Prioritize...
After you complete this section, you should be able to describe how falling precipitation and the presence of snow cover affect temperature. You should also be able to discuss the effects of urbanization on temperature (the "urban heat-island effect").
Read...
If clouds and invisible water vapor can affect temperature, it stands to reason that precipitation falling from the sky (rain, snow, etc.), can also have an impact. And, indeed, it does! We'll explore that in this section, along with how ground cover (particularly snow cover and urbanization) affect temperature.
Precipitation
When precipitation falls from the clouds, the overall impact is usually a cooling of the air. Why is that? When precipitation falls, some rain drops typically evaporate, meaning that they change phase from liquid into water vapor. The process of evaporation requires energy, however, and that energy comes from the surrounding air. As you know, temperature is a measure of energy, so as energy is used for evaporation, the air cools.
You've probably experienced this when you get out of a swimming pool on a hot day -- water drops on your skin begin to evaporate, which cools your skin. You've also noticed "evaporative cooling" in action if you've ever felt a rush of cool air before a shower or thunderstorm arrives. Note that sometimes you'll hear weathercasters say that the rain cools the air, but it's really not the rain causing the cooling, it's the evaporation of rain drops. Of course, if a lot of rain falls and the ground remains wet for a time, daytime temperatures may be suppressed somewhat, because some of the sun's energy will be used to evaporate liquid water on the surface, instead of being absorbed by the ground.
I should also point out that a similar cooling can occur when other types of precipitation fall, like snowflakes. Furthermore, when enough snow falls to cover the ground, the impacts on temperature can last for days! Let's explore.
Snow Cover
A covering of snow increases the albedo of the earth's surface, which means a greater fraction of incoming solar radiation gets reflected back to space. So, during the day, the reflection of some of the sun's energy back to space means less incoming solar radiation gets absorbed by the ground. Plus, a portion of the sun's energy absorbed by the snowpack goes toward melting the snow pack, as well as evaporating meltwater. Like evaporation, melting requires energy, too, and energy used for phase changes is not available to warm the surface.
As a result of these two factors, daytime air temperatures are sometimes noticeably lower than if there had been no snow cover. In the case shown below, chilly air overlying a snow pack slowed the advance of the leading edge of much warmer air from the south and west. Notice the dramatic temperature change from western North and South Dakota (where there was little or no snow cover) to Minnesota (where many areas had at least six inches of snow on the ground).
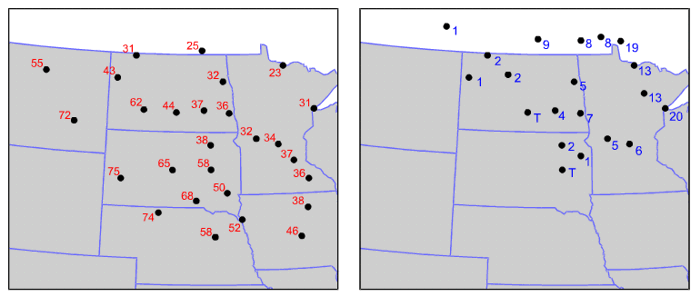
I point out that the effect of lower daytime temperatures in regions that have snow cover is much more pronounced over the Middle West, where there is a lack of forests. In the northeastern U.S., for example, dense coniferous and deciduous forests (which have low albedos) often mask the high albedo of snow on the ground, presenting a much darker appearance to space.
The effect of snowpack on nighttime temperatures can be also be dramatic. Snow readily absorbs and emits infrared radiation, and at night, snow emits infrared radiation with similar efficiency as bare ground. So why would there be any difference in nighttime temperatures over snow cover compared to bare ground? Good question. Snow is an effective insulator, which is why people stranded in the wilderness during winter will dig a protective burrow in the snow to stay warm. In general, a snowpack of two inches or more seals off heat energy stored in the ground, and with heat energy stored in the ground effectively sealed off, the temperature of the snow surface drops precipitously on a clear, calm night. In turn, overlying air temperatures plummet, setting the stage for very low temperatures around dawn.
The bottom line is that the presence of snow cover tends to make both days and nights colder than they otherwise would be (ignoring potential impacts from temperature advection, etc.), and the underlying reasons have to do with the all-important surface energy budget! But, snow isn't the only "ground covering" that impacts the surface energy budget. Urban development, such as paving streets, parking lots, and constructing buildings, changes the surface energy budget, too, so let's look at the effects of urbanization.
