5.2b Cellulose
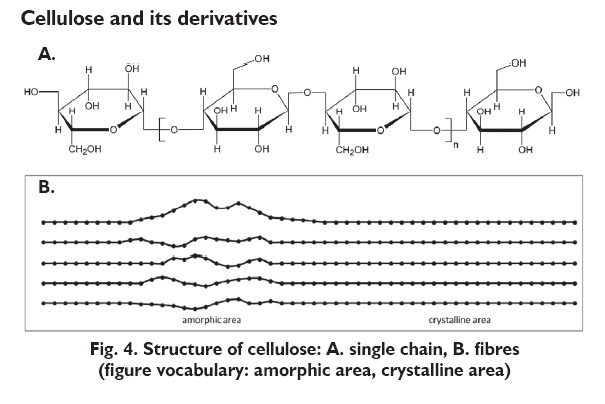
Cellulose is the most abundant polysaccharide, and it is also the most abundant biomass on earth. The linkages are slightly different from starch, called β-1,4-glycosidic linkages, as the bond is in a slightly different configuration or shape. As shown in the figures below, this bond causes the strands of cellulose to be straighter (not helical). The hydrogen on one polymer strand can interact with the OH on another strand; this interaction is known as a hydrogen bond (H-bond), although it isn't an actual bond, just a strong interaction. This is what contributes to the crystallinity of the molecule. [Definition: the H-bond is not a bond like the C-H or C-O bonds are, i.e., they are not covalent bonds. However, there can be a strong interaction between hydrogen and oxygen, nitrogen, or other electronegative atoms. It is one of the reasons that water has a higher boiling point than expected.] The strands of cellulose form long fibers that are part of the plant structure. The average molecular weight is between 50,000 and 500,000, and the average number of glucose units is 300-2500.
The table below shows a comparison of the two types of starch and cellulose. Cellulose forms elongated fibers that stretch out; it doesn’t curl the way the amylose does (remember the helical structure) and doesn’t branch and curl the way the amylopectin does. Because of its chemical structure, it forms a large network where H-bonds stabilize the strand itself, and also the cluster of strands that make up the fibers. The H-bond gives cellulose fibers several important structural features. It is incredibly tough. It is water-impermeable because of the H-bonds and thus excludes water.
| Type of polysaccharide | Starch (Amylopectin) | Starch (Amylose) | Cellulose |
|---|---|---|---|
| Types of linkages |
|
α-1,4-glucosidic | β-1,4-glucosidic |
| Function | Stores energy | Stores energy | Supports and strengthens |
| Molecular weight (amu) | 300,000 | 10,000-50,000 | 50,000-500,000 |
| Size of glucose units | 1800 glucose units | 6-300 glucose units | 300-2500 glucose units |