8.4 Biodiesel Properties and Specifications
To ensure quality biodiesel, there are standards for testing the fuel properly to see that it meets specifications for use. ASTM (an international standards and testing group) has a method to legally define biodiesel for use in diesel engines, labeled ASTM D6751. The table below shows the test methods necessary for all the expected standards for biodiesel.
| Property | ASTM Method | Limits | Units |
|---|---|---|---|
| Ca & Mg, combined | EN 14538 | 5 max | ppm (ug/g) |
| Flash point | D 93 | 93 min | °C |
| Alcohol Control | - | - | - |
| 1. Methanol content | EN14110 | 0.2 max | % mass |
| 2. Flash point | D 93 | 130 min | °C |
| Water & Sediment | D2709 | 0.05 max | % vol |
| Kinematic Viscosity, 40°C | D445 | 1.9-6.0 | mm2/sec |
| Sulfated Ash | D874 | 0.02 max | % mass |
| Sulfur | - | - | - |
| S 15 Grade | D5453 | 0.0015 max (15) |
% mass (ppm) |
| S 500 Grade | D5453 | 0.05 max (500) |
% mass (ppm) |
| Copper Strip Corrosion | D130 | No. 3 max | - |
| Cetane | D613 | 47 min | - |
| Cloud Point | D2500 | report | °C |
| Carbon Residue (100% sample) | D4530 | 0.05 max | % mass |
| Acid Number | D664 | 0.50 max | mg KOH/g |
| Free Glycerin | D6584 | 0.020 max | % mass |
| Total Glycerin | D6584 | 0.240 max | % mass |
| Phosphorus Content | D4951 | 0.001 max | % mass |
| Distillation, T90 AET | D1160 | 360 max | °C |
| Sodium/Potassium, combined | EN 14538 | 5 max | ppm |
| Oxidation Stability | EN 14112 | 3 min | Hours |
| Cold Soak Filtration | Annex to D6751 | 360 max | seconds |
| For use in temperatures below -12 °C | Annex to D6751 | 200 max | seconds |
There are advantages and disadvantages to using biodiesel compared to ultra-low sulfur diesel. It has a higher lubricity, low sulfur content, and low CO and hydrocarbon emissions. This makes it good to blend with diesel from petroleum to be able to achieve the required specifications for ultra-low sulfur diesel because ultra-low sulfur diesel has poor lubricity. But as discussed previously, biodiesel has poor cold weather properties. It really depends on the location; for instance, if using biodiesel in the upper Midwest, there could be problems in the winter.
As with all materials, the production and quality of biodiesel is important. Most importantly, the transesterification reaction should reach completion for the highest production and quality. Due to the nature of the transesterification of triglycerides, a small amount of tri-, di-, and mono-glycerides remain. The figure below shows the changes in these compounds as the glycerides react to form biodiesel. Some terminology to be aware of: 1) bound glycerol is glycerol that has not been completely separated from the glyceride and is the sum of tri-, di-, and mono-glycerides and 2) total glycerol combines the bound glycerol with the free glycerol.
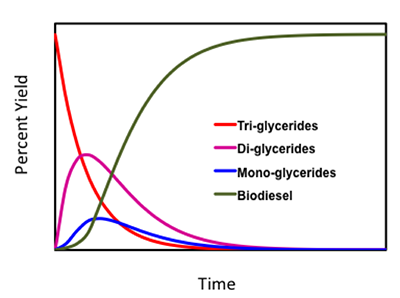
Glycerol content in biodiesel must be as low as possible, as ASTM standards state. The biodiesel will not technically be “biodiesel” unless ASTM standards are met, which means it is below the total glycerol specifications. High glycerol content can cause issues with high viscosity and may contribute to deposit formation and filter plugging. Crude glycerol is often a dark brown color and must be refined and purified before use elsewhere. In biodiesel preparation, brown layers will form, and, possibly, white flakes or sediments, formed from saturated mono-glycerides, will fall to the bottom of the tank the biodiesel is being stored in.
Biodiesel is also a great solvent, better than petroleum-based diesel. It can loosen carbon deposits and varnishes that were deposited by petro-diesel and can cause fuel-filter plugging when switching over to biodiesel. Filters should be changed after the first 1,000 miles with biodiesel.

