Dissolved Nutrients
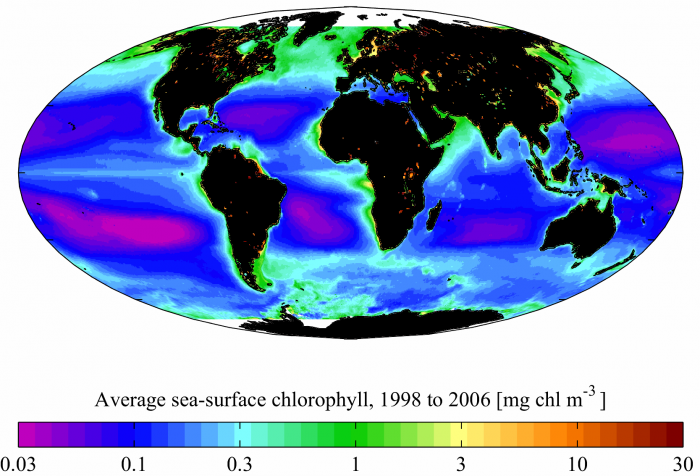
Probably the most important property of seawater in terms of its effect on life in the oceans is the concentration of dissolved nutrients. The most critical of these nutrients are nitrogen and phosphorus because they play a major role in stimulating primary production by plankton in the oceans. These elements are known as limiting because plants cannot grow without them. However, there are a number of other nutrients that also play a role, including silicon, iron, and zinc. Nutrients in the ocean are cycled by a process known as biological pumping, whereby plankton extract the nutrients out of the surface water and combine them in their organic matrix. Then, when the plants die, sink and decay, the nutrients are returned to their dissolved state at deeper levels of the ocean. The abundance of nutrients determines how fertile the oceans are. A measure of this fertility is the primary production, which is the rate of fixation of carbon per unit of water per unit time. Primary production is often mapped by satellites using the distribution of chlorophyll, which is a pigment produced by plants that absorbs energy during photosynthesis. The distribution of chlorophyll is shown in the figure above. You can see the highest abundance close to the coastlines, where nutrients from the land are fed in by rivers. The other location where chlorophyll levels are high is in upwelling zones, where nutrients are brought to the surface ocean from depth by the upwelling process.

The image is a cross-sectional diagram illustrating ocean water movement near Antarctica, focusing on the interaction between different water masses and the ocean floor. It depicts a layered view of the ocean, with the ocean surface at the top and the ocean floor at the bottom. The diagram shows the North Atlantic Deep Water flowing from the left, Antarctic Circumpolar Deep Water in the middle, and various water movements such as upwelling and downwelling. The Antarctic continent is on the right, with a mid-ocean ridge on the ocean floor. Labels indicate key features like the volume front and differences in water masses.
- Top Section
- Feature: Ocean Surface
- Color: Light blue with white patches indicating ice or foam
- Position: Top of the diagram
- Bottom Section
- Feature: Ocean Floor
- Color: Brown
- Position: Bottom of the diagram
- Specific Feature: Mid-ocean Ridge
- Label: Mid-ocean Ridge
- Position: Central area of the ocean floor
- Color: Dark brown
- Water Masses
- North Atlantic Deep Water
- Label: North Atlantic Deep Water
- Position: Left side, flowing from the left
- Color: Dark blue
- Antarctic Circumpolar Deep Water
- Label: Antarctic Circumpolar Deep Water
- Position: Central area, above the mid-ocean ridge
- Color: Medium blue
- North Atlantic Deep Water
- Water Movement
- Upwelling and Downwelling
- Representation: Arrows indicating water flow
- Color: White arrows
- Position: Throughout the diagram, showing vertical movements
- Volume Front
- Label: Volume Front
- Position: Left side, near the surface
- Description: Boundary between water masses
- Differences
- Label: Differences
- Position: Near the surface, above the volume front
- Description: Indicates variations in water properties
- Upwelling and Downwelling
- Antarctic Continent
- Label: Antarctica
- Position: Right side of the diagram
- Color: Brown with a white top indicating ice
- Ocean Layers
- Color: Gradient of blue, darker at the bottom, lighter at the top
- Description: Represents depth and different water masses
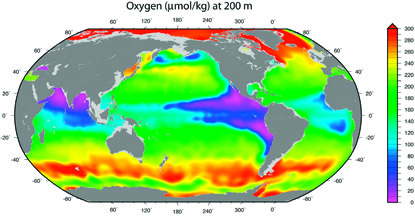
Another critical element for the health of the oceans is the dissolved oxygen content. Oxygen in the surface ocean is continuously added across the air-sea interface as well as by photosynthesis; it is used up in respiration by marine organisms and during the decay or oxidation of organic material that rains down in the ocean and is deposited on the ocean bottom. Most organisms require oxygen, thus its depletion has adverse effects for marine populations. Temperature also affects oxygen levels, as warm waters can hold less dissolved oxygen than cold waters. This relationship will have major implications for future oceans, as we will see.
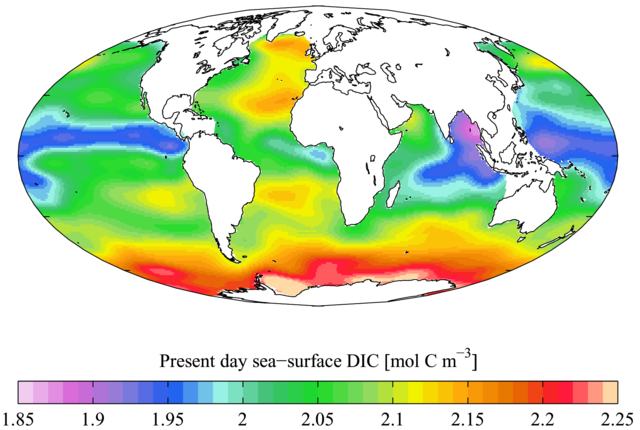
The final seawater property we will consider is the content of dissolved CO2. CO2 is nearly opposite to oxygen in many chemical and biological processes; it is used up by plankton during photosynthesis and replenished during respiration as well as during the oxidation of organic matter. As we will see later, CO2 content has importance for the study of deep-water aging.