additional aluminum is needed which does offset some of the weight savings. The video in our later Synthesis, Fabrication, and Processing of Materials lesson has more on the use of aluminum in the construction of a modern commercial jet airliner. In the photo below is a Boeing 787 Dreamliner which utilizes a composite airframe, not aluminum. Boeing claims that this airliner is 20% more fuel-efficient than previous generations of airliners.

Now please proceed to the next section on one of my favorite alloys, a future star, a non-crystalline metal.
Metallic Glass
Most metals are crystalline. In fact, it is typically very difficult to make a noncrystalline metal. The following short video highlights metals that are noncrystalline, i.e., amorphous. These materials are sometimes referred to as metallic glasses.
To Watch
After viewing this video please proceed to the summary page of this lesson.
Summary and Final Tasks
Summary
The extremely versatile range of different metals and alloys have produced an incredible range of application for these metals and alloys. In this lesson, we have explored how ferrous metals and the many different non-ferrous metals and alloy systems are historically and currently used. Understanding the strengths and weaknesses of these materials can allow one to properly select the right material for the desired application and the environment in which the application exists. In the next lesson, we will be looking at ceramics and their role as one of the primary materials.
Reminder - Complete all of the Lesson 6 tasks!
You have reached the end of Lesson 6! Double-check the to-do list on the Overview page to make sure you have completed all of the activities listed there before you begin Lesson 7.