Lab Activity: Mentos and Magma
In Part 1 of this lab activity, you will conduct the famous "Mentos and Diet Coke eruption", under more-or-less controlled experimental conditions. In Part 2, you will make some degassing calculations of your own. First, you will calculate the amount of CO2 released from a 2-liter bottle of Diet Coke. Then you will apply the same principles to calculate the amount of SO2 released during the 1783-84 eruption of Lakagígar.
Part 1: Mentos and Diet Coke Eruption
Materials
- One pack of mint Mentos
- One two-liter bottle of Diet Coke
- Paper tube made by rolling a half-sheet of paper loosely around the Mentos roll and securing with tape – or you can just use the Mentos label
- Index card or other piece of thin cardboard
- Measuring tape
- Towels, water (to clean up)
Before You Begin
This is going to be messy. I strongly recommend that you perform this experiment outside. If you can’t go outside, you can do it in the bathtub or shower using only one or two Mentos. You can also use soda water in place of Diet Coke. The fountain won’t be as high, but it will be easier to clean up! Make sure you have plenty of water on hand to rinse down the area after your experiment.
Method
- Slowly open the 2 liter bottle of Diet Coke.
- Load the paper tube with four Mentos.
- Place the index card over one end of the tube and invert it.
- Set the card and the tube with the Mentos inside on top of the open mouth of the bottle, making sure the opening of the tube lines up with the mouth of the bottle.
- Quickly remove the index card, allowing the Mentos to drop into the bottle
- Step back quickly!
- Record the height of the geyser
- Optional: repeat using a new bottle of Diet Coke and more Mentos – how high can you go?
- Enter your data on the spreadsheet provided. Once we have data for the entire class, we will do some statistical analysis on our results.
Pointers
- Determining the height can be a little tricky in the moment. It helps to have two people – one to load the bottle, and one to observe. Or set up a video camera and record your experiment so you can estimate the height after the fact.
- If you can, the best way to determine fountain height is to make markers at one-foot intervals on a wall, tree, or pole (see green tape markers on garage door in image below). You can use masking tape or chalk to make the marks. This is easier than trying to hold up a measuring tape.
- Or, you can use a person of known height for a reference. Was the geyser waist-high? Chest-high? Above the head?
- It’s best to put the bottle on a flat surface if possible so it doesn’t tip over.
- Make sure the Mentos can fall freely from the tube before you begin. If the tube is wet or crushed, the Mentos can get stuck inside. If this happens, make a new tube.
- Try not to shake the bottle before you open it, or you will lose some CO2 before the experiment begins. Also, it’s difficult to get the Mentos to go down into a bottle that’s already foaming over.

Just for Fun
If you have someone to help you (or a trusty tripod), record a video of your eruption and turn it in! I’ll make a compilation of “greatest hits” for the website.
Video: Diet Coke & Mentos | MythBusters (1:57)
NARRATOR: The Mythbusters are going to have a blast.
ADAM: The most common theory about what's going on in this reaction between the candy and the soda is what's called nucleation. Basically the idea is that the surface of the candy is covered with microscopic pits and lots of more surface area than you can actually see and each little pit, each little corner provides what's called a nucleation site or a place where carbon dioxide bubble can form and escape.
NARRATOR: Look at a Mentos close up and it's like the surface of the Moon and that might be the candy key. Drop one in cola and every tiny crater provides a site where a CO2 molecule can change to gas. Because Mentos are so pitted the theory is that millions of CO2 bubbles are formed in a very short space of time, and because the candy sinks and this rapid release of gas happens at the bottom of the cola you get that famous fountain.
To test this nucleation theory they're going to start with a control. One regular mint Mentos dropped in soda water and sure enough it gets the bubbly party started. Now to do a comparison.
JAMIE: These two candies are made by the same manufacturer and as far as I'm aware even using the same process, but the colored version of this actually has a glazing over it. It's a wax coating or a sealer that inhibits the nucleation process that the other one achieves quite readily.
NARRATOR: The shiniest surface should lower the nucleation sites meaning less of an immediate eruption.
JAMIE: That's not doing anything more than the wall of the plastic bottle itself.
NARRATOR: Sure enough with the smooth mentos there ain't no Wiz with the fizz. The Mythbusters can say they finally cracked the case of the candy and cola cascade.
