Natural Gas Composition and Specifications
Natural gas as recovered at the wellhead consists of mostly methane (C1), but it contains other hydrocarbons, principally ethane (C2), propane (C3), butanes (C4), and pentanes C5 that constitute the natural gas liquids, as discussed in the previous section. Raw natural gas also contains water vapor, hydrogen sulfide (H2S), carbon dioxide, nitrogen, helium, and other impurities, such as mercury. Table 12.3 gives some examples of the composition of natural gas produced in three different locations, to give an example that methane content of natural gas can be as low as 65%. One can also note in Figure 12.2 that some natural gas streams may contain high concentrations of H2S and N2. Some natural gas streams could be a commercial source for helium [4]. One of the important objectives of natural gas processing is to remove the corrosive and toxic gas H2S and convert it to elemental sulfur, as will be discussed later. Important impurities, including those shown in Table 12.3, that need to be removed from natural gas are listed below [5].
| Canada | Kansas | Texas | |
|---|---|---|---|
| C1 | 77.1 | 73.0 | 65.8 |
| C2 | 6.6 | 6.3 | 3.8 |
| C3 | 3.1 | 3.7 | 1.7 |
| C4s | 2.0 | 1.4 | 0.8 |
| C5s+ | 3.0 | 0.6 | 0.5 |
| H2S | 3.3 | 0.0 | 0.0 |
| CO2 | 1.7 | 0.0 | 0.0 |
| N2 | 3.2 | 14.7 | 25.6 |
| He | 0.0 | 0.5 | 1.8 |
Important impurities found in natural gas [5].
- Water: Most gas produced contains water, which must be removed. Concentrations range from trace amounts to saturation.
- Sulfur species: If the hydrogen sulfide (H2S) concentration is greater than 2 to 3%, carbonyl sulfide (COS), carbon disulfide (CS2), elemental sulfur, and mercaptans may be present.
- Mercury: Trace quantities of mercury may be present in some gases; levels reported vary from 0.01 to 180 μg/Nm3. Typically, the mercury level in pipeline gas should be reduced to 0.01 μg/Nm3.
- Diluents: Although the gases shown in Figure 12.2 are typical, some gases have extreme amounts of undesirable components. For example, some wells in Colorado contain as much as 92% carbon dioxide. High hydrogen sulfide contents (e.g., in Alberta, Canada), and nitrogen contents (e.g., in Texas) have also been observed.
- Oxygen: Some gas-gathering systems in the United States operate below atmospheric pressure. As a result of leaking pipelines, open valves, and other system compromises, oxygen is an important impurity to monitor. A significant amount of corrosion in gas processing is related to oxygen contamination.
Considering that the principal transportation of natural gas over land is by pipeline, natural gas specifications for pipeline transmission have been developed. Table 12.4 gives the natural gas specifications that need to be satisfied for pipeline transportation. Note that in addition to the specified impurity levels for the contaminants, the specifications include the heating value of natural gas (950 -1150 Btu/scf) which depends on the composition, particularly the concentration of inert gases (e.g., N2 and CO2) and other diluents.
Table 12.4. Specifications for pipeline quality natural gas [6].
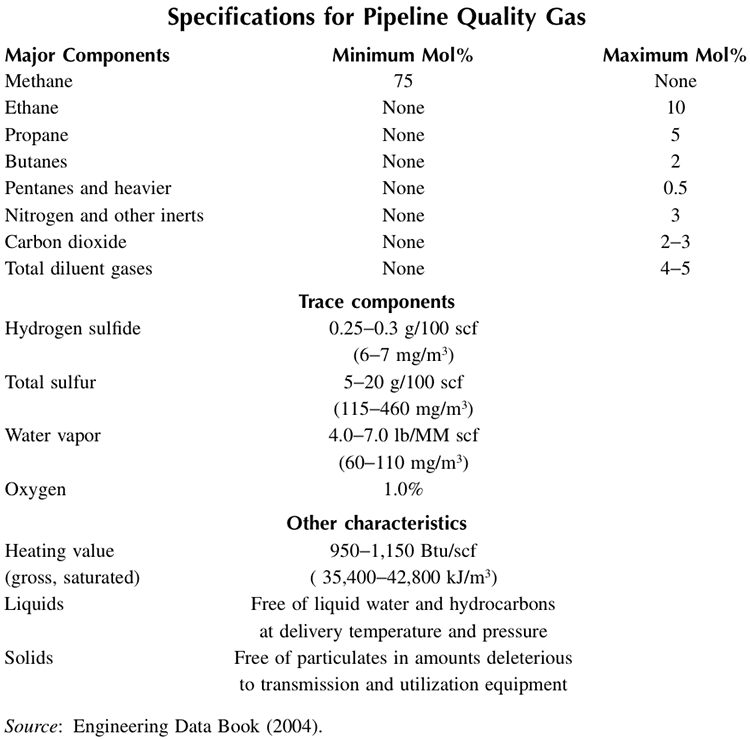
| Major Components | Minimum Mol% | Maximum Mol% |
|---|---|---|
| Methane | 75 | None |
| Ethane | None | 10 |
| Propane | None | 5 |
| Butanes | None | 2 |
| Pentanes and heavier | None | 0.5 |
| Nitrogen and other inerts | None | 3 |
| Carbon dioxide | None | 2-3 |
| Total diluent gases | None | 4-5 |
Trace Components:
- Hydrogen sulfide: 0.25-0.3 g/100 scf (6-7 mg/m3)
- Total sulfur: 5-20 g/100 scf (115-460 mg/m3)
- Water Vapor: 4.0-7.0 lb/MM scf (60-110 mg/m3)
- Oxygen: 1.0%
Other Characteristics:
- Heating calculated (gross, saturated): 950-1,150 Btu/scf (35,400-42,800 kJ/m3)
- Liquids: Free of liquid water and hydrocarbons at delivery temperature and pressure.
- Solids: Free of particulates in amounts deleterious to transmission and utilization equipment.
[4.] A. J. Kidnay and W. R. Parrish, Fundamentals of Natural Gas Processing, CRC Press, Boca Raton, FL, 2006, p.9.
[5.] A. J. Kidnay and W. R. Parrish, Fundamentals of Natural Gas Processing, CRC Press, Boca Raton, FL, 2006, p.10.
[6.] A. J. Kidnay and W. R. Parrish, Fundamentals of Natural Gas Processing, CRC Press, Boca Raton, FL, 2006, p.16.