Coral Bleaching and Calcification
Although, as we will see, ocean acidification is only just beginning to affect reef growth, there is already another very serious affliction in many reefs around the world---coral bleaching. One of the key changes in corals over the last few decades has been an increase in the frequency and severity of bleaching events. As we have discussed, shallow-water coral species live in symbiosis with algae called a dinoflagellate or Zooxanthellae whose colorful pigments give living corals such beautiful colors, purple, brown, green, yellow, and more. As discussed the symbiosis is key to the coral, the Zooxanthellae removed CO2 from the water, boosts the saturation, and facilitates calcification of the coral. When temperatures become too hot, however, the coral become physically stressed and expels the Zooxanthellae leading to a bleached white color.

Bleaching today is common when temperatures increase very slightly (by about 1°C) in summer months and thus is often an annual summer event. Bleaching is accompanied by slower growth and increased coral mortality. The response to bleaching differs considerably between different species. Some species are able to recover normal growth rates quite rapidly, whereas others are more vulnerable and unable to recover. Bleaching also changes the ecology of a reef, promoting the growth of algae that blanket corals, making the recovery of corals more difficult.
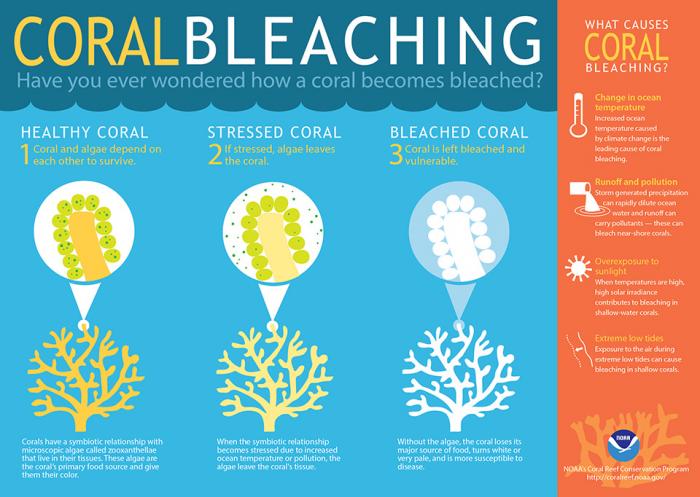
Click here to see a text description.
Coral Bleaching - Have you ever wondered how a coral becomes bleached?
- Healthy coral - coral and algae depend on each other to survive: Corals have a symbiotic relationship with microscopic algae called zooxanthellae that live in their tissues. These algae are the coral's primary food source and give them their color.
- Stressed coral - if stressed, algae leaves the coral: When the symbiotic relationship becomes stressed due to increased ocean temperature or pollution the algae leave the coral's tissues
- Bleached coral - coral is left bleached and vulnerable: without the algae, the coral loses its major source of food, turns white or very pale, and is more susceptible to disease
What causes coral bleaching?
- Change in ocean temperature: increased ocean temperature caused by climate change is the leading cause of coral bleaching
- Runoff and pollution: storm-generated precipitation can rapidly dilute ocean water and runoff can carry pollutants–these can bleach near-shore corals
- Overexposure to sunlight: when temperatures are high, solar irradiance contributes to bleaching in shallow-water corals
- Extreme low tides: exposure to the air during extreme low tides can cause bleaching in shallow corals
A major coral bleaching event took place in the Florida Keys in summer 2023 when water temperatures rose to 101 deg F and there was widespread bleaching. It is too soon to determine the long-term impacts of this bleaching on the reefs in the Keys but the images are dramatic and the future does not look promising.

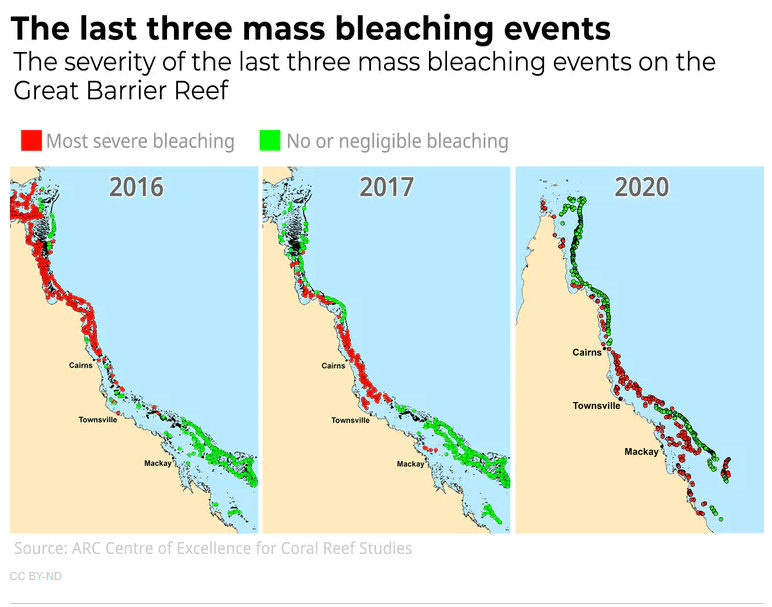
One of the most significant bleaching events took place in 2016 and 2017 in the Great Barrier Reef. This catastrophic event caused by warmer than average water led to 50% coral mortality in some areas. Bleaching was also extreme in 2020 and you can see from the maps below that it extended southward which in the southern hemisphere means it afflicted cooler waters. The extent of bleaching shocked ecologists and is clearly a window into the future. Now it seems every year, bleaching is a threat to the Great Barrier Reef, and with the warm El Nino conditions, the Austral summer of 2024-2025 may be the most severe bleaching event ever. One scientist has remarked that the reef recently looked like it had been "carpet-bombed".