Urbanization
On a sunny day with light winds, temperatures in any big city can be several degrees higher than surrounding rural areas, as paved surfaces and buildings readily absorb solar radiation due to their relatively low albedos. Moreover, heat from cars, industry and other human activities accent the warmer city environment. Meanwhile, over the surrounding countryside, higher albedos typical of vegetation, along with evaporation of water released from trees and plants (which "sweat" in a process called "transpiration") serve to help to keep the daytime rural environment cooler than its urban counterpart. The fact that urban environments tend to be warmer than surrounding suburban and rural areas is often referred to as the urban heat-island effect.
Though noticeable differences between urban and rural temperatures exist during the day, the effect of the urban-heat island stands out more dramatically at night, particularly on clear, calm winter nights that follow sunny days. This is because sunshine warms the city more than surrounding rural areas, so city temperatures are already higher than rural temperatures as the sun starts to set. The temperature gap between the warmer city and the cooler countryside widens throughout the night as concrete and buildings, which absorbed plenty of solar radiation by day, slowly and steadily conduct, convect, and radiate energy to other parts of the urban environment. So, urbanized landscapes tend to be warmer than surrounding suburban and rural landscapes, both during the day and at night.
During summer, when dew points are high, urban nights can be oppressive as water vapor absorbs large amounts of infrared radiation emitted by buildings and concrete. This absorption causes the water vapor to warm and emit even more radiation, thereby slowing or essentially halting nocturnal cooling in the city. To see an example of the urban heat-island effect in summer, check out the image below, showing temperatures in and around Philadelphia, Pennsylvania around dawn on August 5, 2015.
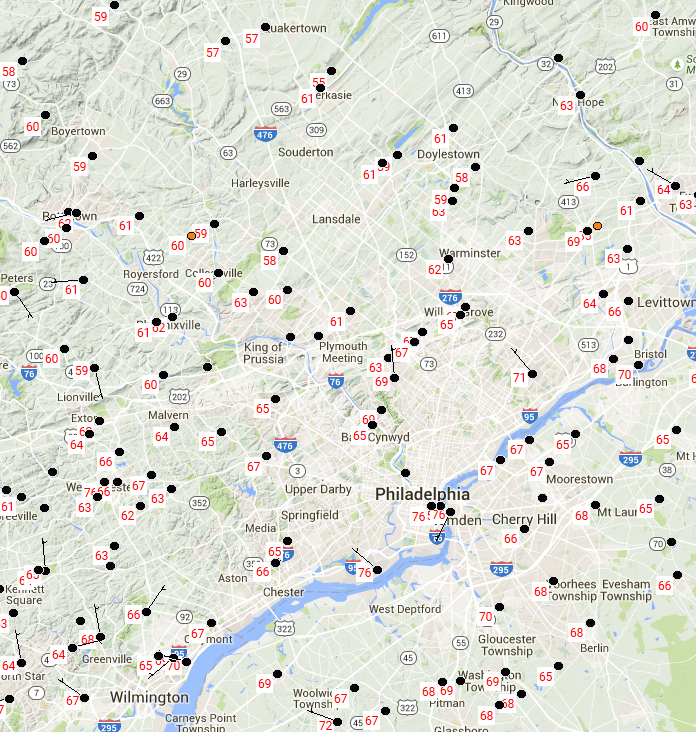
Note that temperatures in Center City Philadelphia and at the airport (just southwest of downtown along the Delaware River) were 76 degrees Fahrenheit. But, the surrounding suburbs were much cooler! Most suburbs were in the 60s, and a few toward the top of the map, such as Quakertown, were actually in the 50s. There was as much as a 20-degree difference between the suburbs and the city itself!
Clear, calm nights maximize the urban heat-island effect, and the above example was no exception (the black circles on the map are not station models, so they don't indicate cloud cover). Notably, wind reduces the temperature contrasts between urban and rural environments because as winds increase, the lower troposphere gets blended and mixed, thereby making surface temperatures more uniform (in much the same way that eddies help to diminish the effects of the nocturnal inversion). Widespread precipitation and its associated evaporative cooling also tend to equalize daytime temperatures between urban and rural environments.
That about wraps up our look at controllers of temperature. But, before we wrap up this lesson, I want to make a point about measuring temperature. As it turns out, it's not as simple as you might think, and not all measurements are trustworthy!