Video: Science of Mentos - Diet Coke explosions explained (1:26)
The fountain of foam produced when you drop mentos into diet coke has been seen millions of times on YouTube. Until now there's not been a scientific explanation. A study in the US has identified the two main factors involved. They turn out to be one the roughness of the surface of the sweet and two how fast it plummets to the base of the bottle. Students compared the effects of fruit and mint Mentos as well as various other ingredients. Here they are testing sound. previous theories put the reaction down to the acid in the coke or the gum arabic and gelatine in the sweets these explanations have not been properly tested. With no ph change occurring and acidic reaction was ruled out as a cause as was gum arabic following less impressive results when tested independently as can be seen here. Instead the fountain effect was explained by the rough dimply surfaces of the mentos encouraging carbon dioxide bubble growth. These are scanning electron microscope images of the mint Mentos on the left and fruit Mentos on the right. The scale bars represent lengths from 20 to 200 micrometers. Mentos are also fairly dense and sink rapidly quickly creating bubbles seed further bubbles as they rise.
[Music]
Video: Original Mentos Diet Coke Geyser - Cool Science Experiment (2:13)
MAN: This is really good. All right. So here's the setup. Mentos, and the reason we choose Mentos is because they have this chalky like quality to it.
WOMAN: Right.
MAN: What we want is.. we're just gonna put them into the test tube so they go down to the test tube like this. So there's your test tube got it. 13 Mentos is just in case you're counting. All right, so there it is. So now let me open this up here. And yours, you can open yours up as well all right okay. So and we're just using diet just because it's less sticky when it finally there's a disaster.
WOMAN: Oh when there is...
All right, so now here's what's going to happen. We put the card on. We turn the whole thing upside down we're gonna put it over the top. We're gonna position it so it's right over the top. You got it? Right there, okay. All right, now when we pull it out those are going to fall real fast and when they do all of the bubbles that are inside are going to rush to them and we might see a little eruption. Are you ready? Alright so we're going to go three, two, one pull ready three, two, one pull good ...ahh!
Tell me that's not awesome. So sorry look at you you! Wasn't that fun? That was fine that's the best I have ever seen! Is that are.. you good? Alright so. So just ask them is it better for diet or regular go ahead and ask?
WOMAN: Is it better for diet or regular?
MAN: Let's go find out. Come on let's go works in a big way it does not let you you go there I'm going go here 'cause you that's this is, this is the regular. All right.
Okay we're good. Alright so you remember then the carbon dioxide is coming out. Alright hey so this is ... you're okay alright so turn it over and remember they're gonna come rush into it. So let's do it together. Ready three, two, one, pull...
Awesome! Mine didn't even fall in ... get out of the way I'll do this with me so here we go. Three, two, one .. now that is awesome! Is that great? So here, yeah you look great no you look awesome, and guess what I put it all on the website so a teacher can be amazing to learn about states of matter and gases. I'm so sorry, look at you, you're a mess.
The Science
A bottle of soda contains dissolved carbon dioxide (CO2) under pressure. When you remove the lid, the pressure is released, and the CO2 exsolves in the form of tiny bubbles. When exsolution occurs faster than the gas can escape, the soda gets whipped up into a foam that quickly overflows the confined volume of the bottle – if you have ever shaken or dropped a bottle of soda before opening it, you have probably observed this effect yourself. In this experiment, the Mentos encourage the rapid formation of bubbles by providing a nucleation site. In the absence of a nucleation site, the CO2 gas must overcome the surface tension of the liquid before it can form a bubble, which inhibits the process a bit, especially at the beginning. Mint-flavored Mentos have a pitted surface with lots of surface area, which provides plenty of nucleation sites for bubble growth. The more Mentos, the more nucleation – hence, a soda eruption! It is less clear why Diet Coke works better than regular Coke, but based on observation this seems to be the case. Some people have suggested a chemical reaction involving the artificial sweeteners. However, any carbonated beverage will produce a fountain when Mentos are added, some will just be more dramatic than others. Incidentally, fruit-flavored Mentos do not produce an eruption. This is because they have a smooth waxy coating that does not provide nucleation sites for bubble formation.
Lab Worksheet
Download the Excel Spreadsheet [3] to enter your experimental results
Download and complete the Worksheet for Lab 2: Degassing [4]
You will need to submit the results spreadsheet and the complete worksheet to the Module 2 Lab Assignment in Canvas.
Part 2a: Degassing Calculations
The idea here is to determine the mass of CO2 you released into the atmosphere during the first part of your experiment. Watch your unit conversions!