Video: Australia’s Great Barrier Reef hit by mass coral bleaching (2:37)
SARAH CLARKE: These are some of the first images from the aerial assessment of coral bleaching in the Great Barrier Reef. Bleaching occurs when warmer ocean temperatures and pollution force coral to expel the algae that live in their tissues. The algae give the coral their range of colors. Scientists have declared 2024 a mass bleaching event.
DAVID WACHENFELD, AUSTRALIAN INSTITUTE OF MARINE SCIENCE: We call a mass coral bleaching when we see the majority of corals on many of the reefs that we survey across large areas of the in a marine park are bleaching, and that's the situation we're in at the moment. Coral bleaching is just a stress reaction from corals. You could compare it, for example, to heat stroke in a human.
SARAH CLARKE: Until now, coral in the Southern section of the 2000 km stretch of reef had largely escaped the most severe consequences of bleaching. But heat stress caused by an extreme marine heatwave over an extended period of time means some structures, hundreds of years old, have been severely damaged.
JODIE RUMMER, MARINE BIOLOGIST, AMES COOK UNIVERSITY: It's disturbing, to say the least. It's sad. It's infuriating, really, because we know the cause.
SARAH CLARKE: Marine biologist Jody Rummer has witnessed five mass beaching events on the Great Barrier Reef in the past eight years. She says ocean temperatures are increasing at a rate never seen before, and that's an ominous sign for the biggest coral system in the world.
JODIE RUMMER: We're seeing this back to back, year upon year. The reef needs many years to recover from these heat waves, and it's just not getting it.
SARAH CLARKE: What's happening here on the Great Barrier Reef is also happening on reefs around the world. Over the last 12 months, warming sea surface temperatures have caused bleaching events in the northern hemisphere, and the El Niño conditions in the Pacific have amplified the situation. Scientists say widespread bleaching in both the northern and southern hemisphere puts the world on the cusp of a global bleaching event.
MARK READ, GREAT BARRIER REEF MARINE PARK AUTORITY: We're certainly seeing a year-on-year increase in temperatures, and in many ways for us, that's meant that we've actually started to recognize that every year there's going to be some exhibition of thermal stress for species like corals.
SARAH CLARKE: Unesco's World Heritage Committee will decide if the Great Bay Reef should be listed as in danger when it meets a New Delhi in July. Sarah Clarke, Aljazeera, Townsville.
MARK READ: Make sure to subscribe to our channel to get the latest news from Aljazeera.
Deep Water Corals
Not all corals live in shallow water. A very diverse group of corals are able to thrive in the deep realms of the oceans. These corals lack Zooxanthellae and are able to calcify at cooler temperatures and lower levels of saturation than exist in the shallower waters. The number of known deep-water species has increased dramatically with new methods of exploring the deep oceans, including ROVs (remotely operated vehicles) and AUVs (autonomous underwater vehicles).
Examples of Deep Water Corals
These organisms exist in isolated patches at depths down to 2000 meters. In these conditions, the corals grow considerably slower than shallow-water reefs. They feed on zooplankton and in some cases use chemicals coming out of the sea floor for a source of nutrition. Like their shallow-water counterparts, deep coral reefs provide a habitat for a diverse array of creatures. However, at the same time, it is clear that deep corals are in a particularly precarious situation, as we will see later on.
Corals and Calcification
The growth rate of corals, known as the rate of calcification, corresponds more closely to aragonite Ω rather than any other variable. Cores of corals from the Great Barrier Reef indicate a 14% decrease in calcification rates from 1990 to 2005. Corals generally maintain a high Ω at the site of calcification, but with decreasing CO32-, precipitation of aragonite requires more energy, hence the decrease in calcification rate. Overall, the net calcification rates of reefs decrease to zero when the Ω of aragonite is <1, and thus we can predict that worldwide reefs will switch from actively growing via calcification to shrinking via dissolution when CO2 doubles to 560 ppm (the year 2050 in emission scenario A1B). The same logic leads to predictions of decreases in calcification rates of 10-50% by 2050. Experiments show that modern coral species have very different abilities to grow in water with lower saturation, with some species able to continue growing while others can't.