Another issue is the cold weather properties of biodiesel. These properties include cloud point, pour point, and cold soak filtration. Biodiesel can form cloud points at a much higher temperature than petro-diesel, close to the freezing point. The cloud point is the temperature at which crystals begin to form; it can cause the biodiesel to gel and flow slower than it should. Once the pour point is reached (basically completely frozen), the fuel cannot move. It depends on the normal temperature of the climate as to whether the fuel can be used or blended with petrodiesel. What can complicate it more is the saturated or unsaturated fatty acid content. High saturated fatty acid content can lead to higher fuel stability but higher pour points. High unsaturated acid content can lead to lower pour points but less stability for storing. The figure below shows a pour point comparison of biodiesels made from various oils (including fatty acid content) compared to petrodiesel. Petro-diesel pour points are significantly lower than biodiesels.
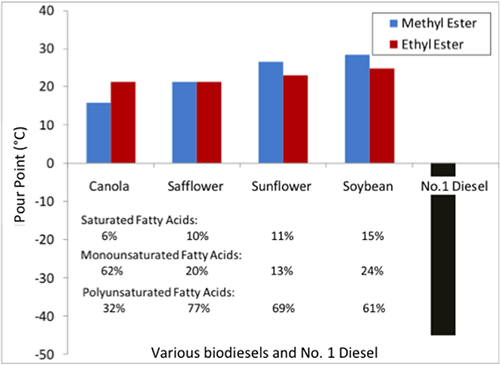
| Biodiesel | Pour Point (Methyl Ester) ºC | Pour Point (Ethyl Ester) ºC | % Saturated Fatty Acids | % Monounsaturated Fatty Acids | % Polyunsaturated Fatty Acids |
|---|---|---|---|---|---|
| Canola | 15 | 22 | 6% | 62% | 32% |
| Safflower | 22 | 22 | 10% | 20% | 77% |
| Sunflower | 24 | 28 | 11% | 13% | 69% |
| Soybean | 25 | 30 | 15% | 24% | 61% |
| asdf | -45 | - | - | - | - |
Cetane number is also an important property for diesel fuels. Cetane number measures the point that the fuel ignites under compression, and this is what we want for a diesel engine. The higher the cetane number, the greater the ease of ignition. Most petro-diesel fuels have a cetane number of 40-50 and meet the ASTM specification for ASTM D975. In general, most biodiesels have higher cetane numbers, 46-60 (some as high as 100), and meet the specifications for ASTM D6751. Because of the higher cetane numbers of biodiesel, the engine running on biodiesel will have an easier time starting and have low idle noise. The table below shows the heat of combustions for various fuels along with their cetane number.
| Fuel | Heat of Combustion (Mj/kg) | Cetane No. |
|---|---|---|
| Methyl Ester (Soybean) | 39.8 | 46.2 |
| Ethyl Ester (Soybean) | 40.0 | 48.2 |
| Butyl Ester (Soybean) | 40.7 | 51.7 |
| Methyl Ester (Sunflower) | 39.8 | 47.0 |
| Methyl Ester (Peanut) | - | 54.0 |
| Methyl Ester (Rapeseed) | 40.1 | - |
| No. 2 Diesel | 45.3 | 47.0 |
If full-strength biodiesel is used (i.e., B100), most engine warranties will not be covered. It will also require replacing rubber seals in older engines. Blends include B2, B10, and B20 (2%, 10%, and 20% biodiesel, respectively). Adding biodiesel as a blend with ultra-low sulfur should improve lubricity for ultra-low sulfur diesel fuel, which will improve engine wear. Emissions of hydrocarbons, CO, NOx, and particulate matter are similar to petrodiesel fuels, although can be reduced in some cases.
Biodiesel is stored very similarly to petrodiesel. It is stored in clean, dark, and dry environments. It can be stored in aluminum, steel, fluorinated polyethylene, fluorinated polypropylene, and Teflon types of containers. It is best to avoid copper, brass, lead, tin, and zinc containers.
In another lesson, we will discuss the economics behind using biodiesel.