First, a few assumptions:
- We shall assume that the physical properties of Diet Coke are the same as those of water (H2O) at room temperature (20˚C)
- We shall assume that a typical soda contains ~0.5% CO2 by weight prior to being opened (in reality this may vary by quite a bit)
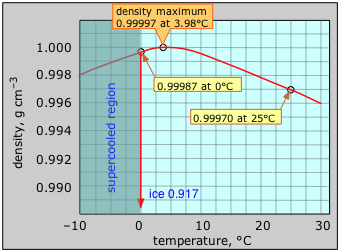
We start by determining the total mass of CO2 present at the beginning of the experiment (prior to opening the bottle). In order to do this, first you will need to determine the mass of Diet Coke. Use the graph below to determine the density of water at 20˚C; we will assume your Diet Coke has the same density. Note that 1 cm3 = 1 mL.
- Approximately how many grams of Diet Coke (water) are in a 2 liter bottle?
- Given that the concentration of CO2 in soda is ~0.5% by weight, how many grams of CO2 are in the bottle?
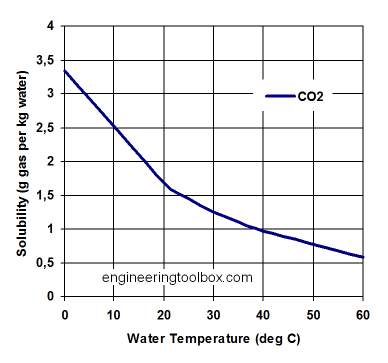
Solubility is the amount of a compound that will remain in solution under a given set of conditions. Use the graph below to estimate the solubility of CO2 in water at 20˚C and atmospheric pressure.
The amount of CO2 released is given by the total amount present prior to opening the bottle minus the amount retained after the degassing experiment.
- How many grams of CO2 will remain dissolved in our 2 liters of Diet Coke after the experiment is done?
- What was the mass of CO2 released?
Part 2b: Degassing of Lakagígar
We can use the same approach to calculate the mass of SO2 released from the lava during the Lakagígar eruption. First, we need to estimate the mass of SO2 dissolved in the magma prior to eruption. But how does one determine the concentration of a volatile component prior to degassing, when all the lava and tephra samples we have are already degassed? The answer lies in tiny bits of glass trapped inside of crystals. We call these bits of glass melt inclusions, because they represent the magma that was present at the time the crystals formed. Once a melt inclusion has been overgrown by a crystal, the volatiles are trapped inside and cannot escape*.
The concentration of sulfur measured in melt inclusions from Lakagígar ranges from ~1200 to 1800 parts per million (ppm). We can use the best estimate of 1675 ppm from Thordarson et al., 1996. In order to convert this concentration into an equivalent mass of S, we need to multiply by the total mass of lava erupted. We can assume a best estimate of 15 km3 of lava erupted.
*In detail this is not entirely true – volatiles can still diffuse out through the solid crystals at high temperatures – but for the purposes of our calculations we can assume that they remain perfectly entrapped.

1. Assuming a basalt density of 2750 kg/m3, what is the total mass of lava erupted in megatons (109 kg)? Watch your units! Not only do you need to convert kilograms to megatons, but you also need to convert cubic kilometers to cubic meters.
Now multiply the mass of lava you just calculated by 1675/106 to get the mass of sulfur in the magma prior to degassing.
2. What is the total mass of sulfur before degassing?
Now, just as with the CO2 in Coke experiment, you will also need to estimate the mass of sulfur after degassing, which is determined by measuring the concentration of sulfur in the degassed tephra and lava. The best estimate given by Thordarson et al. is 205 ppm.
3. Using the same total mass of lava you used above, calculate the mass of sulfur remaining after degassing.
The difference between these two masses is the mass of sulfur released to the atmosphere.
4. What is the total mass of sulfur released to the atmosphere (in megatons)?
One last thing. The sulfur released to the atmosphere is not pure elemental sulfur, it is mostly in the form of SO2 gas. In order to convert the mass of S into the equivalent mass of SO2, you will need to multiply by the mass ratio of SO2 to S. You can use any periodic table (I like WebElements [7]) to calculate the molar mass of SO2. Then simply divide this by the molar mass of S, and you have the mass ratio. Multiply by the total mass of sulfur released, and you’re done!
5. What is the total mass of SO2 released to the atmosphere (in megatons)?
6. Thordarson et al. calculated 122.1 megatons of SO2 released. How close did your calculation come to theirs?