Why did it take so long (~2,000 years) for humankind to apply the concepts of smelting copper and bronze to the development of iron? And then another 3000 years to develop steel?
The major issue with the smelting of iron is that with the technology used for smelting of copper and bronze the temperature that is obtainable results in solid iron. So rather than having molten iron, the smelting of iron results in a sponge-like solid mass of impure iron.
As we will see later in the video, impurities could be pounded out of iron by hitting it. So, until the Industrial Revolution, iron could only be produced as a wrought alloy. A wrought alloy is amenable to being mechanically deformed, i.e., pounding it into a desirable shape. Since iron could not be melted it could not be cast in the molds. There were also limits to controlling impurities.
In England in 1709, Abraham Darby started to use coke instead of charcoal as his fuel source to smelt iron ore. Coke, a form of coal, allowed him to build larger and more efficient furnaces than charcoal could support. These furnaces allowed Darby to reach higher temperatures. The temperatures reached were still not high enough to melt pure iron. However, iron that has around 4.3 weight percentage of carbon has a much lower melting temperature than pure iron. Although not pure iron, the iron that he could cast (since it was molten) allowed him to manufacture cast iron pots that could compete successfully with brass.
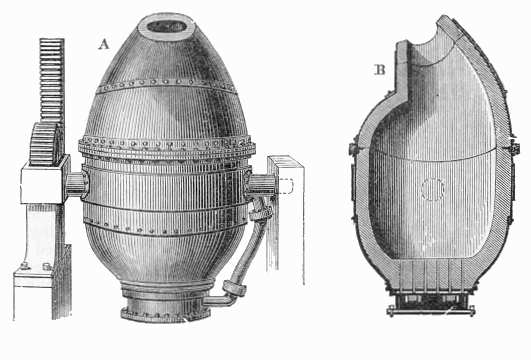
In the 1850s, Henry Bessemer proposed an incredibly bold idea. Bessemer began using very large blast furnaces (shown below), which could produce 3 to 4 tons of molten iron in a single run. Oxygen was blasted through the furnace, which resulted in higher temperatures and the oxygen combining with carbon to form CO2 gas, which bubbled out of the iron. Initially, Bessemer’s process was not reliable. There were issues with phosphorus and sulfur contamination as well as difficulty producing iron with desired target carbon content. This latter issue was resolved by removing all carbon during the process and adding in desired amounts of carbon after purification of the iron.
Now, proceed to the reading and video assignment for this lesson. We will then explore in more detail aluminum alloy and, one of my favorite alloys, metallic glass.