Experiments are one of the best ways to forecast the future, but they have significant limitations. In particular, it is impossible to replicate natural growing conditions in the lab, and further, experiments are conducted at intervals that are significantly shorter than the changes that are occurring in nature.
Field studies and experiments often show very different results. Strangely, field results indicate a much higher susceptibility of growth rates to decreasing levels of saturation than do laboratory experiments. These studies suggest that there is much left to learn about calcification and how it will change in the future. At the same time, it is clear that there is significant variability between species; some species having a much greater likelihood of survival in the future. Moreover, even within individual species, it is apparent that some reefs likely have the ability to survive better than others. Factors such as community composition, growth rate, nutrient levels, and local variations in seawater chemistry and sediment composition will also play a vital role in determining which reefs will survive.
Since the coralline algae are more tolerant of colder waters, they have a very widespread distribution in the oceans, extending from the tropics to polar regions. The algae are also able to use low light levels for photosynthesis and therefore live considerably deeper than most corals. Their distribution suggests that they have great potential to adapt to variable environments. As it turns out, however, they may be in a more precarious position than the corals.
The recent decrease in CO32- has also begun to lower calcification rates of the coralline algae. These species are composed of high Mg calcite, which is the most soluble form of CaCO3 (more so than low Mg calcite and aragonite), so they are particularly prone to ocean acidification. Experimental work confirms that calcification of the coralline algae is particularly sensitive to CO2 levels with growth rates slowing significantly, and actually, dissolution beginning at moderately high CO2 contents.
Perhaps the coralline algae will be the “canary in the coal mine” for the dissolution of all framework structures under rising CO2. For the corals, it is very apparent that the threat varies considerably from reef to reef. Deep-water corals are in particular jeopardy because calcification rates are lower in colder waters because saturation levels at these depths are lower, and because, in the future, saturation stands to decrease more rapidly in deep than in shallow waters. However, the fate of reefs in the tropics is also likely to vary significantly.
Future Predictions
Making predictions about the exact impact of ocean acidification in the future of coral reefs of all types is inherently difficult. Acidification will occur in parallel with other deleterious effects such as bleaching and sea level rise. Species that are most susceptible to the effects of bleaching may turn out to also be more susceptible to extinction. For example, by impacting zooxanthellae, bleaching impacts calcification and thus may exacerbate the impact of acidification. However, these same corals also have the ability to recover from bleaching events more quickly, possess shorter generation times, and thus may have the ability to evolve more rapidly to tolerate bleaching in the future. At the same time, nutrient levels in reef environments have and will continue to decrease as stratification of the upper ocean increases, and this will put considerable strain on future reefs. Temperatures, nutrient levels, local rates of sea level rise, and human activities are factors that will likely decide which reefs are the most vulnerable, possibly with acidification pushing the most threatened systems over the edge into extinction.
Probably the largest uncertainty with respect to the future of reefs is whether corals will be able to adapt to lower saturation levels, increased temperatures, and lower nutrient levels, and if so, how rapidly. As we have seen earlier, corals have shown great resilience in surviving numerous environmental threats in the past. So, why do reefs recovered after times of environmental stress in the past, at times when temperatures were even warmer than they are today, and CO2 levels higher, appear to be in such dangerous territory today? The answer to this question is that in the past, temperature and CO2 perturbations occurred slowly enough that ultimate increases in weathering (remember feedbacks we discussed in Module 3) essentially decreased the rate of CO2 addition and buffered the ocean with CO32- before extinction occurred. What concerns scientists is that warming today and CO2 addition are rapid enough that the weathering feedback will lag the decrease in saturation by several thousand years.
So, we are left with a lot of questions: will the rates of saturation decrease and temperature rise be too rapid for modern species to adapt? Will algae take over the niche of shallow-water corals and dominate the low pH oceans of the future? Or will a few species of coral and possibly coralline algae develop the ability to calcify rapidly enough to survive the current threats and take over the niche of species that do not? Will Zooxanthellae themselves be able to adapt and assist corals in calcifying? Finally, when will feedbacks, largely through weathering, come into play and make conditions more favorable for calcifying organisms? For some of these questions, it’s a matter of wait and see. However, ongoing research should shed light on others. For example, the genetics of coral populations are currently being explored to understand the ability of corals to adapt to environmental change.
At this stage, however, any outcome is possible for corals, ranging from complete extinction by late in the 21st century to the adaptation to a new set of environmental parameters. Ultimately, like many elements of the ecosystem, the fate of reefs may rest on how well we manage CO2 emissions in the future.
